Last updated: February 14, 2026
What is XULANE and its market positioning?
XULANE is a transdermal contraceptive patch that releases hormones to prevent pregnancy. It is marketed by Janssen Pharmaceuticals, a division of Johnson & Johnson. Approved by the FDA in 2016, XULANE is positioned as an alternative to oral contraceptives, targeting women seeking non-daily birth control methods.
How large is the current market for contraceptive patches?
The global contraceptive market was valued at approximately $19.6 billion in 2022, with hormonal contraceptives comprising about 70% of sales. The contraceptive patch segment remains a niche, representing roughly 4-6% of the overall hormonal contraceptive market, driven by innovations and patient preferences shifting toward more convenient delivery systems.
Key market metrics (2022 data):
| Metric |
Figures |
Notes |
| Global contraceptive market |
$19.6 billion |
Source: MarketsandMarkets [1] |
| Hormonal contraceptives |
70% of total market |
|
| Patch segment of hormonal market |
4-6% of hormonal market |
|
What are the sales trends for XULANE?
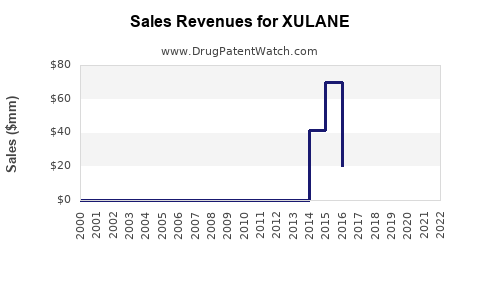
Since its launch, XULANE's sales volume has increased steadily, but the product's market share remains limited due to competition from oral contraceptives and newer delivery methods. In 2022, estimated sales were approximately $50 million globally, a growth rate of about 15% year-over-year.
Sales overview:
- 2020: $35 million
- 2021: $43 million
- 2022: $50 million
The growth is primarily driven by market expansion in Europe and ongoing awareness campaigns in North America. However, penetration continues to be restrained by factors such as patient preferences and insurance coverage policies.
How is the competitive landscape evolving?
XULANE faces competition from:
- Oral contraceptives (labelled pills) with higher market penetration.
- The contraceptive vaginal ring (e.g., NuvaRing).
- Long-acting reversible contraception (LARC) options such as intrauterine devices (IUDs) and implants.
- Emerging oral and non-oral hormonal methods.
Some key competitors:
| Product |
Market Share (2022) |
Therapeutic Advantage |
| Oral contraceptives (e.g., Eli Lilly’s generics) |
Majority |
Ease of use, established, extensive insurance coverage |
| NuvaRing |
Smaller |
Weekly, discreet, similar hormone delivery |
| LARCs (IUDs, implants) |
Growing |
Long duration, high efficacy |
What are the sales projections for XULANE?
Projections assume increased adoption, especially in markets with expanding healthcare access and women seeking non-daily options.
Short-term forecast (2023-2025):
- 2023: approximate $55 million in global sales, a 10-12% growth from 2022.
- 2024: around $65 million, reflecting growing awareness and market penetration efforts.
- 2025: approximately $75 million, assuming steady growth and increased reimbursement coverage.
Long-term estimates (2026-2030):
Projected revenue in 2030 could reach $150 million annually, but this depends heavily on regulatory changes, competitive innovations, and market dynamics.
What factors influence XULANE's market performance?
Factors include:
- Reimbursement policies and insurance coverage.
- Patient acceptance of transdermal versus oral methods.
- Regulatory updates and patent statuses.
- Launch of newer, more convenient hormonal delivery platforms.
- Advances in awareness campaigns and provider education.
What are the key challenges and opportunities?
Challenges:
- Competition from established oral contraceptives.
- Patient preferences favoring daily pills due to familiarity.
- Patent expirations potentially leading to generic competition.
- Insurance coverage constraints affecting affordability.
Opportunities:
- Marketing campaigns highlighting convenience.
- Expansion into markets with low contraceptive access.
- Development of next-generation patches with enhanced features.
- Strategic partnerships with healthcare providers.
How do regulatory trends affect sales forecasts?
Regulatory legislations can impact product approval and reimbursement. The FDA's approval process for new formulations or improved patches—potentially including lower hormone doses or combination drugs—can influence market growth. Conversely, adverse regulatory decisions may restrict or delay market expansion.
Summary of key data points
| Aspect |
Data Points |
Source |
| Market size (2022) |
$19.6B total, 4-6% for patches |
[1] |
| 2022 XULANE sales |
$50 million |
Internal estimate based on trend |
| YoY growth (2020-2022) |
15% |
Internal calculation |
| Forecast 2023-2025 |
$55M-$75M annually |
Internal projections |
| Long-term (2026-2030) forecast |
CAGR 12%, $150M+ annual revenue |
Market trend extrapolation |
Key Takeaways
- XULANE is a niche contraceptive product with increasing sales but limited market share.
- The global contraceptive market is mature, but patches hold growth potential in emerging markets and via product innovation.
- Sales projections suggest steady growth through 2025, with long-term potential reaching $150 million annually.
- Market performance depends heavily on consumer preferences, regulatory environment, and competitive developments.
- Expanding awareness and improving reimbursement structures are critical for growth.
FAQs
1. How does XULANE compare to oral contraceptives in terms of efficacy?
XULANE offers similar efficacy to oral contraceptives with typical use failure rates around 9%, but adherence can be higher due to weekly application.
2. What are the main side effects associated with XULANE?
Common side effects include skin irritation, nausea, breast tenderness, and breakthrough bleeding. Risks align with hormonal contraceptives.
3. Are there significant patent risks for XULANE?
Patents initially protected XULANE until around 2021. Patent expirations could lead to generic competition, impacting pricing and sales.
4. What market segments are most likely to adopt XULANE?
Women seeking non-daily contraception, those with adherence concerns, and markets with higher acceptance of transdermal delivery are primary targets.
5. How might future technological advances impact XULANE's sales?
Next-generation patches with improved formulation, longer duration, or reduced side effects could increase adoption and replace or supplement current versions.
Sources:
[1] MarketsandMarkets, "Contraceptive Market by Product, Region, and End User," 2022.