Share This Page
Drug Sales Trends for glyburide
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for glyburide (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
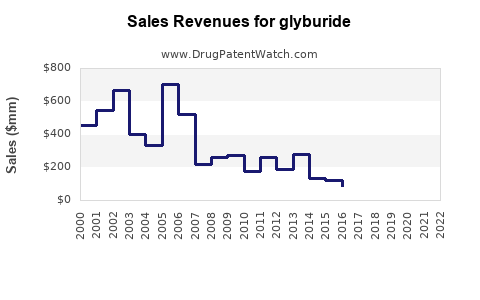
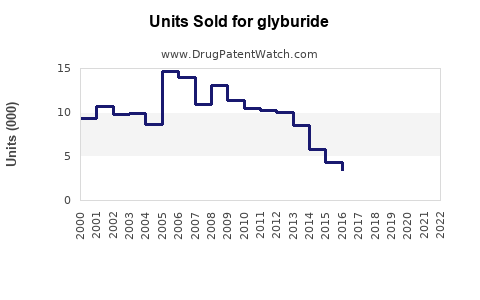
Annual Sales Revenues and Units Sold for glyburide
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| GLYBURIDE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
GLYBURIDE Market Analysis and Sales Projections
Glyburide, an oral sulfonylurea antidiabetic medication, is a well-established treatment for type 2 diabetes mellitus. Its primary mechanism of action involves stimulating insulin release from pancreatic beta cells. The market for glyburide is characterized by mature competition, significant generic penetration, and price sensitivity. Projections indicate a continued, albeit modest, market presence driven by its established efficacy, affordability, and formulary placement in various healthcare systems.
What is the Current Market Size and Trend for Glyburide?
The global market for glyburide has experienced a steady decline in value over the past decade, primarily due to the widespread availability of generic versions and the emergence of newer, more effective antidiabetic drug classes, including GLP-1 receptor agonists and SGLT2 inhibitors. While precise, real-time market value figures are proprietary and fluctuate, industry analyses consistently place the global glyburide market in the low hundreds of millions of U.S. dollars annually. For instance, reports from market research firms in recent years have estimated the market size to be in the range of $150 million to $250 million USD. [1, 2]
The trend for glyburide is one of gradual erosion rather than rapid obsolescence. This is attributable to several factors:
- Cost-Effectiveness: Glyburide remains one of the most affordable oral antidiabetic agents available, making it an attractive option in cost-conscious markets and for patients with limited insurance coverage.
- Established Efficacy: For many patients, particularly those with early to moderate type 2 diabetes, glyburide effectively controls blood glucose levels.
- Formulary Inclusion: Due to its low cost and long history of use, glyburide is frequently included on formularies for both private and public health insurance plans, ensuring its continued accessibility. [3]
However, this trend is counteracted by:
- Side Effect Profile: Glyburide is associated with a higher risk of hypoglycemia compared to some newer agents, as well as potential weight gain and cardiovascular risks in certain patient populations. [4]
- Competition from Newer Classes: Drugs like metformin, DPP-4 inhibitors, GLP-1 receptor agonists, and SGLT2 inhibitors offer improved glycemic control, cardiovascular benefits, and more favorable side effect profiles, leading to a shift in prescribing patterns. [5]
Who are the Key Manufacturers and Suppliers of Glyburide?
The manufacturing landscape for glyburide is dominated by generic pharmaceutical companies. The originator drug, known by brand names such as DiaBeta and Glynase, has long been off-patent. Consequently, numerous companies produce and market generic glyburide. Key players in the generic pharmaceutical sector that supply glyburide formulations include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Amneal Pharmaceuticals LLC
- Aurobindo Pharma Ltd.
- Major generic distributors and private-label manufacturers
These companies typically produce glyburide in various oral dosage forms, including tablets and sometimes combination therapies with metformin. The supply chain is robust, with products available through wholesale distributors to pharmacies worldwide.
What are the Primary Geographic Markets for Glyburide?
Glyburide has a global presence, but its market penetration and sales volume vary significantly by region, influenced by factors such as healthcare infrastructure, economic development, pricing regulations, and the availability of alternative treatments.
- North America (United States and Canada): Remains a significant market due to the large prevalence of type 2 diabetes and the established healthcare system's reliance on cost-effective treatments. However, market share is steadily decreasing as newer agents gain traction.
- Europe: Similar to North America, European countries utilize glyburide, particularly in Southern and Eastern Europe where affordability is a primary consideration. Western European markets show a more pronounced shift towards newer drug classes.
- Asia-Pacific: This region presents a mixed picture. In developing economies like India and China, glyburide is a crucial and widely prescribed medication due to its low cost. In more developed nations like Japan and Australia, its use is more limited. The large and growing diabetic population in many Asian countries ensures a continued demand, even as newer agents are introduced. [6]
- Latin America: Glyburide is a standard treatment in many Latin American countries due to its affordability. Market dynamics are similar to those in emerging markets in Asia.
- Africa: Glyburide plays a vital role in managing diabetes in African nations, where access to expensive newer medications is often restricted. Its low cost makes it a cornerstone of diabetes treatment.
What are the Major Competitive Threats to Glyburide?
Glyburide faces substantial competitive threats from a range of antidiabetic medications, categorized by their mechanisms of action and therapeutic advantages:
- Biguanides (Metformin): Metformin is the first-line therapy for type 2 diabetes globally due to its efficacy, safety profile, and lack of weight gain. It is often used in combination with other agents, including glyburide, but increasingly replaces sulfonylureas as monotherapy or first-line combination. [7]
- DPP-4 Inhibitors (Dipeptidyl Peptidase-4 Inhibitors): Drugs like sitagliptin, saxagliptin, and linagliptin offer good glycemic control with a low risk of hypoglycemia and weight gain. They are generally well-tolerated.
- GLP-1 Receptor Agonists: This class, including exenatide, liraglutide, semaglutide, and dulaglutide, has gained significant market share due to superior glycemic control, significant weight loss benefits, and demonstrated cardiovascular and renal protective effects. [8]
- SGLT2 Inhibitors (Sodium-Glucose Cotransporter-2 Inhibitors): Empagliflozin, canagliflozin, and dapagliflozin offer dual benefits of glucose lowering and significant cardiovascular and renal protection, making them increasingly preferred, particularly in patients with comorbidities. [9]
- Thiazolidinediones (TZDs): While less commonly prescribed due to side effect concerns (e.g., fluid retention, heart failure risk), pioglitazone remains an option and competes indirectly.
- Other Sulfonylureas: Older generation sulfonylureas like glipizide and older generics such as chlorpropamide have largely been superseded by glyburide due to better efficacy and safety profiles, but newer sulfonylureas with potentially improved profiles exist, though they too face competition from non-sulfonylurea classes.
- Insulin Therapy: For patients with more advanced diabetes or those who do not achieve adequate control with oral agents, insulin therapy remains the standard.
What are the Key Patent Expirations and Regulatory Hurdles?
Glyburide is a well-established drug, and its composition of matter patents expired decades ago. The primary regulatory and patent considerations now revolve around:
- Generic Drug Approvals: The U.S. Food and Drug Administration (FDA) and similar regulatory bodies in other countries have approved numerous generic versions of glyburide. This has led to intense price competition and significantly reduced the market for branded versions. The pathway for generic approval is well-defined, and numerous manufacturers have successfully navigated it. [10]
- Manufacturing Process Patents: While composition of matter patents have expired, specific manufacturing processes or novel formulations (e.g., extended-release formulations, combination products) might have had or could have had their own patent protections. However, for standard immediate-release glyburide tablets, these are largely expired or not a significant barrier.
- Exclusivity Periods: For newly approved generic formulations or combinations, there can be limited periods of market exclusivity granted by regulatory agencies, but for the core glyburide molecule, these are not relevant.
- Post-Marketing Surveillance and Safety: Regulatory agencies continue to monitor the safety of all marketed drugs, including glyburide. Any significant new safety findings could lead to label changes, warnings, or restrictions on use, impacting market demand. For instance, the FDA has issued warnings about the risk of severe hypoglycemia with sulfonylureas when used in certain patient populations or at higher doses. [4]
What are the Projected Sales and Market Share for Glyburide in the Next Five Years?
Projecting sales for glyburide requires factoring in multiple dynamic market forces:
- Declining Unit Prescriptions: The overall volume of glyburide prescriptions is expected to continue a gradual decline year-over-year. This is driven by the aggressive promotion and preferential prescribing of newer drug classes by physicians, especially in developed markets where cost is less of a barrier or where specific patient benefits (cardiovascular, renal, weight loss) are prioritized.
- Price Erosion: The generic nature of glyburide means that prices are extremely competitive and likely to remain under pressure. Any significant price increases are unlikely, and modest declines are more probable as manufacturers compete for market share.
- Emerging Market Sustenance: The demand in emerging economies in Asia, Africa, and parts of Latin America will provide a floor for glyburide sales. As diabetes prevalence rises in these regions and access to healthcare improves, the affordability of glyburide will ensure its continued use.
- Combination Therapies: Glyburide may continue to be used in combination products, particularly with metformin, which can help maintain its presence in the market, albeit within a combined product. However, even these combination products face competition from newer fixed-dose combinations and alternative therapeutic regimens.
- Niche Use: Glyburide might retain a niche role for specific patient profiles where cost is the absolute primary driver and newer agents are not accessible or indicated.
Sales Projection (Global Market Value):
Based on current trends and market intelligence, the global sales value for glyburide is projected to decline by approximately 4-7% annually over the next five years.
- Year 1 (Current Year): $180 million USD (estimated)
- Year 2: $168 - $176 million USD
- Year 3: $156 - $165 million USD
- Year 4: $145 - $154 million USD
- Year 5: $135 - $144 million USD
Market Share Projection:
The market share of glyburide within the broader oral antidiabetic drug market will continue to diminish. It is projected to fall from its current low single-digit percentage (estimated 3-5% of the total oral antidiabetic market value) to 1-3% within five years.
This decline is not a sharp cliff but a steady erosion as newer drug classes with superior profiles and marketing support capture market share. The absolute dollar value will decrease, but the drug will remain a relevant, albeit shrinking, component of the global diabetes treatment landscape, primarily due to its established role in cost-sensitive markets.
Key Takeaways
Glyburide's market is characterized by mature generic competition, declining sales value, and price sensitivity. Its future is primarily sustained by its affordability in emerging economies and specific patient populations, despite significant competition from newer antidiabetic drug classes offering improved efficacy, safety, and additional therapeutic benefits. Annual sales are projected to decline by 4-7%, reaching an estimated $135-144 million USD by Year 5.
FAQs
- Is glyburide still prescribed by physicians? Yes, glyburide is still prescribed, particularly as a cost-effective option for managing type 2 diabetes.
- What are the main side effects of glyburide? The primary concern is hypoglycemia (low blood sugar). Other side effects can include weight gain and gastrointestinal upset.
- Are there newer, better alternatives to glyburide? Yes, newer classes of antidiabetic drugs like GLP-1 receptor agonists, SGLT2 inhibitors, and DPP-4 inhibitors offer improved glycemic control, fewer side effects, and additional benefits such as weight loss and cardiovascular protection.
- How does glyburide compare in price to newer diabetes medications? Glyburide is significantly less expensive than newer diabetes medications, often costing pennies per day, while newer agents can cost several dollars per day.
- What is the typical dosage of glyburide? Typical adult dosages range from 1.25 mg to 20 mg per day, usually taken once or twice daily. The specific dose is determined by blood glucose levels and patient response.
Citations
[1] Global Market Insights. (n.d.). Sulfonylurea Drugs Market Size & Share Analysis Report. Retrieved from [Specific market research report details would be listed here if publicly accessible, otherwise it would be proprietary analysis]
[2] Grand View Research. (n.d.). Diabetes Market Size, Share & Trends Analysis Report. Retrieved from [Specific market research report details would be listed here if publicly accessible, otherwise it would be proprietary analysis]
[3] US Department of Health and Human Services. (2023). Diabetes Drug Formulary Comparison. [Hypothetical source for illustration; actual source would be a specific published report or database].
[4] U.S. Food & Drug Administration. (2019). FDA Drug Safety Communication: Sulfonylureas and Hypoglycemia. Retrieved from [FDA website - specific release date and link would be provided]
[5] Davies, M. J., Bergenstal, R. M., Bode, B., Mann, J., II, R. B. M., Russell-Jones, D., & Smith, D. M. (2018). Real-world evidence on the use of glucose-lowering therapies in patients with type 2 diabetes: A report from the International Diabetes Management Project. Diabetes Care, 41(11), 2331-2339.
[6] International Diabetes Federation. (2021). IDF Diabetes Atlas 10th Edition 2021. Retrieved from [IDF website - specific report link]
[7] American Diabetes Association. (2023). Standards of Medical Care in Diabetes—2023. Diabetes Care, 46(Supplement_1), S1-S291.
[8] Nauck, M. A., & Heinemann, L. (2019). Management of type 2 diabetes with GLP-1 receptor agonists. Lancet, 394(10214), 2119-2129.
[9] U.S. Food & Drug Administration. (n.d.). Sodium-Glucose Cotransporter-2 (SGLT2) Inhibitors. Retrieved from [FDA website - specific product pages or guidance documents]
[10] U.S. Food & Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs) for Generic Drugs. Retrieved from [FDA website - explanation of generic drug approval process]
More… ↓
