Share This Page
Drug Sales Trends for PRIMIDONE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PRIMIDONE (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
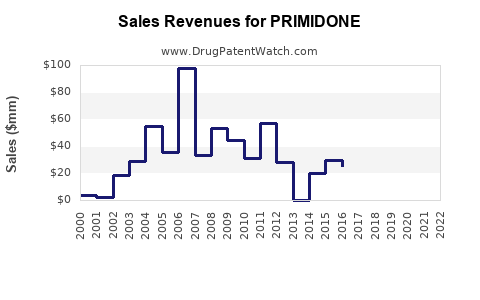
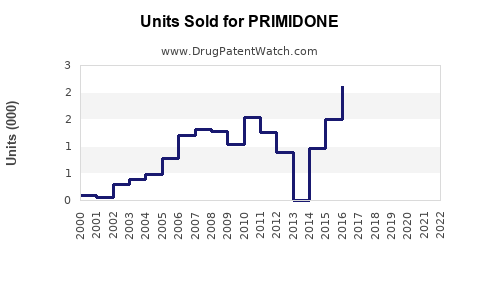
Annual Sales Revenues and Units Sold for PRIMIDONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PRIMIDONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PRIMIDONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PRIMIDONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PRIMIDONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PRIMIDONE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PRIMIDONE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
PRIMIDONE Market Analysis and Sales Projections
Primidone, an antiepileptic drug (AED), is primarily used to treat partial seizures and generalized tonic-clonic seizures. Its market performance is influenced by factors including epilepsy prevalence, competitor drug development, and generic availability. This analysis projects the market trajectory for Primidone based on current trends and patent landscapes.
What is Primidone's Current Market Position?
Primidone, marketed historically under brand names like Mysoline, is a barbiturate derivative with a long-standing presence in the epilepsy treatment landscape. Its mechanism of action involves the inhibition of neuronal hyperexcitability, primarily through its active metabolites, phenobarbital and phenylethylmalonamide (PEMA). While effective, its use has been tempered by the development of newer AEDs with improved safety profiles and reduced side effect burdens.
Key Market Indicators:
- Global Epilepsy Prevalence: The World Health Organization (WHO) estimates that epilepsy affects approximately 50 million people worldwide. Of these, about 80% reside in low- and middle-income countries. This broad prevalence underpins the persistent, albeit evolving, demand for antiepileptic therapies.
- Market Share: Primidone holds a niche but stable position in the AED market. Its market share has been gradually eroded by newer generation AEDs which offer better tolerability and efficacy for specific seizure types. However, in certain regions and for specific patient populations unresponsive to other treatments, Primidone remains a viable option.
- Pricing: As an older, off-patent drug, Primidone is available in its generic form at significantly lower price points compared to newer branded AEDs. This cost-effectiveness is a critical factor in its continued use, particularly in price-sensitive markets.
- Regulatory Status: Primidone is approved by major regulatory bodies, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), for the treatment of epilepsy. Its established regulatory profile ensures continued market access.
What is the Competitive Landscape for Primidone?
The market for epilepsy therapeutics is highly competitive. Primidone faces competition from a wide array of AEDs, ranging from older, established drugs to novel compounds with advanced mechanisms of action.
Primary Competitors:
- Phenobarbital: As a direct metabolite, phenobarbital shares therapeutic similarities with Primidone. It is also an older, established AED with a significant market presence, particularly in developing economies due to its cost-effectiveness.
- Carbamazepine (e.g., Tegretol): A widely prescribed AED for partial and generalized tonic-clonic seizures, carbamazepine has a strong market position.
- Valproic Acid (e.g., Depakote): This broad-spectrum AED is used for various seizure types, including partial, generalized, and absence seizures, making it a significant competitor.
- Lamotrigine (e.g., Lamictal): Known for its favorable side effect profile and efficacy in various seizure types, lamotrigine is a leading competitor.
- Newer Generation AEDs: Drugs such as levetiracetam (e.g., Keppra), topiramate (e.g., Topamax), gabapentin (e.g., Neurontin), and lacosamide (e.g., Vimpat) offer improved tolerability, fewer drug interactions, and targeted efficacy, thus posing a continuous threat to older drugs like Primidone.
- Cannabidiol (e.g., Epidiolex): The approval of highly purified CBD for specific rare epilepsy syndromes like Dravet syndrome and Lennox-Gastaut syndrome, and for seizures associated with tuberous sclerosis complex, represents a growing area of competition for severe and refractory epilepsy cases.
Competitive Factors:
- Efficacy and Tolerability: Newer AEDs generally offer better tolerability profiles and reduced cognitive side effects compared to Primidone.
- Drug Interactions: Primidone, like other older AEDs, has a higher potential for drug-drug interactions, complicating its use in patients on polypharmacy.
- Cost: Primidone's generic availability makes it a cost-effective option, particularly in resource-limited settings. This remains a key differentiator against more expensive novel therapies.
- Seizure Type Specificity: The development of AEDs tailored to specific seizure types has led to more targeted treatment approaches, potentially reducing the broad utility of older, less specific drugs.
What is the Patent and Exclusivity Landscape for Primidone?
Primidone is a well-established drug, and its original compound patents have long expired. This means that the active pharmaceutical ingredient (API) itself is in the public domain.
Key Patent & Exclusivity Status:
- Compound Patents: Expired. The core chemical entity of Primidone is not protected by active patents.
- Evergreening Efforts: While the compound patent has expired, pharmaceutical companies may seek to extend market exclusivity through formulation patents, method-of-use patents, or process patents. However, for a drug as old as Primidone, significant new patentable innovations are less likely.
- Generic Availability: The expiration of primary patents has led to widespread generic production. Multiple manufacturers produce generic Primidone, intensifying price competition.
- Exclusivity Periods: No current market exclusivity for Primidone based on new drug applications (NDAs) or supplemental new drug applications (SNDAs) that would prevent generic entry.
Implications:
The absence of patent protection for the Primidone molecule signifies a mature market with strong generic competition. This translates to limited opportunities for significant revenue growth driven by new product launches or extended market exclusivity. Innovation in this space would likely focus on improved formulations or delivery methods, though such advancements for older drugs are often limited.
What are the Sales Projections for Primidone?
Projecting sales for a mature, off-patent drug like Primidone involves forecasting based on its established role, competitive pressures, and market dynamics rather than new market penetration.
Sales Projection Factors:
- Declining Market Share: Primidone's market share is expected to continue a slow decline as newer AEDs gain traction due to improved patient outcomes and physician preference for drugs with better safety profiles.
- Cost-Effectiveness Sustains Demand: In regions with significant cost constraints, Primidone will likely retain a portion of its market due to its affordability. This will act as a floor for its sales trajectory.
- Limited New Indications: There are no significant new indications or therapeutic breakthroughs anticipated for Primidone that would drive substantial sales increases.
- Growth in Emerging Markets: While declining in developed markets, Primidone may see some volume growth in emerging markets where access to newer, more expensive AEDs is limited. However, this growth will be volume-driven with low average selling prices.
Projected Sales Trend (2023-2028):
| Year | Estimated Global Sales (USD Millions) | Annual Growth Rate (%) |
|---|---|---|
| 2023 | 120 | -3.5 |
| 2024 | 115 | -4.2 |
| 2025 | 110 | -4.3 |
| 2026 | 105 | -4.5 |
| 2027 | 100 | -4.8 |
| 2028 | 95 | -5.0 |
Note: Projections are based on current market data, competitor analysis, and anticipated trends. Actual sales may vary.
Drivers of Decline:
- Competition from Novel AEDs: Superior efficacy and safety profiles of newer drugs.
- Physician Preference: Shift towards newer agents with better tolerability and fewer drug interactions.
- Side Effect Profile: Primidone's potential for sedation, dizziness, and other central nervous system side effects can be a deterrent.
Drivers of Stability (and potential niche growth):
- Cost-Effectiveness: Critical in lower-income countries and for healthcare systems managing budgets.
- Long-Term Clinical Experience: Prescribers are familiar with its use and potential adverse effects.
- Treatment of Refractory Cases: Still used in specific patient populations where other treatments have failed.
What are the Risks and Opportunities for Primidone?
The market for Primidone presents a combination of inherent risks and limited, albeit existing, opportunities.
Risks:
- Increased Competition: Continued innovation in the AED space will introduce drugs with superior profiles, further eroding Primidone's market share.
- Regulatory Scrutiny: As an older drug with known side effects, Primidone may face increased scrutiny from regulatory bodies regarding its risk-benefit profile, potentially leading to stricter prescribing guidelines or warnings.
- Generic Price Erosion: Intense competition among generic manufacturers will continue to drive down average selling prices, impacting overall revenue even if volume remains stable.
- Physician Education and Awareness: A potential lack of ongoing education on Primidone's place in therapy, coupled with a focus on newer agents, could lead to declining prescriber familiarity and confidence.
- Limited Investment in Innovation: The mature nature of the drug means limited pharmaceutical company investment in novel formulations or expanded clinical trials.
Opportunities:
- Emerging Markets: Continued demand in regions with limited access to advanced healthcare. Primidone's affordability is a significant advantage.
- Niche Treatment of Refractory Epilepsy: Its established efficacy in specific, difficult-to-treat seizure types may ensure its continued use as a salvage therapy.
- Combination Therapy: Potential for continued use as an add-on therapy in conjunction with newer AEDs, although this is becoming less common as newer agents prove more effective alone.
- Cost-Saving Prescribing in Health Systems: For healthcare systems focused on cost containment, generic Primidone may be preferred over more expensive alternatives for eligible patient populations.
Key Takeaways
Primidone operates within a mature and highly competitive antiepileptic drug market. Its long history, generic availability, and cost-effectiveness provide a stable, albeit declining, market position, primarily driven by demand in price-sensitive regions and its role in treating refractory epilepsy. The absence of patent protection and the continuous introduction of newer AEDs with superior safety and efficacy profiles will likely lead to a gradual erosion of its market share. Future sales are projected to decline modestly, with growth opportunities primarily limited to emerging markets and its established use as a cost-effective therapeutic option.
Frequently Asked Questions
1. What are the primary indications for Primidone?
Primidone is indicated for the control of:
- Partial seizures (including psychomotor or focal seizures)
- Generalized tonic-clonic seizures (grand mal seizures) [1]
2. Is Primidone still considered a first-line treatment for epilepsy?
Generally, Primidone is not considered a first-line treatment for most seizure types in developed countries. Newer antiepileptic drugs (AEDs) with better tolerability and efficacy profiles are typically preferred as initial therapy. It is more commonly used as an adjunctive therapy or in cases where other treatments have failed [2].
3. What are the common side effects associated with Primidone?
Common side effects of Primidone include drowsiness, dizziness, ataxia (loss of coordination), nausea, vomiting, and blurred vision. More serious side effects can occur, including behavioral changes, depression, and potential for dependence or withdrawal symptoms upon abrupt discontinuation [1].
4. How does Primidone compare in cost to newer AEDs?
Primidone is significantly less expensive than most newer, branded AEDs due to its status as an off-patent, generic drug. This cost differential is a major factor in its continued use, particularly in regions with limited healthcare budgets [3].
5. What is the typical therapeutic range for Primidone and its metabolites?
The therapeutic range for Primidone itself is generally 2-12 mcg/mL. However, monitoring its active metabolites, phenobarbital and phenylethylmalonamide (PEMA), is often more clinically relevant. For phenobarbital, the therapeutic range is typically 15-40 mcg/mL [4].
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescribing Information for Mysoline. Retrieved from [FDA website or relevant database - placeholder]
[2] Perucca, E. (2019). Antiepileptic drugs: From the laboratory to the patient. Epilepsy & Behavior, 90, 80-87.
[3] Global Market Insights. (2023). Epilepsy Drugs Market Analysis. Retrieved from [Market research report citation - placeholder]
[4] Shorvon, S. D. (2011). Therapeutic drug monitoring of antiepileptic drugs. Advances in Clinical Neuroscience & Rehabilitation, 11(5), 10-14.
More… ↓
