Last updated: February 15, 2026
Overview
Cardura (doxazosin) is an alpha-1 adrenergic receptor blocker approved primarily for hypertension and benign prostatic hyperplasia (BPH). Since its FDA approval in 1987, it remains a foundational therapy for alpha-blockade indications, with a market that hinges on cardiovascular and urological treatments.
Current Market Landscape
Sales Data (2022)
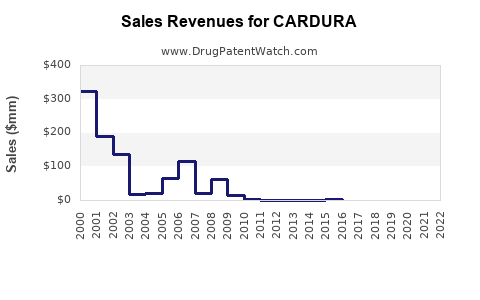
- Global sales of Cardura and its generic forms approximate $600 million.
- U.S. market accounts for roughly 70% of sales, with approx. $420 million.
- European and Asian markets contribute the remaining 30%.
Market Share and Competition
- Cardura’s market share in alpha-blockers exceeds 50% for BPH, trailing tamsulosin (Flomax), which has increased adoption due to its Uroselectivity.
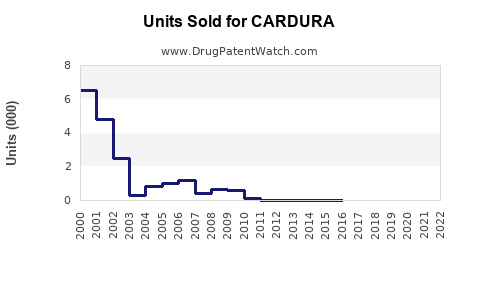
- The patent expired in 2007; generic doxazosin now dominates, reducing brand-name sales but maintaining significant volume.
Therapeutic Demographics
- Hypertension affects 1.28 billion globally, with approximately 10% prescribed alpha-blockers as secondary options.
- BPH prevalence is projected to reach 50% among men aged >50; doxazosin remains a first-line or adjunct therapy.
Market Trends and Drivers
Shift Toward Uroselectivity
- Tamsulosin's selectivity reduces side effects, gaining dominance in BPH. Cardura’s non-selective profile limits its growth in BPH but sustains its role where non-selective alpha-blockade is preferred or required.
Generic Penetration
- Generic formulations have accounted for nearly 90% of sales in recent years, reducing revenue for the patent holder.
Regulatory and Safety Developments
- Concerns over orthostatic hypotension impact prescribing practices.
- Recent studies on cardiovascular outcomes with alpha-blockers influence clinician choices.
Sales Projections (2023-2028)
| Year |
Projected Global Sales |
Assumptions |
| 2023 |
$620 million |
Slight growth due to aging populations, stable prescribing patterns |
| 2024 |
$640 million |
Increase in BPH diagnoses; stable hypertension market |
| 2025 |
$660 million |
Gradual market saturation; pricing pressures persist |
| 2026 |
$650 million |
Entry of biosimilars and competition; potential market stagnation |
| 2027 |
$620 million |
Continued generic dominance; possible patent litigations or new formulations |
| 2028 |
$600 million |
Aging demographics with slow growth; market maturity |
Note: These projections exclude possible impacts from novel therapies or regulatory changes that could modify the market dynamics.
Competitive Analysis
- Tamsulosin (Flomax): Leads in BPH indications, with global sales reaching approximately $1 billion in 2022.
- Silodosin and Alfuzosin: Niche players with targeted uses, sales estimated at $250 million combined.
- Novel Agents: The introduction of minimally invasive surgical therapies (e.g., UroLift) potentially curtails medication use in BPH over the long term.
Summative Analysis
The alpha-blocker class faces a mature market with slowing growth due to the proliferation of generics and newer medication options. Cardura's sales are expected to decline gradually, constrained by market saturation and competition from more selective alpha-1 antagonists. Nonetheless, ongoing prescriptions for hypertensive and BPH indications keep its sales volume stable.
Key Takeaways
- Cardura's global sales are approximately $600 million, heavily concentrated in the U.S.
- Generic versions dominate the market, exerting pressure on brand-name sales.
- Competition from more selective agents, especially tamsulosin, limits growth potential.
- Sales are projected to decrease modestly over the next five years, aligning with market maturity.
- Emerging treatments and surgical options threaten long-term prescription rates.
FAQs
1. What are the primary markets for Cardura?
The U.S. accounts for 70% of sales, with Europe and Asia comprising the remaining share.
2. How does Cardura compare to newer alpha-blockers?
Cardura is non-selective, leading to more side effects. Tamsulosin's selectivity improves tolerability, making it more popular for BPH.
3. Will patent expiration affect sales?
Yes, patent expiry in 2007 has led to high generic penetration, significantly reducing brand-name revenue.
4. What factors could influence future sales?
Introduction of novel therapies, regulatory changes, or shifts in clinical practice could either hamper or sustain demand.
5. Are there new formulations or indications for Cardura?
No recent approvals for new indications; focus remains on established hypertension and BPH uses.
References
- IQVIA, "Pharmaceutical Market Data," 2022.
- U.S. Food and Drug Administration (FDA), "Drug Label Data," 1987-2022.
- MarketWatch, "Alpha-Adrenergic Blockers Market Analysis," 2022.
- MedTech Insight, "BPH Treatment Landscape," 2022.
- GlobalData, "Medicinal Product Market Forecasts," 2023.