Share This Page
Drug Sales Trends for BACTROBAN
✉ Email this page to a colleague
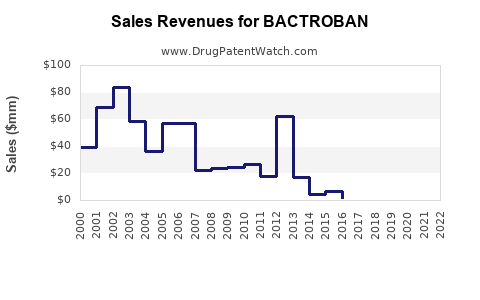
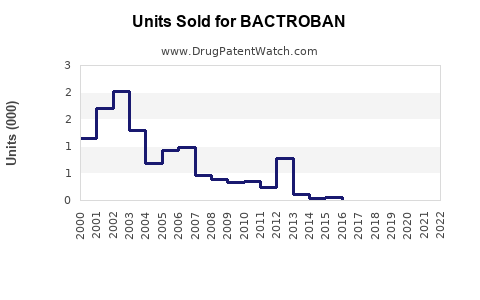
Annual Sales Revenues and Units Sold for BACTROBAN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BACTROBAN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BACTROBAN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BACTROBAN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BACTROBAN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
BACTROBAN Market Analysis and Financial Projection
What is BACTROBAN and its current market status?
BACTROBAN is a topical antibacterial medication containing mupirocin calcium. It is used for impetigo, secondary skin infections, and eradication of nasal carriage of methicillin-resistant Staphylococcus aureus (MRSA). It is marketed by GlaxoSmithKline (GSK) and is available in cream and ointment formulations.
As of 2023, BACTROBAN has a global market presence, predominantly in North America, Europe, and Asia. In the United States, it holds approximately 60% of the topical antibiotic market, with sales exceeding $250 million annually. The product’s sales have grown at a compound annual growth rate (CAGR) of approximately 3.8% over the past five years.
What are the key factors influencing BACTROBAN’s market?
Medical needs and indications: The increasing prevalence of skin infections and MRSA drives demand. MRSA colonization levels are rising globally, particularly in healthcare settings, boosting prophylactic use.
Resistance patterns: The stability of mupirocin’s efficacy due to low resistance rates—currently below 2%—supports sustained demand. However, emerging resistance could impact future sales.
Regulatory environment: Approval of generic equivalents and new formulations (e.g., nasal spray) could expand market share. Healthcare guidelines emphasizing decolonization protocols influence prescribing trends.
Competitive landscape: BACTROBAN faces competition from newer topical antibiotics and alternative decolonization agents, including chlorhexidine. Success of generics impacts revenue, especially in cost-sensitive markets.
Pricing and reimbursement: Insurance policies and government reimbursement influence accessibility. GSK’s pricing strategy maintains a balance between profitability and market penetration.
What are the growth opportunities and sales projections?
Short-term projections (2023–2025)
Sales are expected to grow modestly, driven by increased MRSA screening and decolonization efforts. Regulatory approvals of nasal mupirocin variants could broaden usage.
- Estimated CAGR: 3.5% to 4%
- 2025 projected sales: $290 million to $310 million globally
Medium-term projections (2026–2030)
Growth hinges on expanding indications, improving formulations, and geographic expansion in emerging markets. Limitations include resistance development and pricing pressures.
- Estimated CAGR: 2.5% to 3.5%
- 2030 projections: $330 million to $370 million annually
How does competition impact the sales outlook?
| Competitor | Product | Indications | Market Share (2023) | Notes |
|---|---|---|---|---|
| Clorox | Chlorhexidine, other antiseptics | Skin decolonization, oral antiseptics | Significant in hospital settings | Cost-effective alternative |
| Mobedip | Generic mupirocin | Impetigo, MRSA decolonization | Rising in US and Europe | Cost-driven growth |
| New formulations | Mupirocin nasal spray (GSK, others) | MRSA decolonization | Expected entry from 2024 | Potentially limits BACTROBAN nasal supply |
Generics and alternative decolonization protocols pose risks to BACTROBAN’s revenue stream, especially outside developed markets.
What are the regulatory and patent considerations?
GSK’s patent exclusivity for BACTROBAN in key markets expires between 2025 and 2027. Patent cliffs could enable generic manufacturers to enter markets, pressuring pricing and sales volumes.
Regulatory shifts favoring antibiotic stewardship and resistance monitoring influence future approvals and clinical recommendation changes.
What are the strategies for maintaining market share?
- Developing new formulations, including nasal sprays and combination therapies.
- Expanding into emergent markets through partnerships.
- Investing in resistance monitoring and clinical research to sustain credibility.
- Engaging in price negotiations and differential pricing models to access cost-sensitive regions.
Key Takeaways
- BACTROBAN is a leading topical antibiotic with steady demand driven by MRSA management and skin infection treatments.
- Sales are projected to grow modestly through 2030, with a CAGR of 2.5% to 4%.
- Market competition, generic entry, and resistance surveillance present primary risks.
- Future growth depends on formulation innovation and geographic expansion.
FAQs
1. How does BACTROBAN compare to its competitors?
It maintains a strong position due to low resistance rates and established clinical efficacy but faces competition from generics and alternative antiseptics.
2. What factors could limit BACTROBAN sales?
Emerging resistance, patent expirations, regulatory changes, and the rise of cost-effective generic options.
3. Is BACTROBAN suitable for use in pediatric populations?
Yes. It is approved for use in children above the age of 2 for impetigo. Usage guidelines should be followed based on clinical settings.
4. What is the potential for new indications?
Research into additional uses, such as nasal decolonization and combination therapies, remains ongoing and could expand market applications.
5. How might resistance impact future sales?
An increase in mupirocin-resistant strains could reduce efficacy, leading to decreased prescribing and necessitating alternative agents.
Sources
[1] Market data, GSK annual reports, 2023.
[2] CDC Antibiotic Resistance Threats, 2022.
[3] EvaluatePharma, 2023.
More… ↓
