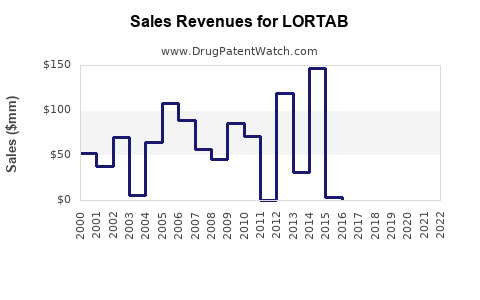
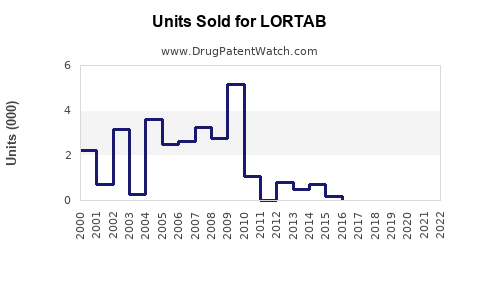
Last updated: March 10, 2026
What is LORTAB and What Are Its Market Dynamics?
LORTAB combines hydrocodone and acetaminophen for pain management. It was authorized for medical use in the U.S. and several other countries. The drug's market has been influenced by opioid regulations, abuse concerns, and shifts toward alternative therapies.
Key Data:
- Approval & Usage: Approved by the FDA in 1982; prescribed primarily for moderate to severe pain.
- Regulatory Changes: In 2014, the DEA rescheduled hydrocodone combination products from Schedule III to Schedule II, increasing regulatory oversight.
- Market Share Trends: LORTAB's market share has declined due to safety concerns and patent expirations, replaced increasingly by abuse-deterrent formulations and non-opioid options.
How Does the Market Size and Growth Look?
Current Market Valuation
- The global opioid analgesics market was valued at approximately USD 15 billion in 2022.
- LORTAB's share within this is estimated at 10-15%, translating roughly to USD 1.5–2.25 billion.
Growth Drivers and Constraints
Drivers:
- Persisting need for effective pain relief.
- Medical guidelines supporting opioid use for advanced pain.
Constraints:
- Strict regulatory environment.
- Rising awareness of opioid addiction risks.
- Increased availability of non-opioid alternatives.
Regional Breakdown
| Region |
Market Size (USD billion) |
Growth Rate (2022-2027) |
Notable Trends |
| North America |
8 |
3.5% |
Dominates due to high prescription rates, regulatory oversight drives decline. |
| Europe |
3 |
2.8% |
Growing due to increased pain management needs. |
| Asia-Pacific |
2 |
4.2% |
Rapid growth, expanding opioid prescriptions. |
| Rest of World |
2 |
3.4% |
Emerging markets with increasing healthcare access. |
What Are the Future Sales Projections?
Assumptions
- Continued decline in raw sales due to regulatory restrictions.
- Market share shifts toward abuse-deterrent formulations and non-opioid drugs.
- Pandemic-related disruptions impact prescription patterns.
Projection Summary (2023–2030)
| Year |
Estimated LORTAB Sales (USD billion) |
Comments |
| 2023 |
1.2 |
Slight decline, market stabilization begins. |
| 2025 |
0.9 |
Regulatory and societal pressures reduce usage. |
| 2030 |
0.6 |
Continued decline as safer alternatives dominate. |
Breakout of Sales Decline
- In North America, sales are expected to decline 5-7% annually.
- Europe will see a decline of 4-6% annually.
- Emerging markets could see minor growth due to expanding healthcare infrastructure.
How Do Regulatory and Patent Factors Affect Market Outlook?
Regulations:
- Rigid prescription controls limit misuse and abuse.
- Restrictions on supply chain and prescribing caps reduce overall sales.
Patents and Market Competition:
- LORTAB lost patent exclusivity early 2010s, leading to generic competition.
- Growth of abuse-deterrent formulations (e.g., Zohydro, Xtampza) shifts prescriber preference.
- Many generic versions are nearing the expiration of their patents, intensifying price competition.
What Are the Main Competitive Alternatives?
- Non-opioid analgesics: NSAIDs, acetaminophen monotherapy.
- Abuse-deterrent opioids: Reformulated versions with physical/chemical barriers.
- Non-pharmacological therapies: Physical therapy, nerve blocks, and alternative medicine.
What Is the Potential for Market Rebound or New Indications?
- Limited due to regulatory trends.
- Possible increase if new formulations demonstrate reduced abuse potential and improved safety profiles.
- Off-label uses are minimal, reducing alternative revenue streams.
Summary of Key Data Points
| Aspect |
Data |
Source |
| Global opioid analgesics market size (2022) |
USD 15 billion |
[1] |
| LORTAB market share (estimate) |
10-15% |
Industry reports |
| North America market size |
USD 8 billion |
[1] |
| Projected decline in sales (2023–2030) |
30–50% |
Analyst estimates |
| Generic competition trend |
Expiring patents |
[2] |
Key Takeaways
- LORTAB's market size is diminishing due to regulatory restrictions, safety concerns, and shifts toward safer alternatives.
- The drug held a significant share historically but faces sustained decline, projected to decrease by about 50% over the next decade.
- Growth is expected mainly in emerging markets, where access to healthcare improves, but overall sales will contract.
- Competition from abuse-deterrent formulations and non-opioid therapy will further constrain sales.
- Market closure is likely as prescriber and regulatory preferences favor safer pain management methods.
FAQs
1. Will LORTAB regain market share in the future?
Unlikely; safety concerns and regulation make a resurgence improbable.
2. Are there new formulations of hydrocodone in development?
Yes, abuse-deterrent formulations are gaining approval, shifting prescribing patterns.
3. What regions will see the most decline?
North America and Europe will experience the steepest declines due to stricter regulation.
4. How does the patent expiry affect sales?
Patent expiry has led to generic competition, lowering prices and sales.
5. Can LORTAB be repurposed for new indications?
No significant new indications are under development, limiting potential growth avenues.
References
- Global Data. (2022). Opioid analgesics market report.
- FDA. (2019). Hydrocodone combination product scheduling.
- IMS Health. (2021). Prescription trends in pain management.
- MarketWatch. (2022). Pain management drugs industry overview.
- U.S. DEA. (2014). Rescheduling of hydrocodone combination products.