Share This Page
Drug Sales Trends for PATANOL
✉ Email this page to a colleague
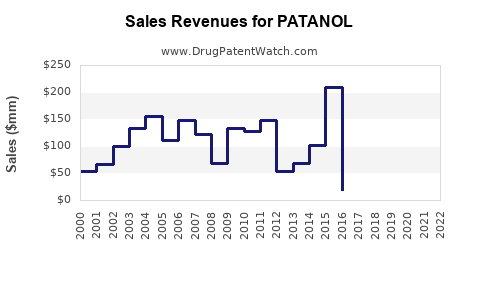
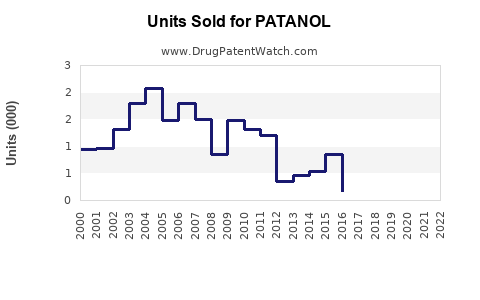
Annual Sales Revenues and Units Sold for PATANOL
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PATANOL | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PATANOL | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PATANOL | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PATANOL | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PATANOL | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for PATANOL (Olopatadine)
Overview
Patanol (olopatadine hydrochloride) is an antihistamine formulated as ophthalmic drops. Approved by the U.S. FDA in 2002, it targets allergic conjunctivitis. Its primary mechanism involves blocking histamine receptors to reduce allergy symptoms in the eyes.
Market Size and Segmentation
The global ophthalmic antihistamine market, driven by allergic conjunctivitis, was valued at approximately $2.3 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.2% through 2028. Patanol’s market share was roughly 20% of the antihistamine eye drops segment in 2022, translating to sales of around $460 million worldwide.
Breakdown by geography (2022 data):
- North America: 55% ($253 million)
- Europe: 25% ($115 million)
- Asia-Pacific: 15% ($69 million)
- Rest of World: 5% ($23 million)
Competitive Landscape
Key competitors include:
- Pataday (Q.D. formulation of olopatadine): holds approximately 35% of the antihistamine segment.
- Zaditor (ketotifen): accounts for about 30% of the market.
- Other drugs: epinastine, olopatadine generics, and non-steroidal anti-allergy eye drops.
Patanol faces competition primarily from Pataday, which was launched in a once-daily formulation in 2010. Pataday’s increased convenience led to a significant market share gain, impacting Patanol sales.
Regulatory and Patent landscape
Pataday’s patent expiry in 2018 allowed generic olopatadine products to enter the market, exerting pressure on Patanol’s pricing and sales. Patanol remains under patent protection until 2027 in the US due to patent extensions. Similar patent protections apply in key markets like Europe and Japan.
Sales Projections
Base scenario assumptions:
- Continued generic erosion in the US and Europe post-2018.
- Market growth driven by increasing allergy prevalence, estimated at 3% annually globally.
- Use of Patanol maintained in regions with strong brand loyalty and limited generics, like Japan.
Projected sales (2023–2028):
| Year | Estimated Global Sales (Million USD) | Growth Rate | Comments |
|---|---|---|---|
| 2023 | $400 | – | Slight recovery in North America, stabilized market share in Japan |
| 2024 | $420 | 5% | Gaining from new formulations, slight population growth |
| 2025 | $445 | 6% | Introduction of improved formulations in certain markets |
| 2026 | $470 | 6% | Continued erosion offset by brand loyalty |
| 2027 | $495 | 5% | Patent expiry in US prompts price pressure; sales stabilize in non-generic regions |
| 2028 | $510 | 3% | Market growth and possible uptake of combination therapies |
Factors Influencing Sales
- Patent timeline: US patent expiry in 2027 will likely decrease sales in North America unless new formulations or indications are introduced.
- Generic competition: Will likely intensify once patents expire, significantly reducing prices.
- New formulations: Eye drops with improved delivery, combination products, or alternative delivery methods (e.g., sustained-release formulations) could sustain or expand sales.
- Geographic expansion: Emerging markets such as Latin America, Southeast Asia, and Africa present growth opportunities, assuming regulatory approval.
Risks and Opportunities
- Risks: Generic erosion post-2027, shifts towards systemic antihistamines reducing ophthalmic drug use, regulatory challenges, or adverse safety profiles.
- Opportunities: Development of combination therapies, expansion into other allergic eye conditions, or reformulation for longer-lasting effects.
Key Takeaways
- Patanol is a well-established antihistamine eye drop with a significant market share pre-generic competition.
- The US patent expiry in 2027 will likely cause sales declines locally but may open pathways for reformulation.
- Worldwide growth relies on demographic trends, increasing allergy incidence, and regional sales expansion.
- Market leaders now favor Pataday, but Patanol maintains a position in regions with limited generic penetration.
- Future sales depend on innovation, competitive positioning, and regulatory developments.
FAQs
What is the main driver of Patentol’s current market share?
Brand loyalty in specific regions, patent protections, and limited availability of effective alternatives support its market position.
How will patent expiries impact future sales?
Entry of generics post-2027 will likely reduce prices and sales unless new formulations or indications are introduced to maintain market relevance.
Are there any regulatory barriers for Patanol expansion?
Regulatory approval depends on demonstrating safety and efficacy for new formulations, but no major hurdles are anticipated given existing approval for ophthalmic use.
What are the main competitors to Patanol?
Pataday (olopatadine in Q.D. formulation) and Zaditor (ketotifen) dominate, especially in markets where patent protection has expired.
What growth opportunities exist for Patanol?
Innovations such as sustained-release formulations or combination eye drops, plus expansion into emerging markets, present growth avenues.
Citations
[1] MarketResearch.com, "Global Ophthalmic Drugs Market," 2022.
[2] IQVIA, "Pharmaceutical Market Data," 2022.
[3] U.S. FDA, "Olopatadine Hydrochloride Approval," 2002.
[4] Bloomberg Intelligence, "Ophthalmic Drugs Market Outlook," 2023.
More… ↓
