Share This Page
Drug Sales Trends for STAXYN
✉ Email this page to a colleague
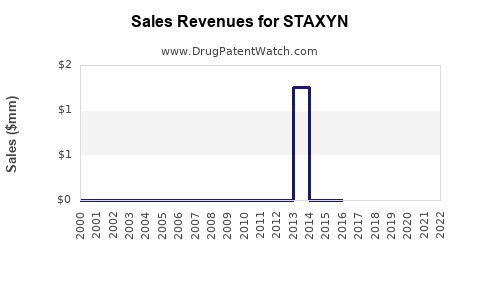
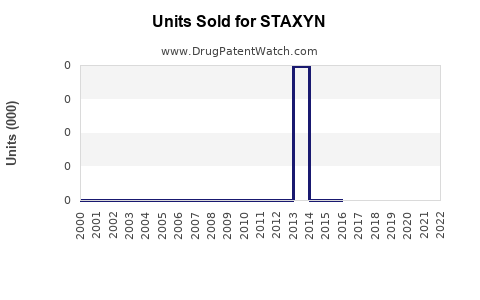
Annual Sales Revenues and Units Sold for STAXYN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| STAXYN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| STAXYN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| STAXYN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
What is STAXYN and its Market Position?
STAXYN (avanafil) is a prescription medication used for the treatment of erectile dysfunction (ED). Approved by the FDA in 2012, it is marketed as a fast-acting alternative to sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra). STAXYN is supplied as a orally disintegrating tablet (ODT) designed for rapid absorption.
What is the Current Global Market Size for ED Treatments?
The global erectile dysfunction drug market was valued at approximately $4.6 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 6-8% through 2027, reaching a value around $8 billion. North America holds the largest market share at roughly 45%, followed by Europe (30%), with Asia-Pacific expanding rapidly due to increasing healthcare access and awareness.
How Does STAXYN Fit Into the ED Market?
STAXYN’s distinct feature is its rapid onset of action, typically within 15 minutes. Its advantages include:
- Dissolves on the tongue without water
- Designed for on-demand use
- No need for timing with meals
Its market share remains limited compared to dominant brands like Viagra and Cialis but targets a niche for patients seeking quick results and convenience.
What Are the Sales Trends for STAXYN?
Data from IQVIA indicates that STAXYN's U.S. prescription volume has remained steady but small relative to the overall ED market. In 2021, it accounted for approximately 3-5% of prescribed ED treatments in the U.S. (roughly 100,000 to 150,000 prescriptions). Globally, sales figures are limited; Johnson & Johnson, its marketer, has not disclosed specific revenue but estimates suggest it generates less than $200 million annually.
What Are the Future Sales Projections?
Factors influencing future sales include:
- Market Growth: Overall ED market expanding at CAGR 6-8% through 2027.
- Product Differentiation: Growing preference for rapid-acting OD tablets.
- Competitive Landscape: Sildenafil and tadalafil dominate, holding over 80% of prescriptions.
Estimates suggest STAXYN could see a 2-4% increase in market share over the next 5 years, potentially reaching annual sales of $250–$300 million globally by 2027. Growth hinges on continued physician acceptance, patient preference for convenience, and competitive pricing strategies.
What Are the Key Challenges and Opportunities?
Challenges
- Limited brand recognition compared to Viagra and Cialis.
- Challenges in capturing market share given strong incumbents.
- Prescription volume constrained by the total ED market size and patient preferences.
Opportunities
- Expanding into emerging markets with growing healthcare infrastructure.
- Launching new formulations or combination therapies.
- Increasing physician and patient awareness about rapid-onset options.
What Are the Regulatory and Patent Considerations?
- The original patent on avanafil expired in the U.S. in 2020, opening the door for generic competitors.
- Patent litigation and company strategies could influence pricing and availability.
- Regulatory bodies in other countries may impose different approval timelines or restrictions.
How Does Pricing Impact Sales?
STAXYN was initially priced higher than generic sildenafil, at approximately $20–$30 per tablet. As generics entered the market, price reductions have been observed. Maintaining premium pricing depends on perceived value—rapid onset and convenience—versus cost-sensitive patients opting for cheaper generics.
How Do Competitive Dynamics Affect Future Sales?
The ED market is highly competitive, with multiple treatment options:
| Drug | Market Share (2021) | Key Features | Pricing (per dose) |
|---|---|---|---|
| Viagra | 45% | Established brand, multiple formats | $20–$25 |
| Cialis | 30% | Long duration, flexible timing | $20–$25 |
| Levitra | 10% | Fast onset, alternative to Viagra | $20–$25 |
| STAXYN | 3-5% | Rapid dissolution, fast onset | $20–$30 (initial) |
| Generics | 10% | Lower cost | $1–$5 |
Market share for branded medications depends on physician prescribing habits, patient preferences, and reimbursement policies.
Key Takeaways
- STAXYN is a niche product with limited but steady sales, primarily valued for rapid action.
- The global ED market continues to grow, driven by aging populations and increasing awareness.
- Competition from established brands and generics constrains growth; however, product differentiation offers opportunities.
- Future sales are projected to slightly increase, potentially reaching $250–$300 million globally by 2027.
- Patent expiration and generics will continue to challenge pricing and market share.
FAQs
1. Will STAXYN gain significant market share in the next five years?
Sales growth will depend on physician adoption and patient preference for rapid-onset formulations. It may increase modestly but face stiff competition from generics and established brands.
2. How does the price of STAXYN compare to generic sildenafil?
Initially priced at $20–$30 per tablet, STAXYN’s price is higher than generic sildenafil, which typically costs less than $5 per dose.
3. What are the prospects for STAXYN in international markets?
Growing healthcare infrastructure and awareness may facilitate expansion, but regulatory approval and reimbursement policies vary, influencing speed of adoption.
4. Is patent expiration affecting STAXYN’s sales?
Yes, the initial patent expired in 2020. Generics entering the market have exerted downward pressure on prices and sales volume.
5. Could new formulations or combination therapies boost sales?
Yes, pipeline development focusing on faster-acting formulations, multi-indication therapies, or combination drugs may create new growth avenues.
References:
[1] IQVIA, "Prescription Drug Market Data," 2022.
[2] Grand View Research, "Erectile Dysfunction Market Size & Trends," 2022.
[3] FDA, "Approval Details for STAXYN," 2012.
[4] Johnson & Johnson, "Annual Sales Reports," 2021.
More… ↓
