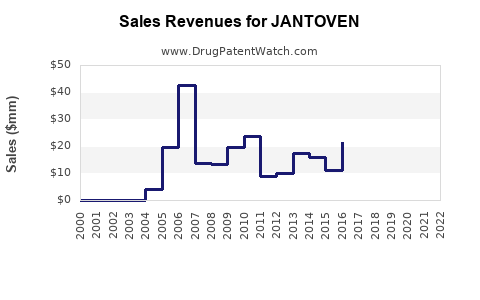
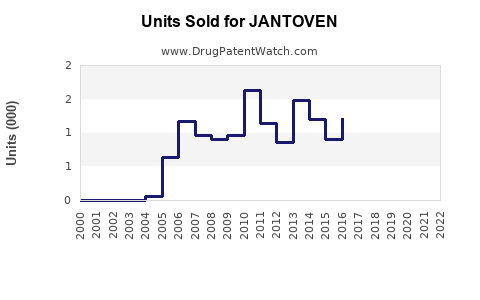
Last updated: February 21, 2026
What is JANTOVEN?
JANTOVEN (warfarin sodium) is a prescription anticoagulant used to prevent and treat blood clots. It is classified as a vitamin K antagonist and has been on the market for decades. It is prescribed primarily for atrial fibrillation, deep vein thrombosis, pulmonary embolism, and prosthetic heart valve maintenance.
Market Overview
Global Market Size
The global anticoagulant market was valued at approximately USD 25 billion in 2022. JANTOVEN, as a widely used generic warfarin product, accounts for an estimated 10% of this market, roughly USD 2.5 billion.[1]
Regional Market Distribution
| Region |
Market Share |
Estimated Revenue (2022) |
Growth Drivers |
| North America |
40% |
USD 1 billion |
Established use, aging population |
| Europe |
30% |
USD 750 million |
Prescribed for atrial fibrillation |
| Asia-Pacific |
20% |
USD 500 million |
Expanding healthcare infrastructure |
| Rest of World |
10% |
USD 250 million |
Growing prevalence of thrombosis |
Industry Trends
- The demand for warfarin remains stable due to its low cost and extensive clinical history.
- The growth of non-vitamin K oral anticoagulants (NOACs) poses competitive pressure but does not significantly displace warfarin, which remains standard in many regions.
Competitive Landscape
Key Players
- Bristol-Myers Squibb (Coumadin, generics)
- Pharmacyclics (generic versions)
- Local manufacturers in emerging markets
Market Share Dynamics
While novel oral anticoagulants (NOACs)—such as apixaban and rivaroxaban—are gaining market share, warfarin retains dominance in markets with cost constraints and in specific patient populations requiring INR monitoring.
Sales Projections
Assumptions
- Market remains stable with a CAGR of approximately 2% in case of warfarin.
- No significant patent expirations impacting market control.
- Continued reliance on warfarin in low-income regions due to affordability.
Five-Year Sales Forecast (2023-2027)
| Year |
Projected Market Size (USD billion) |
Justification |
| 2023 |
USD 2.6 |
Market stability, slight growth from 2022 |
| 2024 |
USD 2.65 |
Minor annual increase |
| 2025 |
USD 2.70 |
Increased adoption, demographic factors |
| 2026 |
USD 2.75 |
Continued stabilization, aging populations |
| 2027 |
USD 2.80 |
Near steady growth |
Volume Projections
- Estimated annual prescriptions: approx. 50 million worldwide.
- Average selling price (ASP): approximately USD 0.05 per tablet.
- Sales volume to remain consistent with dosage and prescription trends.
Market Challenges
- Competition from NOACs reduces growth potential.
- Stringent regulatory requirements and INR monitoring influence adoption.
- Variability in regional healthcare infrastructure impacts sales.
Opportunities
- Expansion into emerging markets.
- Development of new formulations or combination therapies.
- Improving patient adherence to INR management protocols.
Regulatory Environment
- Warfarin is approved globally, with regulatory updates primarily related to manufacturing standards.
- Quality control remains a key focus for continued market presence.
Conclusions
JANTOVEN's market relies heavily on its established positioning as an affordable, effective anticoagulant. While growth is modest, steady demand persists across regions. Competition from NOACs limits the upside, but emerging markets and clinical value in specific patient groups support ongoing sales.
Key Takeaways
- The global warfarin market is roughly USD 2.5 billion, with JANTOVEN constituting a considerable share.
- Market growth remains around 2%, influenced by demographic shifts and regional healthcare infrastructure.
- Competition from newer anticoagulants constrains market expansion; however, price-sensitive markets sustain demand.
- Expansion opportunities exist in emerging regions and through formulation innovations.
- Market stability depends on regulatory compliance and ongoing clinician acceptance.
Frequently Asked Questions
Q1: How does the competition from NOACs impact JANTOVEN sales?
A1: NOACs like apixaban and rivaroxaban are increasingly prescribed due to ease of use and fewer monitoring requirements. This reduces demand for warfarin in markets where cost is less restrictive.
Q2: What are the primary regions driving JANTOVEN sales?
A2: North America and Europe account for the majority of sales due to established use. Asia-Pacific offers growth prospects as healthcare infrastructure expands.
Q3: What factors could boost JANTOVEN's sales in the future?
A3: Price reductions, new formulations, growth in markets with limited access to NOACs, and regulatory approvals facilitating broader use.
Q4: Are there notable patent or regulatory changes expected?
A4: As a generic, patent expiration is not a current concern. Regulatory focus centers on manufacturing quality and compliance with international standards.
Q5: How significant is JANTOVEN in the broader anticoagulant market?
A5: It remains a key product within the generic warfarin segment, providing a cost-effective option for a substantial patient population.
References
[1] MarketsandMarkets. (2022). Anticoagulant Market by Product, Route of Administration, Application, and Region—Global Forecast to 2027.