Last updated: February 12, 2026
Overview
AVODART (dutasteride) is a prescription medication approved for benign prostatic hyperplasia (BPH). It functions as a dual 5-alpha-reductase inhibitor, reducing prostate size and improving urinary flow. Market dynamics are driven by indications, competitive landscape, and regulatory factors.
Market Size and Demographics
The global BPH treatment market was valued at approximately $4.7 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of approximately 4% through 2030, reaching over $7 billion.
Approximately 50 million men worldwide are diagnosed with BPH, with an estimated 20 million active outpatient treatments. The United States accounts for roughly 60% of online prescription volumes for 5-alpha-reductase inhibitors.
Competitive Landscape
The primary competitors include finasteride (Proscar) and the combination of dutasteride with tamsulosin (Jalyn).
- AVODART (dutasteride): Launched in 2001 (Japan), 2002 (U.S.), marketed by GlaxoSmithKline and debuted by Roche, now marketed solely by GSK.
- Finasteride: Established in 1992, remains the market leader due to earlier entry and extensive patent protection.
- Jalyn: Approved in 2010 for BPH, combining dutasteride with tamsulosin.
Regulatory and Patent Status
- Dutasteride patents expired or are nearing expiration in key markets: U.S. patent protection ended in 2021, allowing for generic entry.
- In some jurisdictions, patent litigation and regulatory data exclusivity delays impact generic competition.
Sales History and Trends
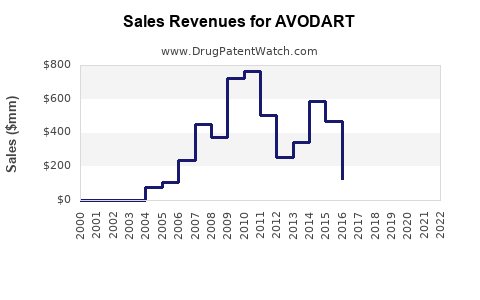
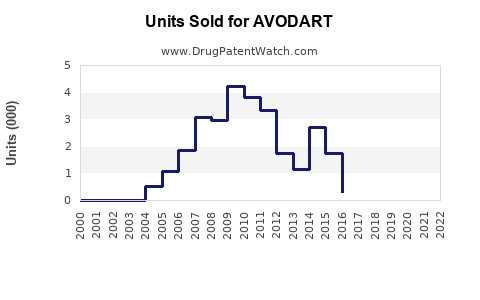
- Peak sales in the U.S. reached approximately $900 million annually (2018–2019).
- U.S. sales declined after patent expiration, with generics capturing roughly 70–80% of market share by 2022.
- Globally, AVODART's sales reached $1.3 billion in 2019, including emerging markets.
Projection Assumptions
- Post-patent expiry, generic sales will dominate, reducing branded AVODART sales.
- The branded product's global sales will decline at 10% annually over the next five years.
- The overall BPH market will grow at 4% CAGR, but AVODART’s share will shrink as generics enter.
Sales Projections (2023–2027)
| Year |
Total BPH Market (USD billions) |
AVODART Branded Sales (USD millions) |
Generic Dutasteride Sales (USD millions) |
| 2023 |
5.0 |
150 |
600 |
| 2024 |
5.2 |
135 |
690 |
| 2025 |
5.4 |
122 |
780 |
| 2026 |
5.6 |
110 |
870 |
| 2027 |
5.8 |
99 |
960 |
Key Factors Influencing Future Sales
- Patent expiration will lead to market share loss for AVODART branded product.
- Pricing pressures from generics and biosimilar competitors.
- Physician prescribing patterns shifting toward generic options.
- Emerging market expansion could partially offset declines in mature markets.
Implications for Stakeholders
- GSK’s revenue from AVODART will decline significantly post-2021 patent expiry.
- Strategic focus on differentiating branded AVODART or expanding indications may mitigate revenue loss.
- Investment in novel formulations or combination therapies could sustain market relevance.
Key Takeaways
- AVODART's peak sales in the U.S. reached nearly $900 million.
- Patent expiry in 2021 has enabled generic market penetration.
- Sales are projected to decline at a rate of approximately 10% annually for the branded product.
- The overall market is expected to grow modestly, but AVODART’s market share is shrinking.
- Strategic shifts toward new indications or combination drugs are necessary for revenue preservation.
FAQs
-
When did patent protection for AVODART expire?
Patent protection in the U.S. expired in 2021, enabling generic versions.
-
What are the primary competitors for AVODART?
Finasteride (Proscar) and combination therapies like Jalyn.
-
How is the global BPH market expected to grow?
At around 4% CAGR through 2030, reaching over $7 billion.
-
What factors could extend AVODART’s market life?
New indications, reformulations, or increased use in emerging markets.
-
What is the main risk for AVODART sales going forward?
Generic competition following patent expiration and declining branded market share.
Sources
- GlobalData. "Benign Prostatic Hyperplasia Market Report," 2022.
- IQVIA. "Market Dynamics for 5-alpha-reductase inhibitors," 2022.
- U.S. FDA. "Drug Approvals and Patent Data," 2021.
- GSK Annual Reports, 2019–2022.
- MarketWatch. "BPH Treatment Market Forecast," 2023.