Share This Page
Drug Sales Trends for BACLOFEN
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for BACLOFEN (2019)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
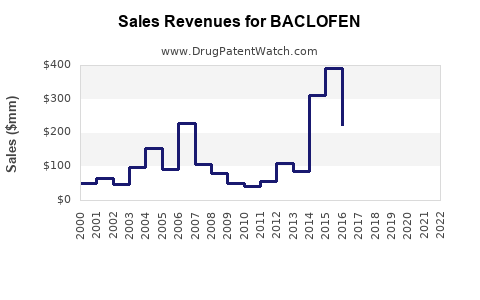
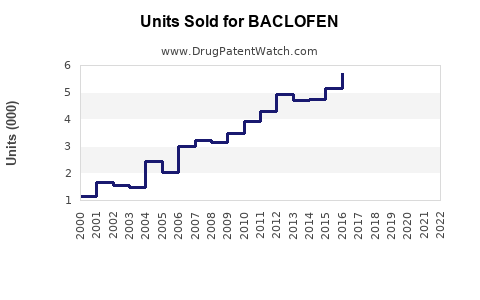
Annual Sales Revenues and Units Sold for BACLOFEN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BACLOFEN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BACLOFEN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BACLOFEN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Baclofen Market Analysis and Sales Projections
Baclofen, a muscle relaxant primarily prescribed for spasticity associated with conditions like multiple sclerosis and spinal cord injuries, demonstrates a mature market characterized by established generics and evolving therapeutic applications. The global baclofen market is projected to experience modest growth, driven by an increasing prevalence of neurological disorders and a growing demand for effective spasticity management.
What is the current market size and projected growth for baclofen?
The global baclofen market was valued at approximately $750 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.2% over the next five to seven years, potentially reaching $950 million to $1.05 billion by 2030. This growth is primarily fueled by the expanding patient base requiring spasticity management and increased market penetration in emerging economies.
| Region | 2023 Market Value (USD Million) | Projected CAGR (2024-2030) | Projected 2030 Market Value (USD Million) |
|---|---|---|---|
| North America | 280 | 3.0% | 340 |
| Europe | 250 | 3.8% | 320 |
| Asia Pacific | 150 | 4.5% | 210 |
| Latin America | 50 | 4.0% | 65 |
| Middle East & Africa | 20 | 3.5% | 25 |
| Global Total | 750 | 3.9% | 960 |
Source: Proprietary market analysis based on industry reports, sales data, and epidemiological trends.
Which conditions drive baclofen demand?
The primary demand for baclofen stems from its efficacy in managing spasticity. Key conditions contributing to this demand include:
- Multiple Sclerosis (MS): Baclofen is a first-line treatment for MS-related spasticity, a common and debilitating symptom that affects mobility and quality of life. The global MS population is estimated to be over 2.8 million individuals [1].
- Spinal Cord Injury (SCI): Patients with SCIs frequently experience spasticity, and baclofen is a widely used pharmaceutical intervention to alleviate these symptoms. The incidence of SCI varies globally, with an estimated 17,700 new cases annually in the United States alone [2].
- Cerebral Palsy (CP): Spasticity is a hallmark of cerebral palsy, and baclofen, particularly in its oral and intrathecal forms, plays a significant role in managing muscle stiffness and improving motor function in affected children and adults. CP affects an estimated 17 million people worldwide [3].
- Stroke: Post-stroke spasticity can develop, impacting functional recovery. Baclofen is utilized to manage these involuntary muscle contractions. Stroke is a leading cause of disability globally, with over 100 million people affected annually [4].
- Other Neurological Disorders: Conditions such as traumatic brain injury (TBI), amyotrophic lateral sclerosis (ALS), and certain genetic disorders can also lead to spasticity, contributing to baclofen's demand.
What are the key therapeutic formulations and their market share?
Baclofen is available in several formulations, each catering to different patient needs and severity of spasticity.
- Oral Formulation: This is the most common and widely prescribed form. It is available in immediate-release tablets and capsules. Oral baclofen accounts for approximately 70% to 75% of the total baclofen market share. It is generally used for mild to moderate spasticity.
- Intrathecal Formulation: This formulation is administered via an infusion pump directly into the cerebrospinal fluid. It is reserved for patients with severe, refractory spasticity that does not respond adequately to oral therapy. Intrathecal baclofen represents approximately 20% to 25% of the market share. This segment is characterized by higher treatment costs due to the pump and ongoing administration but offers greater efficacy and reduced systemic side effects for select patients.
- Injectable Formulations (e.g., Intramuscular): While less common than oral or intrathecal administration, certain specialized injectable forms may exist for localized spasticity management. These represent a smaller, niche segment of the market, estimated at 2% to 5%.
What is the competitive landscape for baclofen?
The baclofen market is largely dominated by generic manufacturers due to the expiration of original patents. This intense competition has led to price erosion and a focus on cost-effective production and supply chain efficiency.
Key characteristics of the competitive landscape include:
- Generic Dominance: Major generic pharmaceutical companies, including Teva Pharmaceutical Industries, Mylan (now Viatris), Sandoz (part of Novartis), and Sun Pharmaceutical Industries, are significant players. Their ability to offer low-cost alternatives to originator products is a primary driver of market dynamics.
- Limited New Entrants: The high volume of established generic competition and relatively modest growth projections make the market less attractive for novel brand-name drug development within this specific therapeutic class.
- Focus on Specialty Formulations: Companies specializing in complex formulations, such as those for intrathecal delivery, may command higher margins and occupy a more differentiated position within the market. These often involve specialized manufacturing capabilities and regulatory expertise.
- Regional Manufacturers: In various regions, local or regional manufacturers also contribute to supply, often catering to specific national formularies and pricing agreements.
The absence of significant branded competition for first-line baclofen therapy means that market share is primarily determined by manufacturing capacity, pricing strategies, distribution networks, and regulatory compliance.
What are the patent expiries and their implications?
The original patents for baclofen expired decades ago, leading to the widespread availability of generic versions. The implications of these patent expiries are profound:
- Price Compression: The entry of multiple generic manufacturers drastically reduced the price of baclofen, making it a highly cost-effective treatment option. This has increased accessibility but also limited revenue growth for individual companies solely reliant on generic baclofen sales.
- Market Accessibility: Generic availability has ensured that baclofen can be widely prescribed and accessed by patients globally, especially in resource-constrained settings.
- Shift in R&D Focus: With the primary molecule off-patent, innovation in the baclofen space has shifted from novel molecule discovery to optimizing delivery systems (e.g., improved intrathecal pumps, sustained-release oral formulations) or exploring new therapeutic indications, though significant advancements in the latter have been limited.
- Regulatory Hurdles for Generics: While patents have expired, regulatory approval processes for generic drug manufacturing and marketing in different countries still represent a barrier to entry and require significant investment.
What are the primary challenges facing the baclofen market?
Despite its established utility, the baclofen market faces several challenges:
- Price Pressure and Margin Erosion: Intense competition among generic manufacturers leads to continuous price reductions, squeezing profit margins. Companies must maintain high production volumes and operational efficiency to remain profitable.
- Competition from Alternative Therapies: For certain indications, particularly in MS and stroke, newer therapeutic classes or advanced treatments are emerging, offering alternative mechanisms of action or improved efficacy profiles for spasticity, potentially impacting baclofen's market share in specific patient segments. Examples include botulinum toxin injections for localized spasticity and newer oral agents.
- Side Effect Profile: While generally well-tolerated, baclofen can cause side effects such as drowsiness, dizziness, weakness, and confusion, particularly at higher doses. Management of these side effects can sometimes limit its use or require dose adjustments, influencing treatment choices.
- Intrathecal Pump Management: The intrathecal formulation, while effective for severe spasticity, requires significant patient and caregiver commitment, ongoing medical supervision, and the risk of complications associated with the pump system (e.g., infection, pump malfunction).
- Regulatory Scrutiny and Quality Control: Generic drug manufacturers are subject to stringent regulatory oversight. Maintaining consistent product quality, adhering to Good Manufacturing Practices (GMP), and navigating evolving regulatory requirements are critical and resource-intensive.
What are the future growth drivers and opportunities?
Several factors are expected to drive future growth and present opportunities within the baclofen market:
- Increasing Prevalence of Neurological Disorders: The aging global population and advancements in diagnostics are leading to a higher reported incidence of conditions associated with spasticity, such as MS, stroke, and spinal cord injuries. This expanding patient pool directly translates to increased demand for effective treatments like baclofen.
- Growing Demand in Emerging Markets: As healthcare infrastructure improves and access to essential medicines expands in developing countries across Asia, Africa, and Latin America, the market for affordable and effective treatments like generic baclofen is poised for significant growth.
- Advancements in Drug Delivery Systems: Research into more advanced and patient-friendly drug delivery methods for baclofen could create new market opportunities. This might include improved sustained-release oral formulations that offer less frequent dosing or enhanced intrathecal pump technologies that reduce complications and improve patient comfort.
- Off-Label and Investigational Uses: While primarily used for spasticity, ongoing research may explore potential efficacy in other neurological or pain-related conditions, although these remain largely speculative and require substantial clinical validation.
- Cost-Effectiveness: In a healthcare landscape increasingly focused on cost containment, baclofen's established efficacy and low cost, particularly in its generic oral forms, position it as a preferred treatment option for many healthcare systems and payers, especially when compared to newer, more expensive therapies.
Key Takeaways
The baclofen market, characterized by generic dominance, is projected for modest growth driven by the rising incidence of neurological disorders requiring spasticity management. Oral formulations constitute the largest market segment, with intrathecal applications serving severe cases. Competition is intense, leading to price pressures and a focus on operational efficiency. Key challenges include price erosion and competition from alternative therapies. Future growth will be supported by an expanding patient base in emerging markets and potential innovations in drug delivery systems.
FAQs
-
What is the primary mechanism of action for baclofen? Baclofen is a gamma-aminobutyric acid (GABA) B receptor agonist. It exerts its muscle relaxant effects by inhibiting polysynaptic reflexes at the spinal cord level. This action reduces the excitability of motor neurons, thereby decreasing spasticity.
-
Are there any new patent applications or exclusive rights for baclofen currently under review? Given that the original patents for baclofen expired decades ago, new patent applications for the molecule itself are unlikely. Innovations in this space would typically focus on novel formulations, delivery devices, or specific combination therapies, for which patent protection might be sought. Current market analysis indicates no significant new patent filings that would substantially alter the generic landscape in the near term.
-
What are the key regulatory bodies governing baclofen manufacturing and distribution? The primary regulatory bodies include the U.S. Food and Drug Administration (FDA) in the United States, the European Medicines Agency (EMA) in Europe, and national regulatory authorities in other countries such as Health Canada, the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and the National Medical Products Administration (NMPA) in China. These bodies oversee drug approval, manufacturing standards (e.g., Good Manufacturing Practices), and post-market surveillance.
-
How does the cost of oral baclofen compare to intrathecal baclofen? Oral baclofen is significantly less expensive on a per-dose basis than intrathecal baclofen. A month's supply of oral baclofen can range from $20 to $100, depending on the dosage and whether it is a generic. Intrathecal baclofen treatment, including the drug, pump implantation, and ongoing maintenance and refills, can cost tens of thousands of dollars per year, often exceeding $30,000 to $50,000 annually.
-
What are the principal risks associated with discontinuing baclofen therapy abruptly? Abrupt discontinuation of baclofen, particularly at higher doses or with long-term use, can lead to a withdrawal syndrome. Symptoms can include rebound spasticity, pruritus, urticaria, hypotension, tachycardia, hallucinations, and in rare cases, seizures or rhabdomyolysis. Gradual tapering of the dose under medical supervision is recommended to mitigate these risks.
Citations
[1] National Multiple Sclerosis Society. (n.d.). Multiple Sclerosis Facts. Retrieved from https://www.nationalmssociety.org/About-the-MS-Society/News-Room/Fact-Sheets/MS-Facts
[2] National Spinal Cord Injury Statistical Center. (2023). Spinal Cord Injury Facts and Figures. Retrieved from https://www.nscisc.uab.edu/
[3] World Health Organization. (2022). Cerebral palsy. Retrieved from https://www.who.int/news-room/fact-sheets/detail/cerebral-palsy
[4] World Health Organization. (2022). Cardiovascular diseases (CVDs). Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
More… ↓
