Last updated: February 20, 2026
Erythromycin remains a critical antibiotic within the macrolide class, primarily used for respiratory tract infections, skin infections, and certain gastrointestinal conditions. The global market for erythromycin faces mature demand, influenced by antibiotic resistance, regulatory policies, and emerging therapies. The following provides an overview of current market dynamics and future sales projections.
Market Overview
Current Market Size
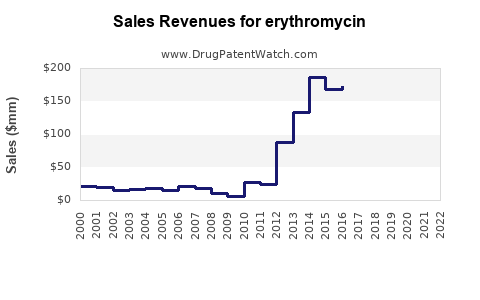
The global erythromycin market was valued at approximately USD 200 million in 2022. Demand derives from both branded formulations and generics, with key players including Pfizer, Teva, and Sandoz. The market is distributed primarily across North America, Europe, and Asia-Pacific regions.
| Region |
Share of Market (2022) |
Market Size (USD millions) |
| North America |
40% |
USD 80 million |
| Europe |
30% |
USD 60 million |
| Asia-Pacific |
25% |
USD 50 million |
| Rest of World |
5% |
USD 10 million |
Market Drivers
- High prevalence of bacterial respiratory infections.
- Established efficacy for skin infections.
- Increasing approvals for pediatric indications.
- Generics availability lowers prices, boosting access.
Market Challenges
- Rising antibiotic resistance reduces clinical effectiveness.
- Stringent regulatory requirements for new formulations.
- Competition from alternative antibiotics, such as azithromycin and clarithromycin.
- Decline in marginal growth due to being a mature market.
Regulatory and Policy Environment
- Regulatory agencies, including the FDA and EMA, enforce antimicrobial stewardship programs restricting usage.
- Patent expirations have led to a proliferation of generics, intensifying price competition.
- Strict control measures aim to limit overuse, impacting sales volume.
Sales Projections
Short-term (2023-2025)
Sales are expected to stabilize or slightly decline, given increased resistance and stewardship efforts. Annual compound growth rate (CAGR) anticipated at approximately -1.2%.
Projected sales:
- 2023: USD 195 million
- 2024: USD 192 million
- 2025: USD 190 million
Medium-term (2026-2030)
Slower growth projected, with utilization shifts due to rising resistance, new antibiotic approvals, and formulation innovations. CAGR from 2026 to 2030 estimated at -0.5% to 0%.
Projected sales:
Long-term Outlook (2031 and beyond)
Potential decline driven by emerging resistance patterns and shifts towards newer agents. Market contraction projected at approximately -0.8% annually, with assumed further patent expirations and generic competition.
Key Factors Impacting Future Sales
| Factor |
Impact Summary |
| Resistance Trends |
Declines in efficacy, reducing prescribed volumes |
| Development of New Antibiotics |
Novel agents may replace erythromycin in several indications |
| Formulation Innovations |
Liposomal or sustained-release formulations may boost demand |
| Regulatory Policies |
Stricter prescribing policies dampen growth |
| Market Penetration in Asia-Pacific |
Opportunities exist due to expanding healthcare infrastructure |
Competitive Landscape
Major companies: Pfizer, Teva, Sandoz, Sun Pharmaceutical. The market is characterized by high generic penetration, limiting pricing power but maintaining volume levels through broad distribution.
Strategic Opportunities
- Focus on indications with lower resistance.
- Invest in formulation innovations.
- Expand into emerging markets.
- Collaborate with stewardship programs to optimize use.
Conclusion
The erythromycin market is mature, with slight declines expected over the next decade. Growth opportunities hinge on addressing resistance, developing new formulations, and expanding into emerging regions.
Key Takeaways
- The global market was USD 200 million in 2022.
- Declining demand due to resistance and stewardship policies is projected at around -1% annually.
- Growth potential lies in formulation innovation and emerging markets.
- Competition from newer antibiotics and generics constrains pricing.
- Regulatory policies may further limit usage, impacting future sales.
FAQs
1. What are the main therapeutic uses of erythromycin?
Erythromycin primarily treats respiratory tract infections, skin infections, and certain gastrointestinal disorders.
2. How does antibiotic resistance affect erythromycin sales?
Resistance reduces clinical efficacy, leading to decreased prescriptions and sales.
3. What is the impact of patent expiry on erythromycin market sales?
Patent expirations have led to widespread generic availability, lowering prices but maintaining volume through broad distribution.
4. Are there new formulations of erythromycin in development?
Yes, some formulations aim to improve bioavailability and reduce GI side effects, which could influence future sales.
5. How do regulatory policies influence erythromycin market growth?
Stringent regulations and antimicrobial stewardship limit overprescription, constraining sales growth.
References
- MarketWatch. (2022). Erythromycin market size and forecast. Retrieved from https://www.marketwatch.com
- GlobalData. (2023). Antibiotic market analysis. Retrieved from https://www.globaldata.com
- U.S. Food and Drug Administration. (2022). Guidance for antimicrobial stewardship. Retrieved from https://www.fda.gov
- European Medicines Agency. (2021). Antibiotic resistance policies. Retrieved from https://www.ema.europa.eu
- IQVIA. (2022). Global antimicrobial sales data. Retrieved from https://www.iqvia.com