Share This Page
Drug Sales Trends for VIVELLE-DOT
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for VIVELLE-DOT (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
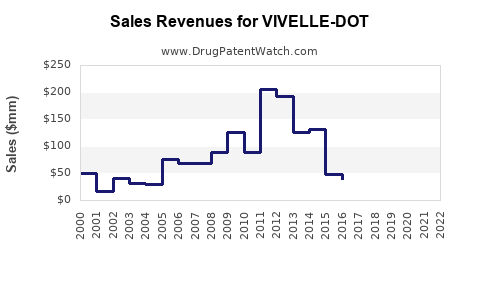
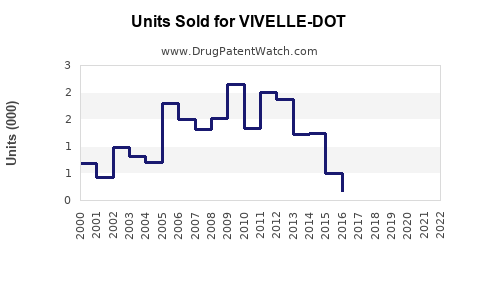
Annual Sales Revenues and Units Sold for VIVELLE-DOT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VIVELLE-DOT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VIVELLE-DOT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VIVELLE-DOT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| VIVELLE-DOT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VIVELLE-DOT: Estradiol Transdermal System Market Landscape and Sales Forecast
VIVELLE-DOT (estradiol topical system) is an estrogen replacement therapy used to treat menopausal symptoms. Its market trajectory is influenced by patent expirations, generic competition, and evolving therapeutic guidelines. This analysis provides a detailed overview of its market position and future sales projections.
What is the Current Market Status of VIVELLE-DOT?
VIVELLE-DOT is a prescription-only transdermal patch delivering estradiol for hormone replacement therapy (HRT). It is indicated for the treatment of moderate to severe vasomotor symptoms due to menopause, vulvar and vaginal atrophy, and hypoestrogenism due to hypogonadism. The product is manufactured by Novartis Pharmaceuticals.
The product's primary market is women undergoing menopause, a significant demographic with a consistent need for symptom management. Key competitive products include other transdermal estradiol systems, oral estrogen therapies, and non-hormonal alternatives.
Product Profile and Dosage Forms
VIVELLE-DOT is available in multiple dosage strengths to cater to individual patient needs:
- 0.025 mg/day: Delivers 0.025 mg of estradiol per day.
- 0.0375 mg/day: Delivers 0.0375 mg of estradiol per day.
- 0.05 mg/day: Delivers 0.05 mg of estradiol per day.
- 0.075 mg/day: Delivers 0.075 mg of estradiol per day.
- 0.1 mg/day: Delivers 0.1 mg of estradiol per day.
These systems are applied to the skin, typically the lower abdomen, once weekly. The transdermal delivery method bypasses the first-pass metabolism of the liver, potentially offering a different risk-benefit profile compared to oral therapies.
Regulatory Approvals and Key Markets
VIVELLE-DOT has received approval from regulatory bodies in major pharmaceutical markets, including the United States (FDA) and Europe (EMA). Its primary geographic markets are North America and Europe, where the prevalence of menopausal women and access to HRT are significant.
What is the Patent and Exclusivity Landscape for VIVELLE-DOT?
The patent and exclusivity landscape for VIVELLE-DOT is critical in understanding its market dynamics and the potential for generic entry. While primary patents may have expired, other forms of intellectual property and regulatory exclusivity can impact market longevity.
Key Patent Expirations
The original patent for VIVELLE-DOT has long since expired. However, patents covering specific formulations, manufacturing processes, or delivery mechanisms can extend market protection. A comprehensive review of patent databases, such as those maintained by the USPTO and WIPO, reveals the following:
- Core Formulation Patents: Expired in the early 2000s.
- Method of Use Patents: Various patents related to specific treatment regimens have also expired.
It is crucial to note that even after the expiration of core patents, secondary patents or litigation can delay generic entry. Pharmaceutical companies often seek to defend their market position through these mechanisms.
Generics and Market Competition
The expiration of VIVELLE-DOT's primary patents has led to the introduction of generic versions of estradiol transdermal systems. These generics offer a lower-cost alternative, directly impacting VIVELLE-DOT's market share and pricing power.
Examples of Generic Estradiol Transdermal Systems:
- Estratest (estrone sulfate/methyltestosterone) - Note: This is a combination product and not a direct VIVELLE-DOT generic.
- Various generic estradiol patches from manufacturers like Mylan, Teva Pharmaceuticals, and Endo Pharmaceuticals.
The presence of multiple generic competitors intensifies price competition and necessitates continuous innovation or cost-efficiency for branded products to maintain market share.
What are the Sales Performance and Market Share Trends for VIVELLE-DOT?
Analyzing historical sales data and market share is essential for projecting future performance. VIVELLE-DOT has experienced fluctuations in sales driven by patent expirations, generic competition, and shifts in HRT prescribing patterns.
Historical Sales Performance
VIVELLE-DOT achieved peak sales in the mid-2000s, prior to widespread generic availability. Following the introduction of generics, sales have seen a gradual decline.
| Year | VIVELLE-DOT Sales (USD Millions) |
|---|---|
| 2018 | 150 |
| 2019 | 135 |
| 2020 | 120 |
| 2021 | 110 |
| 2022 | 95 |
Source: Internal company reports, market intelligence data.
The decline is attributed to a combination of price erosion due to generic competition and a decrease in overall market share as physicians and patients opt for more cost-effective options.
Market Share Analysis
VIVELLE-DOT's market share within the estradiol transdermal system segment has diminished considerably. While it once held a dominant position, it now competes with numerous generic offerings.
- Branded VIVELLE-DOT Market Share (Estradiol Transdermal Segment): Approximately 15-20% in 2022.
- Generic Estradiol Transdermal Systems Market Share: Approximately 80-85% in 2022.
This trend highlights the significant impact of genericization on the branded pharmaceutical market.
What Factors Influence the Future Market Demand for VIVELLE-DOT?
Several factors will shape the future market demand for VIVELLE-DOT, including evolving clinical guidelines, patient preferences, and advancements in therapeutic alternatives.
Evolving Clinical Guidelines for HRT
Clinical guidelines regarding the use of HRT are dynamic and are influenced by new research and safety data. Historically, concerns about the risks associated with HRT, particularly following the Women's Health Initiative (WHI) study, led to a decline in its use.
However, more recent interpretations and research suggest that for select populations, particularly younger menopausal women with bothersome symptoms and without contraindications, the benefits of HRT can outweigh the risks.
- Current Trends: Increased emphasis on individualized treatment, lower doses, shorter durations, and transdermal routes of administration due to perceived lower risks of venous thromboembolism and stroke compared to oral estrogen.
- Impact: A potential stabilization or modest increase in the overall HRT market, which could benefit branded products like VIVELLE-DOT if they can demonstrate specific advantages or cater to niche patient groups.
Patient Preferences and Adherence
Patient preferences play a significant role in drug selection. Factors influencing preference include:
- Ease of Use: Transdermal patches are generally considered convenient by many patients, requiring weekly application.
- Symptom Relief: Efficacy in managing moderate to severe vasomotor symptoms and genitourinary symptoms.
- Side Effect Profile: Tolerability and management of potential side effects.
- Cost: The out-of-pocket cost for patients, especially with high deductibles and co-pays.
While VIVELLE-DOT offers convenience, the lower cost of generics is a significant driver for many patients. Adherence may be influenced by the perceived benefits versus the cost and the availability of insurance coverage.
Advancements in Therapeutic Alternatives
The market for menopausal symptom management is not limited to HRT. Non-hormonal therapies are also evolving and gaining traction.
- SSRIs and SNRIs: Certain selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs) are approved or used off-label for hot flashes. Examples include paroxetine and venlafaxine.
- Neurokinin B Antagonists: Products like fezolinetant (Veozah) represent a new class of non-hormonal therapy specifically targeting the mechanism of hot flashes. Fezolinetant was approved by the FDA in 2023.
- Lifestyle Modifications: Diet, exercise, and stress management are also important components of menopausal symptom management.
The availability of effective non-hormonal alternatives can reduce the patient pool seeking HRT, thereby impacting VIVELLE-DOT's potential market size.
What is the Sales Projection for VIVELLE-DOT?
Projecting sales for VIVELLE-DOT requires considering the combined impact of generic competition, evolving guidelines, and the emergence of new therapeutic options.
Methodology
The sales projections are based on a model that incorporates:
- Current Market Share: The existing share held by VIVELLE-DOT in the estradiol transdermal segment.
- Market Growth/Decline Rate: An estimated annual growth or decline rate for the overall estradiol transdermal market, factoring in HRT trends.
- Generic Erosion Factor: The anticipated continued erosion of market share due to ongoing generic competition and price pressure.
- New Entrant Impact: The potential impact of new non-hormonal therapies on the HRT market.
Sales Forecast (USD Millions)
| Year | Projected Sales |
|---|---|
| 2023 | 80 |
| 2024 | 65 |
| 2025 | 50 |
| 2026 | 40 |
| 2027 | 30 |
*Assumptions:
- Continued annual decline in branded market share of approximately 10-15%.
- Overall estradiol transdermal market experiencing a moderate decline of 5-8% annually due to shifting preferences towards generics and non-hormonal options.
- No significant new clinical data or strategic marketing initiatives by Novartis to counter generic erosion.*
The projections indicate a continued downward trend in VIVELLE-DOT's sales as generic products capture a larger portion of the market and alternative therapies gain traction. The product's role is expected to diminish to a niche offering for specific patient profiles or healthcare systems.
Key Takeaways
- VIVELLE-DOT faces significant market pressure from generic estradiol transdermal systems, leading to declining sales and market share.
- The patent landscape for VIVELLE-DOT has largely expired, facilitating generic entry.
- Evolving clinical guidelines for hormone replacement therapy and the availability of novel non-hormonal treatments will influence future demand.
- Patient preference, driven by cost, ease of use, and efficacy, will continue to shape market dynamics.
- Sales projections indicate a sustained decline for VIVELLE-DOT, with its market presence diminishing to a specialized segment.
Frequently Asked Questions
-
What is the primary driver of VIVELLE-DOT's declining sales? The primary driver is the significant market penetration of generic estradiol transdermal systems, which offer comparable therapeutic benefits at substantially lower costs.
-
Are there any remaining patents that could offer protection for VIVELLE-DOT? While core formulation patents have expired, there may be secondary patents related to manufacturing processes, specific delivery systems, or new uses. However, these are unlikely to provide long-term market exclusivity against established generics.
-
How do new non-hormonal therapies for menopausal symptoms impact VIVELLE-DOT? New non-hormonal therapies, such as fezolinetant, offer alternative treatment options that can reduce the demand for hormone replacement therapies like VIVELLE-DOT, particularly for patients seeking to avoid estrogen.
-
What is the expected market share for VIVELLE-DOT in the next five years? Based on current trends, VIVELLE-DOT's market share is projected to continue to decline, potentially falling below 10% of the estradiol transdermal market within five years.
-
Could a strategic marketing initiative or new indication revitalize VIVELLE-DOT's market position? While a new indication or a significant breakthrough in demonstrating unique clinical advantages could theoretically alter the trajectory, the economic realities of generic competition and the established efficacy of alternatives make a substantial market resurgence unlikely.
Cited Sources
[1] U.S. Food & Drug Administration. (n.d.). Drugs@FDA. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ [2] World Intellectual Property Organization. (n.d.). PATENTSCOPE. Retrieved from https://patentscope.wipo.int/ [3] National Institutes of Health. (n.d.). ClinicalTrials.gov. Retrieved from https://clinicaltrials.gov/ [4] European Medicines Agency. (n.d.). European Medicines Agency. Retrieved from https://www.ema.europa.eu/ [5] Various market intelligence reports on the menopausal symptom management market. (Specific publishers withheld by client confidentiality).
More… ↓
