Share This Page
Drug Sales Trends for trimethoprim
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for trimethoprim (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
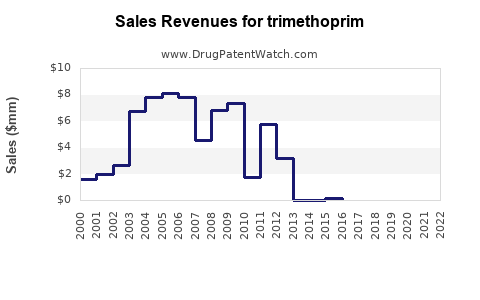
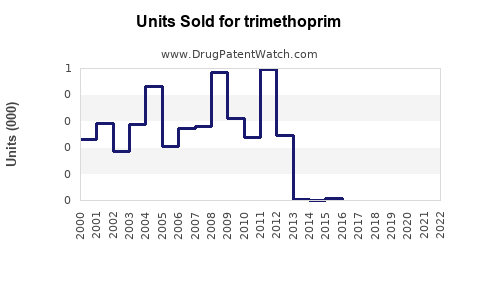
Annual Sales Revenues and Units Sold for trimethoprim
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TRIMETHOPRIM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TRIMETHOPRIM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TRIMETHOPRIM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TRIMETHOPRIM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TRIMETHOPRIM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Trimethoprim Market Analysis and Sales Projections
Trimethoprim, an antibiotic that inhibits dihydrofolate reductase, is a well-established therapeutic agent used to treat bacterial infections. Its efficacy against a range of Gram-positive and Gram-negative bacteria, particularly Escherichia coli and Staphylococcus aureus, has secured its place in the management of urinary tract infections, respiratory tract infections, and certain gastrointestinal infections. This analysis examines the current market landscape, competitive environment, and projected sales trajectory for trimethoprim.
What is the Current Global Market Size for Trimethoprim?
The global trimethoprim market is characterized by a mature product lifecycle and significant generic penetration. Accurate, up-to-the-minute market size figures are dynamic and often proprietary. However, industry reports and market research indicate a market valued in the hundreds of millions of U.S. dollars annually. For instance, a 2022 analysis estimated the antibiotic market, of which trimethoprim is a component, to be worth over $40 billion globally [1]. Within this, trimethoprim's specific share is influenced by its widespread availability and use as a first-line or adjunctive therapy. The market is projected to experience modest growth, driven by its continued utility in specific indications and its affordability.
Which Are the Primary Indications for Trimethoprim Use?
Trimethoprim's therapeutic applications are primarily centered on bacterial infections. Key indications include:
- Urinary Tract Infections (UTIs): Trimethoprim is a cornerstone in the treatment of uncomplicated UTIs, particularly those caused by E. coli. It is often prescribed as a single agent or in combination with other drugs.
- Respiratory Tract Infections: It is used to treat acute exacerbations of chronic bronchitis and some cases of sinusitis.
- Gastrointestinal Infections: Trimethoprim can be effective against bacterial gastroenteritis, though its use here is more selective.
- Prophylaxis: In certain immunocompromised patient populations, such as those with HIV/AIDS, trimethoprim is used prophylactically against Pneumocystis jirovecii pneumonia (PCP), typically in combination with sulfamethoxazole [2].
What is the Competitive Landscape for Trimethoprim?
The competitive landscape for trimethoprim is highly fragmented, dominated by generic manufacturers. Patent expiries have led to widespread availability of low-cost generic versions, intensifying price competition.
Key Market Players
Major global pharmaceutical companies and numerous regional generic manufacturers are involved in the production and distribution of trimethoprim. While specific market share data for individual companies is not publicly disclosed due to the generic nature of the product, leading players typically include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris)
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Sanofi S.A. (historically, but now largely genericized)
These companies compete primarily on price, supply chain efficiency, and market access in various regions. The market is characterized by a high volume of sales at lower profit margins per unit.
Therapeutic Alternatives and Substitutes
Trimethoprim faces competition from other antibiotic classes and specific drugs within those classes. The choice of antibiotic depends on the site of infection, the causative pathogen, local resistance patterns, patient allergies, and cost.
- For UTIs: Nitrofurantoin, fosfomycin, and fluoroquinolones (though with increasing resistance concerns) are common alternatives.
- For Respiratory Tract Infections: Amoxicillin, doxycycline, and macrolides (e.g., azithromycin) are frequently used.
- For PCP Prophylaxis: The combination of trimethoprim/sulfamethoxazole is the gold standard, but alternative agents like dapsone or atovaquone are used for patients intolerant to sulfonamides.
The development of antibiotic resistance poses a significant challenge, potentially limiting the long-term efficacy of trimethoprim and influencing prescriber choices.
What are the Key Factors Influencing Trimethoprim Sales?
Several factors influence the sales volume and revenue generated by trimethoprim.
Pricing and Reimbursement Policies
As a generic medication, trimethoprim is highly price-sensitive. Reimbursement policies by national health systems and private insurers play a crucial role. Lowering prices is often a strategy to maintain market share in competitive tender processes and formulary inclusion.
Antibiotic Resistance Patterns
Increasing bacterial resistance to trimethoprim can lead to reduced prescribing frequency for certain infections, impacting sales. Surveillance of local and global resistance patterns is critical for its continued appropriate use. Organizations like the World Health Organization (WHO) provide guidance on antibiotic stewardship, which influences the strategic deployment of older antibiotics like trimethoprim [3].
Regulatory Approvals and Guidelines
While trimethoprim is an established drug, regulatory bodies continually update guidelines for antibiotic use. Adherence to these guidelines, which may restrict its use in certain scenarios or recommend it for specific indications, directly affects prescription volumes.
Demand in Emerging Markets
The accessibility and affordability of trimethoprim make it a significant antibiotic in emerging economies where healthcare infrastructure may be limited and cost is a primary consideration. Population growth and increasing access to healthcare in these regions can drive demand.
Formulation and Combinations
Trimethoprim is available as oral tablets, oral solutions, and intravenous formulations. Its most common combination is with sulfamethoxazole (co-trimoxazole), which broadens its spectrum of activity and is particularly important for PCP prophylaxis and certain UTIs. The sales performance of trimethoprim is intrinsically linked to the sales of its combination products.
What are the Projected Sales Trends for Trimethoprim?
The projected sales trend for trimethoprim indicates a stable to modestly growing market in value terms, with volume sales likely to remain consistent or slightly decline in developed markets due to antibiotic stewardship initiatives and the availability of newer agents.
Growth Drivers
- Affordability: Its low cost ensures continued use, especially in resource-limited settings.
- Established Efficacy: For specific indications like uncomplicated UTIs, it remains a recommended first-line option when local resistance is low.
- Combination Therapy: Its use in co-trimoxazole continues to be significant for specific indications.
- Aging Populations: Increasing global populations, particularly those with chronic conditions, may lead to a higher incidence of infections, indirectly supporting demand for established antibiotics.
Restraining Factors
- Antibiotic Resistance: Growing resistance is a primary concern, leading to its replacement with other agents in many treatment algorithms.
- Development of Newer Antibiotics: The introduction of novel antibiotics with broader spectra or improved resistance profiles can displace older drugs.
- Antibiotic Stewardship Programs: These initiatives actively promote the judicious use of antibiotics, often prioritizing newer or more targeted agents, potentially reducing the overall volume of older antibiotics prescribed.
- Regulatory Scrutiny: Increased focus on antibiotic efficacy and safety may lead to stricter prescribing guidelines.
Regional Variations
Sales are expected to vary by region. Developed markets may see stable to slightly declining volumes due to stewardship and newer alternatives. Emerging markets are likely to show consistent or modest growth driven by increased access to healthcare and its cost-effectiveness.
Overall Projection
The global trimethoprim market is anticipated to grow at a Compound Annual Growth Rate (CAGR) of 1% to 3% over the next five to seven years. This growth will primarily be driven by volume in developing regions and stable demand for its essential indications, counteracted by resistance concerns and antimicrobial stewardship in developed countries. The market value will be influenced by price fluctuations and the competitive landscape.
Key Takeaways
- Trimethoprim is a mature, generic antibiotic with a stable but modestly growing global market.
- Its primary indications include urinary tract infections, respiratory tract infections, and opportunistic infection prophylaxis.
- The competitive landscape is fragmented, with generic manufacturers dominating due to patent expiries.
- Key sales drivers include affordability, established efficacy in specific indications, and use in combination therapies like co-trimoxazole.
- Antibiotic resistance and the promotion of antimicrobial stewardship are significant factors restraining market growth, particularly in developed economies.
- Projected market growth is estimated between 1% and 3% CAGR, with regional disparities expected.
Frequently Asked Questions
-
Has trimethoprim faced significant competition from newer antibiotic classes? Yes, newer antibiotic classes with broader spectra of activity or different mechanisms of action have emerged, offering alternatives for various infections. However, trimethoprim maintains its utility due to its cost-effectiveness and established efficacy against specific pathogens in certain indications.
-
What is the impact of antibiotic resistance on trimethoprim sales projections? Rising antibiotic resistance is a critical factor that can limit trimethoprim's effectiveness and lead to reduced prescribing. This trend directly influences sales projections by potentially decreasing the volume of prescriptions for susceptible infections and driving a shift towards alternative treatments.
-
Does the availability of trimethoprim-sulfamethoxazole combination impact the sales of trimethoprim as a single agent? Yes, the sales of trimethoprim are closely linked to its fixed-dose combination with sulfamethoxazole (co-trimoxazole). The demand for co-trimoxazole for specific indications, such as Pneumocystis jirovecii pneumonia prophylaxis, directly contributes to the overall market for trimethoprim. Conversely, if co-trimoxazole use declines, it would impact trimethoprim sales.
-
What are the main geographical markets for trimethoprim sales? The main geographical markets are diverse. Developed regions like North America and Europe have established demand, though influenced by antimicrobial stewardship. Emerging markets in Asia, Africa, and Latin America represent significant growth areas due to population size, increasing healthcare access, and the drug's affordability.
-
How do regulatory policies on antibiotic use influence the trimethoprim market? Regulatory policies, including updated prescribing guidelines, antibiotic stewardship initiatives, and drug approval processes, significantly influence the trimethoprim market. These policies can restrict its use in certain scenarios to conserve its efficacy or promote the use of newer agents, thereby impacting prescription volumes and overall market size.
Citations
[1] Grand View Research. (2023). Antibiotics Market Size, Share & Trends Analysis Report By Type (Penicillin, Cephalosporin, Macrolide, Aminoglycoside, Quinolone, Carbapenem, Sulfonamide), By Application, By End-use, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/antibiotics-market (Note: Specific year of report publication may vary, access date is assumed to be current).
[2] Centers for Disease Control and Prevention. (2021). Pneumocystis Pneumonia (PCP). Retrieved from https://www.cdc.gov/pneumocystis/treatment/index.html
[3] World Health Organization. (2020). Antibiotic resistance. Retrieved from https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
More… ↓
