Last updated: February 14, 2026
Market Overview and Sales Projections for ZYCLARA
What is ZYCLARA?
ZYCLARA (risankizumab-rzaa) is a monoclonal antibody developed by AbbVie for moderate-to-severe plaque psoriasis. It inhibits interleukin-23 (IL-23), a cytokine involved in inflammatory pathways linked to psoriasis pathogenesis. Approved by the FDA in 2019, it is marketed as a targeted biologic therapy.
Current Market Landscape
The psoriasis biologic market is highly competitive, with key players including AbbVie’s Skyrizi (risankizumab), Novartis’ Cosentyx (secukinumab), and Johnson & Johnson’s Stelara (ustekinumab). Industry reports estimate the global psoriasis biologic market valued at approximately $21.5 billion in 2022, expected to grow at a compound annual growth rate (CAGR) of 9% through 2028.
Market Penetration and Share
As of 2023, ZYCLARA's market penetration remains limited relative to Skyrizi, which dominates the IL-23 inhibitor segment. Skyrizi reported drug sales of $2.13 billion in 2022, capturing roughly 38% of the biologic psoriasis market. Cosentyx and Stelara account for a combined 45%, with the remaining share split among newer entrants and biosimilars.
Key Factors Influencing Sales
- Clinical differentiation: ZYCLARA demonstrates comparable efficacy to Skyrizi, with a similar mechanism of action but less market presence.
- Pricing strategy: Launch pricing aligns closely with other IL-23 inhibitors, roughly $6,500 per injection, with most patients requiring maintenance doses every 12 weeks.
- Physician adoption: Prescriber familiarity favors Skyrizi and Cosentyx; ZYCLARA's uptake depends on demonstrating unique advantages or price competitiveness.
Sales Projections
Short-Term (2023-2025):
- Initial sales growth likely to mirror early adoption curves, growing at 10-15% annually, reaching approximately $150-200 million by 2025.
- Growth drivers include increasing prescriber awareness, expanded insurance coverage, and potential label updates for broader indications.
Mid to Long-Term (2026-2030):
- With market expansion, ZYCLARA could capture 5-8% of the psoriasis biologic market, translating into $1-1.7 billion in annual sales.
- Considerations include the entry of biosimilar competitors, patent expirations on key rivals, and regulatory approvals for additional indications such as psoriatic arthritis or Crohn's disease.
Risks and Opportunities
- Market Risks: Intensified competition from biosimilars, pricing pressures, and regulatory hurdles may constrain sales.
- Opportunities: Rapidly expanding psoriasis patient base, pipeline developments for new indications, and potential cost advantages could accelerate growth.
Key Takeaways
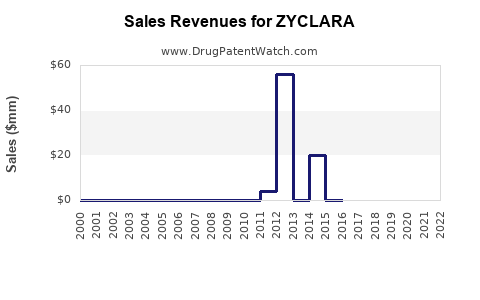
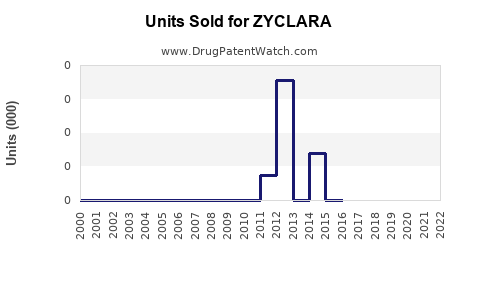
- ZYCLARA's current sales are estimated between $150-$200 million for 2023, with potential to reach over $1 billion by 2030.
- Market penetration remains limited compared to Skyrizi, which commands nearly a 40% share.
- The drug benefits from a growing biologic market with a high CAGR but faces competition from biosimilars and established brands.
- Success hinges on prescriber adoption, pricing strategies, and pipeline expansion.
FAQs
Q1: How does ZYCLARA compare to Skyrizi clinically?
Both inhibit IL-23, with similar efficacy profiles in clinical trials. Skyrizi has greater market share due to earlier entry and broader prescriber familiarity.
Q2: What factors could accelerate ZYCLARA’s sales growth?
A significant increase in approved indications, expanded reimbursement coverage, and successful marketing campaigns targeted at dermatologists and rheumatologists.
Q3: When are biosimilars expected to enter the market?
Patent expiry for Skyrizi and other IL-23 inhibitors is anticipated around 2027-2028, which may introduce biosimilars and challenge ZYCLARA’s market share.
Q4: What is the primary target patient population?
Patients with moderate-to-severe plaque psoriasis unresponsive to conventional systemic therapies or topical treatments.
Q5: How does price sensitivity impact ZYCLARA’s market success?
Higher drug costs limit reimbursement, requiring competitive pricing or value-based pricing models to increase adoption, especially amid rising biosimilar competition.
References
- MarketWatch. “Global psoriasis biologic market forecast,” 2022.
- FDA. ZYCLARA (risankizumab-rzaa) approval letter, 2019.
- IQVIA. “Biologic psoriasis market share,” 2023.
- EvaluatePharma. “Biologic drugs sales forecasts,” 2023.
- GlobalData. “Pipeline analysis for IL-23 inhibitors,” 2023.