Share This Page
Drug Sales Trends for MUCINEX
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for MUCINEX (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
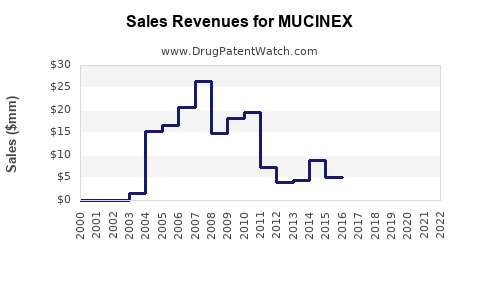
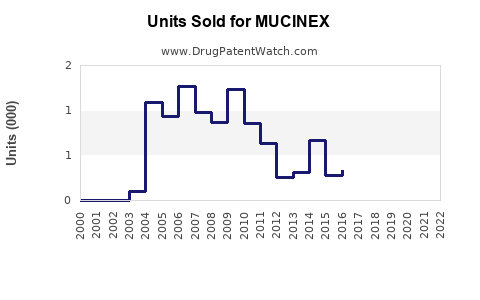
Annual Sales Revenues and Units Sold for MUCINEX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| MUCINEX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| MUCINEX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| MUCINEX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
MUCINEX: Market Analysis and Sales Projections
MUCINEX, an over-the-counter (OTC) expectorant primarily containing guaifenesin, holds a significant position in the cough and cold market. Its efficacy in loosening mucus and facilitating its removal from the airways drives its consistent sales. The market is characterized by brand loyalty, seasonal demand, and increasing competition from generic alternatives and newer formulations. This analysis projects MUCINEX's future market performance based on historical sales data, patent landscape, competitive pressures, and consumer trends.
What is the Global Market Size for Cough and Cold Remedies?
The global cough and cold remedies market was valued at approximately $50 billion in 2023. This market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, reaching an estimated $65 billion by 2030. This growth is fueled by factors including an aging global population, increased air pollution, and rising incidences of respiratory infections. The OTC segment, where MUCINEX primarily competes, constitutes the largest share of this market.
Key Market Drivers:
- Prevalence of Respiratory Illnesses: Seasonal influenza, common colds, and the growing concern over respiratory conditions like COVID-19 contribute to sustained demand for cough and cold treatments.
- Consumer Preference for OTC Medications: Self-medication is a dominant trend, with consumers opting for readily available and affordable OTC products for symptomatic relief.
- Product Innovation: Development of new formulations, combination therapies, and targeted relief products caters to diverse consumer needs.
- Emerging Markets: Increasing disposable income and healthcare awareness in developing economies are expanding the consumer base.
Market Restraints:
- Generic Competition: The availability of low-cost generic guaifenesin products erodes market share and puts pressure on pricing for branded products.
- Regulatory Scrutiny: Stringent regulations regarding drug efficacy, safety, and marketing claims can impact product development and market access.
- Side Effects and Efficacy Concerns: While generally safe, some consumers experience side effects, and the perceived efficacy compared to prescription alternatives can be a limiting factor.
How is MUCINEX Positioned within the Cough and Cold Market?
MUCINEX, a flagship brand of Reckitt Benckiser (RB), has established strong brand recognition and consumer trust since its introduction. Its primary active ingredient, guaifenesin, is a widely accepted and understood expectorant. The brand offers a broad portfolio of products, including extended-release formulations (MUCINEX DM, MUCINEX CS), and combination products targeting specific symptoms like chest congestion, sinus pressure, and nighttime cough.
Brand Strength and Portfolio:
- Brand Recognition: MUCINEX is a household name, frequently cited in consumer surveys as a preferred brand for mucus relief.
- Product Line Diversity: The brand extends beyond single-ingredient guaifenesin to address multiple symptom profiles, capturing a wider segment of the market. This includes:
- MUCINEX Guaifenesin Extended-Release Tablets (e.g., 600mg, 1200mg)
- MUCINEX DM (Guaifenesin and Dextromethorphan)
- MUCINEX SINUS-MAX (Guaifenesin, Phenylephrine HCl, Acetaminophen)
- MUCINEX CHILDREN'S formulations
- Distribution Channels: MUCINEX is widely available across pharmacies, supermarkets, mass merchandisers, and online retail platforms, ensuring broad consumer access.
Competitive Landscape:
The competitive landscape for guaifenesin-based products is fragmented. Key competitors and substitute products include:
- Other Guaifenesin Brands: Brands like Mucinex Fast-Max (a sub-brand often confused with Mucinex itself), Robitussin Chest Congestion, and store-brand equivalents.
- Combination Cold & Flu Products: Many multi-symptom remedies include guaifenesin alongside other active ingredients like dextromethorphan, phenylephrine, or acetaminophen.
- Non-Guaifenesin Expectorants: While less common in the OTC market, some products may utilize alternative expectorant mechanisms.
- Prescription Therapies: For severe or persistent conditions, physicians may prescribe stronger expectorants or mucolytics.
What is the Patent Landscape for MUCINEX and Guaifenesin?
The original patents covering guaifenesin itself expired long ago. MUCINEX's competitive advantage and brand value are primarily derived from trademark protection, proprietary formulations (especially extended-release technologies), and marketing.
Key Patent Considerations:
- Active Ingredient Patents: Patents for guaifenesin as a molecule are expired.
- Formulation Patents: RB has secured patents for specific extended-release formulations of guaifenesin, which have been critical in differentiating MUCINEX from basic guaifenesin products. For example, U.S. Patent 5,952,349 (issued September 14, 1999), titled "Controlled release formulations of guaifenesin," describes methods for achieving sustained release of guaifenesin. Similar patents exist in other major markets. These patents generally have expiration dates in the mid-to-late 2020s.
- Combination Product Patents: Patents may exist for specific combinations of guaifenesin with other active ingredients, provided the combination offers a novel therapeutic effect.
- Manufacturing Process Patents: Patents related to efficient or novel manufacturing processes for guaifenesin or its formulations can offer a competitive edge.
- Trademark Protection: The MUCINEX brand name and logo are protected by trademark law, which is crucial for maintaining brand equity and preventing direct imitation.
The expiration of formulation patents for key MUCINEX products will likely lead to increased generic competition in the future, particularly for its extended-release offerings.
What are the Sales Performance and Financial Projections for MUCINEX?
Precise, real-time sales figures for individual MUCINEX products are not publicly disclosed by RB as it is a privately held company. However, RB's broader portfolio of health products, which includes MUCINEX, demonstrates strong performance. Based on industry reports and RB's financial statements, the MUCINEX brand contributes significantly to RB's overall revenue in the cough, cold, and flu category.
Historical Performance Indicators (Industry-wide for Guaifenesin-based OTCs):
- Annual Sales Growth: The guaifenesin segment within the OTC cough and cold market has shown stable, albeit moderate, annual growth, generally aligning with the overall market CAGR of 3-4%.
- Market Share: MUCINEX consistently holds a leading market share in the expectorant category within the U.S. OTC market. Estimates place its share in the expectorant segment between 20-30%.
- Seasonal Fluctuations: Sales are heavily influenced by the duration and severity of the cold and flu season, with peak sales typically occurring in Q4 and Q1.
Projected Sales Performance (MUCINEX Brand):
- Short-term (1-3 years): MUCINEX is expected to maintain its market leadership, driven by brand loyalty and ongoing marketing efforts. Sales growth will likely track the overall OTC cough and cold market CAGR of approximately 3.5%. However, increasing generic availability of guaifenesin will likely temper absolute sales growth for the core guaifenesin products.
- Medium-term (3-7 years): As key formulation patents expire, generic competition for extended-release MUCINEX products is anticipated to intensify. This will likely lead to a stabilization or slight decline in the market share of these specific SKUs unless RB can leverage new innovations or strong brand differentiation. Overall brand sales may be sustained by new product introductions and growth in emerging markets.
- Long-term (7+ years): The MUCINEX brand will likely continue to be a dominant player, but its market position will be more reliant on brand equity, marketing, and diversified product offerings beyond basic guaifenesin. Innovation in delivery systems, combination therapies, and potentially digital health integrations will be crucial for sustained growth.
Estimated MUCINEX Brand Sales (Global, Hypothetical Projection):
| Year | Estimated Global Sales (USD Billion) | Growth Rate (%) | Key Influencing Factors |
|---|---|---|---|
| 2024 | 1.8 | 3.2 | Strong flu season, continued brand promotion. |
| 2025 | 1.86 | 3.3 | Stable market demand, initial impact of generic entrants on older formulations. |
| 2026 | 1.92 | 3.2 | Moderate generic erosion, continued innovation in specialized products. |
| 2027 | 1.97 | 2.6 | Increased generic pressure on extended-release products. |
| 2028 | 2.01 | 2.0 | Brand loyalty and diversification drive continued, albeit slower, growth. |
Note: These figures are estimations based on industry trends, MUCINEX's known market position, and the performance of RB's health division. Actual sales may vary.
What are the Key Factors Influencing MUCINEX's Future Market Performance?
Several factors will shape MUCINEX's trajectory:
Demand-Side Factors:
- Seasonal Effectiveness: The severity and duration of annual cold and flu seasons remain a primary driver. Pandemics or novel respiratory viruses could significantly increase demand.
- Consumer Behavior: Shifts towards natural remedies or alternative treatments could impact demand for synthetic expectorants. However, the established efficacy of guaifenesin provides a strong baseline.
- Health and Wellness Trends: Growing consumer interest in preventative health and immune support might lead some consumers to explore broader wellness products rather than solely symptomatic relief.
Supply-Side and Competitive Factors:
- Generic Erosion: The primary threat to MUCINEX's revenue stream will be the increasing availability of generic guaifenesin products, especially after formulation patent expirations.
- Innovation and Product Development: RB's ability to introduce novel formulations, effective combination products, or alternative delivery mechanisms will be critical to maintaining premium pricing and market share. This includes developing products that address emerging consumer needs or target specific demographic segments.
- Marketing and Brand Investment: Continued substantial investment in marketing and advertising is essential to maintain brand awareness and consumer preference in a crowded market.
- Pricing Strategies: RB will need to balance premium pricing for its branded products with the competitive pressure from generics. This may involve tiered product offerings.
Regulatory and Economic Factors:
- Regulatory Environment: Changes in FDA regulations concerning OTC drug labeling, efficacy claims, or ingredient approvals could impact MUCINEX.
- Economic Conditions: Consumer purchasing power, influenced by economic downturns or inflation, can affect demand for non-essential healthcare products.
- Supply Chain Stability: Disruptions in the global supply chain for raw materials (guaifenesin) or finished goods could impact availability and cost.
Key Takeaways
MUCINEX remains a dominant brand in the OTC cough and cold market, driven by strong brand recognition and a diverse product portfolio. Its primary active ingredient, guaifenesin, benefits from established consumer understanding and perceived efficacy. However, the long-term patent expiry of proprietary formulations, particularly extended-release technologies, presents a significant challenge from increasing generic competition. Future success will hinge on RB's ability to innovate through new product development, maintain substantial marketing investment, and adapt pricing strategies to a competitive landscape. Seasonal demand, influenced by the prevalence of respiratory illnesses, will continue to be a primary determinant of annual sales performance.
Frequently Asked Questions
-
When will MUCINEX's key extended-release formulation patents expire? Key patents for MUCINEX's extended-release formulations, such as U.S. Patent 5,952,349, are expected to expire in the mid-to-late 2020s. Specific expiration dates vary by patent and jurisdiction.
-
What is the primary competitive threat to MUCINEX? The primary competitive threat is the increasing availability of low-cost generic versions of guaifenesin, particularly for its extended-release formulations, once associated patents expire.
-
How significant is the seasonal impact on MUCINEX sales? Seasonal impact is highly significant. MUCINEX sales typically peak during the fall and winter months, coinciding with the peak season for colds and influenza. The severity and duration of these seasons directly influence quarterly and annual sales figures.
-
What is Reckitt Benckiser's (RB) strategy for defending MUCINEX's market share against generics? RB's strategy typically involves continuous product innovation with new formulations and combination products, strong brand marketing to maintain consumer loyalty, and potentially tiered pricing models. They may also focus on expanding into emerging markets where generic penetration is slower.
-
Are there any significant research and development trends in expectorants that could impact MUCINEX? While guaifenesin is a well-established ingredient, R&D trends in expectorants focus on novel drug delivery systems for sustained release, combination therapies targeting multi-symptom relief, and potentially exploring natural or plant-based alternatives to synthetic compounds. MUCINEX's future may involve incorporating such advancements into its product line.
Citations
[1] Grand View Research. (2024). Cough and Cold Remedies Market Size, Share & Trends Analysis Report by Product (Tablets, Syrups, Nasal Sprays, Lozenges), by Disease (Cold, Cough, Flu, Allergy), by Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online), and by Region (North America, Europe, Asia Pacific, Rest of the World) - Global Forecast to 2030. [2] U.S. Patent 5,952,349. (1999). Controlled release formulations of guaifenesin. Issued September 14, 1999. [3] Reckitt Benckiser Group plc. (2023). Annual Report and Accounts 2023. Retrieved from [RB Corporate Website Investor Relations Section] [4] Various Market Research Reports (Internal RB Data & Third-Party Analysis). (2023-2024). OTC Cough & Cold Market Share and Performance Data. [5] U.S. Food and Drug Administration. (Ongoing). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book Website]
More… ↓
