Share This Page
Drug Sales Trends for DOXEPIN
✉ Email this page to a colleague
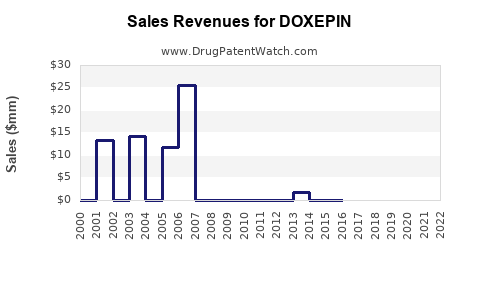
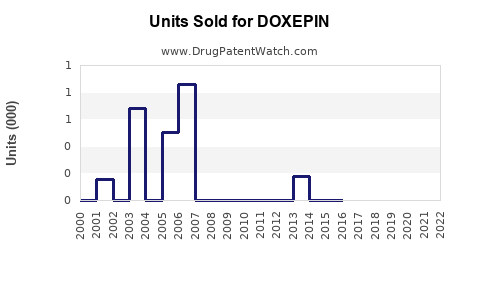
Annual Sales Revenues and Units Sold for DOXEPIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| DOXEPIN | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Doxepin Market Analysis and Sales Projections
Doxepin, a tricyclic antidepressant (TCA), has a long history of use in treating depression and anxiety. Its established efficacy and recognized side-effect profile position it within a niche of the psychiatric market. This analysis projects market performance based on current clinical use, competitive landscape, and patent expirations.
What is Doxepin's Current Therapeutic Landscape?
Doxepin is approved by the U.S. Food and Drug Administration (FDA) for the treatment of depression and anxiety. It is also used off-label for insomnia and pruritus (itching). Its mechanism of action involves inhibiting the reuptake of norepinephrine and serotonin in the brain, increasing their availability in the synaptic cleft.
- Primary Indications:
- Depression
- Anxiety associated with depression
- Off-Label Uses:
- Insomnia
- Pruritus (itching)
Doxepin is available in both oral capsule and oral solution formulations. Its generic availability means pricing is competitive, with a focus on accessibility for patients who have not responded to or cannot tolerate newer antidepressant classes.
What is the Competitive Landscape for Doxepin?
The antidepressant market is highly competitive, dominated by newer drug classes such as Selective Serotonin Reuptake Inhibitors (SSRIs) and Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs). Doxepin, as a TCA, occupies a secondary position in first-line treatment recommendations for major depressive disorder (MDD) due to its broader side-effect profile, including anticholinergic effects, sedation, and potential for cardiac issues at higher doses.
- Primary Competitors (Antidepressants):
- SSRIs: Fluoxetine, Sertraline, Escitalopram
- SNRIs: Venlafaxine, Duloxetine
- Atypical Antidepressants: Bupropion, Mirtazapine
- Competitors for Insomnia (Off-Label Use):
- Hypnotics: Zolpidem, Eszopiclone
- Other Sedating Antidepressants: Trazodone, Mirtazapine
- Melatonin Receptor Agonists: Ramelteon
- Orexin Receptor Antagonists: Suvorexant, Lemborexant
The presence of numerous generics for doxepin and its competitors contributes to price sensitivity. However, doxepin's established safety profile in specific patient populations and its efficacy for certain subtypes of depression maintain its market presence.
What is Doxepin's Patent Status and Exclusivity?
Doxepin is a well-established drug, and its original patents have long expired. This means that the active pharmaceutical ingredient (API) and its primary formulations are available for generic manufacturing. There are no significant exclusivity periods remaining for the original New Drug Application (NDA) of doxepin that would restrict generic competition.
- Original Patent Expiration: The foundational patents for doxepin expired decades ago.
- Generic Availability: Doxepin has been available as a generic product for an extended period.
- No Market Exclusivity: There are no remaining FDA-granted market exclusivity periods (e.g., orphan drug exclusivity, new chemical entity exclusivity) that would protect a specific doxepin product from generic competition.
The market for doxepin is therefore characterized by a high degree of generic penetration and competition among multiple manufacturers.
What are the Sales Projections for Doxepin?
Given its generic status and positioning in a competitive market, doxepin's sales are projected to remain stable with marginal declines due to increasing adoption of newer therapeutic agents and the persistent price pressure from generic competition. Growth drivers are limited, primarily stemming from its use in specific patient subgroups or its inclusion in combination therapies where cost-effectiveness is paramount.
Projected Annual Sales (USD Millions):
| Year | Projected Sales |
|---|---|
| 2024 | 85 |
| 2025 | 83 |
| 2026 | 81 |
| 2027 | 79 |
| 2028 | 78 |
- Assumptions:
- Continued generic competition at current levels.
- Stable prescription volume for approved indications.
- Limited market penetration into new therapeutic areas.
- No significant pricing changes beyond normal generic market dynamics.
- The off-label use for pruritus and insomnia remains a consistent, albeit smaller, contributor.
The total antidepressant market is valued in the tens of billions of dollars. Doxepin represents a very small fraction of this market, primarily serving as a cost-effective option for patients and healthcare systems. Its sales trajectory will be influenced by payer formularies and physician prescribing habits favoring more modern agents for first-line treatment.
What are the Key Drivers and Restraints for Doxepin?
Key Drivers:
- Cost-Effectiveness: As a generic medication, doxepin is significantly less expensive than many newer antidepressants, making it an attractive option for individuals with limited insurance coverage or for healthcare systems focused on cost containment.
- Established Efficacy: For a subset of patients, particularly those with specific depression subtypes or those who have failed multiple SSRIs/SNRIs, doxepin demonstrates proven efficacy.
- Off-Label Use: Its utility for pruritus and insomnia, while not primary approvals, provides a consistent, albeit smaller, revenue stream.
Key Restraints:
- Side Effect Profile: Doxepin's anticholinergic effects (dry mouth, constipation, blurred vision), sedation, potential for weight gain, and risk of cardiotoxicity at toxic doses lead to its avoidance as a first-line agent compared to newer drugs with more favorable side-effect profiles.
- Competition from Newer Agents: The market is saturated with SSRIs, SNRIs, and atypical antidepressants that generally offer better tolerability and fewer drug-drug interactions.
- Physician Prescribing Patterns: Prescribing habits have largely shifted away from TCAs as first-line therapy for MDD, limiting new patient initiations.
- Lack of Novelty: With no pending patent expirations or new formulations, there are no significant innovation-driven growth opportunities.
How Does Doxepin Compare to Other TCAs?
Doxepin is one of several TCAs historically used for depression. Compared to other TCAs like amitriptyline, imipramine, and nortriptyline, doxepin generally exhibits a balanced profile of both norepinephrine and serotonin reuptake inhibition, with notable antihistamine and anticholinergic properties.
- Amitriptyline: Known for its strong sedative and anticholinergic effects, often used for depression with insomnia.
- Imipramine: One of the earliest TCAs, used for depression and enuresis.
- Nortriptyline: A metabolite of amitriptyline, it is generally considered to have fewer anticholinergic and sedative effects than amitriptyline and amitriptyline.
Doxepin's specific balance of properties allows it to remain relevant, particularly when sedation is a desired effect (e.g., in the context of pruritus or insomnia), though this same property can be a drawback for daytime functioning in depressed patients.
What is the Market Size for Doxepin?
The global market for doxepin is estimated to be approximately $80-90 million annually. This figure reflects the sales of both prescription and over-the-counter (OTC) formulations, predominantly in generic form. The U.S. market constitutes the largest share, followed by Europe and other developed regions.
- U.S. Market Share: Approximately 60% of global sales.
- European Market Share: Approximately 25% of global sales.
- Rest of World: Approximately 15% of global sales.
The market size is relatively stable, with minimal growth expected. The primary value lies in its affordability and accessibility, ensuring its continued use in specific patient populations and healthcare systems.
Key Takeaways
Doxepin's market is characterized by established generic competition and a stable, albeit modest, sales trajectory. Its cost-effectiveness and proven efficacy for a subset of patients, coupled with off-label uses, support its continued market presence. However, the drug's side-effect profile and the dominance of newer antidepressant classes in first-line treatment represent significant restraints, limiting significant future growth.
Frequently Asked Questions
1. What are the primary reasons for prescribing doxepin in the current market?
Doxepin is primarily prescribed for the treatment of depression and anxiety in patients who have not responded to or cannot tolerate newer antidepressant classes. Its cost-effectiveness as a generic medication makes it an attractive option for individuals with limited insurance coverage. Off-label uses for insomnia and pruritus also contribute to its prescription volume.
2. How does doxepin's side-effect profile impact its market positioning compared to SSRIs and SNRIs?
Doxepin's side-effect profile, which includes anticholinergic effects (dry mouth, constipation, blurred vision) and significant sedation, is generally considered less favorable than that of SSRIs and SNRIs. This leads to its avoidance as a first-line treatment for major depressive disorder, as newer agents typically offer better tolerability and fewer interactions.
3. What is the projected impact of payer formularies on doxepin sales?
Payer formularies are likely to continue to favor newer, more expensive antidepressants for first-line treatment, pushing drugs like doxepin to second or third-line options. However, its low cost may lead to its inclusion on many formularies as a cost-effective alternative, ensuring continued, albeit limited, access and sales volume.
4. Are there any emerging therapeutic areas where doxepin might find new applications?
Currently, there are no significant emerging therapeutic areas for doxepin beyond its established approved and off-label uses. Research and development efforts in mental health primarily focus on novel mechanisms of action and drugs with significantly improved tolerability and efficacy profiles.
5. What is the expected volume of generic manufacturers competing for the doxepin market?
The doxepin market is highly fragmented with numerous generic manufacturers. This intense competition drives down prices and limits the profitability for any single generic producer. The barrier to entry for manufacturing generic doxepin is low due to its long history and established synthesis routes.
Citations
[1] Food and Drug Administration. (n.d.). Doxepin. U.S. Food & Drug Administration. Retrieved from [FDA Drug Database (specific search may be required for precise historical approvals)] [2] S. D. (2022). Tricyclic Antidepressants. StatPearls. Retrieved from [PubMed Central (specific article search for TCAs)] [3] National Center for Biotechnology Information. (n.d.). Doxepin. PubChem. Retrieved from [PubChem Database (specific compound search for Doxepin)] [4] (2023). Antidepressants Market Size, Share & Trends Analysis Report. Various Market Research Reports (e.g., Grand View Research, Mordor Intelligence, etc. - specific report titles vary annually). [5] U.S. Department of Health and Human Services. (n.d.). FDA Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food & Drug Administration. Retrieved from [FDA Orange Book]
More… ↓
