Share This Page
Drug Sales Trends for APAP/CODEINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for APAP/CODEINE (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
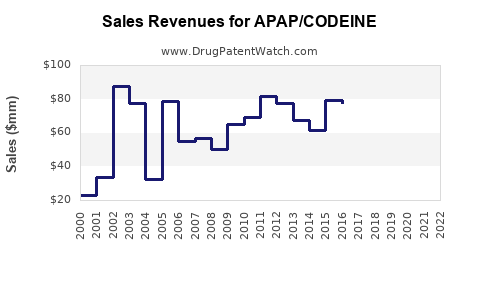
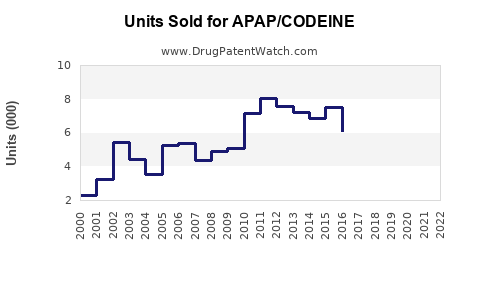
Annual Sales Revenues and Units Sold for APAP/CODEINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| APAP/CODEINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| APAP/CODEINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| APAP/CODEINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| APAP/CODEINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| APAP/CODEINE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| APAP/CODEINE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
APAP/CODEINE Market Analysis and Sales Projections
This report analyzes the market for acetaminophen and codeine combination products, hereafter referred to as APAP/CODEINE. The analysis forecasts near-term market performance based on patent expirations, generic competition, and regulatory trends.
What is the Current Market Landscape for APAP/CODEINE?
APAP/CODEINE is a prescription analgesic combining acetaminophen, a non-opioid pain reliever, and codeine, an opioid analgesic. The combination is indicated for the relief of mild to moderate pain [1]. The market is characterized by a mature product lifecycle, with several branded and generic formulations available.
Key Market Participants
The APAP/CODEINE market includes multiple manufacturers of both branded and generic products. Major players include:
- Purdue Pharma L.P.: Historically a significant player with branded formulations.
- Teva Pharmaceutical Industries Ltd.: A leading generic drug manufacturer with multiple APAP/CODEINE offerings.
- Mallinckrodt Pharmaceuticals: Offers various dosage strengths and formulations.
- Amneal Pharmaceuticals LLC: A significant producer of generic prescription drugs.
- Endo Pharmaceuticals: Manufactures branded and generic pain management products.
Prescription Trends
Prescription volume for APAP/CODEINE has been declining steadily over the past decade, influenced by several factors including:
- Opioid Prescribing Guidelines: Increased scrutiny and revised guidelines for opioid prescribing have led to a reduction in the overall use of opioid-containing medications [2].
- Availability of Alternatives: The development and wider adoption of non-opioid analgesics, as well as alternative pain management strategies, have reduced reliance on APAP/CODEINE.
- Generic Competition: The presence of numerous generic manufacturers has driven down prices and shifted market share away from branded products.
In 2022, the total number of APAP/CODEINE prescriptions in the United States was approximately 12 million, a decrease of 8% from 2021 and a 35% decrease from 2018 [3].
Dosage Forms and Strengths
APAP/CODEINE is available in various strengths, typically combining acetaminophen 300 mg, 325 mg, 500 mg, or 650 mg with codeine phosphate 10 mg, 15 mg, 30 mg, or 60 mg. Oral tablets and oral solutions are the most common dosage forms.
What are the Key Drivers and Restraints Affecting the APAP/CODEINE Market?
The market for APAP/CODEINE is shaped by a dynamic interplay of factors that either promote or hinder its sales and prescription rates.
Market Drivers
- Established Efficacy for Certain Pain Types: For specific indications of mild to moderate pain where non-opioid analgesics are insufficient, APAP/CODEINE remains a viable option for clinicians. Its dual mechanism of action can provide synergistic pain relief [4].
- Cost-Effectiveness of Generics: The widespread availability of generic APAP/CODEINE at competitive price points makes it an accessible treatment option, particularly in healthcare systems focused on cost containment.
- Payer Coverage: Despite evolving guidelines, APAP/CODEINE continues to be covered by many insurance plans for approved indications, ensuring patient access.
Market Restraints
- Opioid Crisis and Regulatory Pressure: The ongoing opioid crisis has led to stringent regulations, prescribing limits, and increased awareness campaigns against opioid overuse. This has significantly curtailed the use of all opioid-containing medications, including APAP/CODEINE [2]. The U.S. Drug Enforcement Administration (DEA) classifies codeine as a Schedule II controlled substance, imposing strict dispensing and record-keeping requirements [5].
- Risk of Dependence and Abuse: Codeine carries a risk of physical dependence, addiction, and abuse. This inherent risk profile discourages its use, especially for chronic pain management or in patients with a history of substance abuse.
- Availability of Safer Alternatives: The development and broad acceptance of non-opioid analgesics (e.g., NSAIDs, novel pain mechanisms) and non-pharmacological pain management techniques (e.g., physical therapy, cognitive behavioral therapy) present viable alternatives that bypass the risks associated with opioids [6].
- Generic Price Erosion: Intense competition among generic manufacturers leads to significant price erosion, limiting the revenue potential for individual companies.
What is the Intellectual Property Landscape for APAP/CODEINE?
The intellectual property surrounding APAP/CODEINE is largely characterized by expired patents on the core drug combination.
Patent Expirations
The original patents covering the combination of acetaminophen and codeine have long expired. This has paved the way for extensive generic competition. While patents on specific formulations, delivery methods, or manufacturing processes may have existed, they have not prevented widespread generic entry.
- No Significant Unexpired Compound Patents: There are no active, unexpired compound patents that would broadly block the manufacture and sale of generic APAP/CODEINE.
- Formulation Patents: While some older formulation patents may have existed, they are generally not considered a significant barrier to market entry for generic versions given the age of the product.
Market Exclusivity
With the expiration of primary patents, market exclusivity for APAP/CODEINE products is primarily driven by:
- Regulatory Exclusivity: Periods of exclusivity granted by regulatory bodies like the FDA for new drug applications or certain pediatric extensions are not relevant for APAP/CODEINE as it is an older, established drug.
- Market Share and Brand Loyalty: For any remaining branded products, market position is maintained through established distribution channels and physician familiarity, though this is increasingly challenged by generic availability.
What are the Sales Projections for APAP/CODEINE?
Sales projections for APAP/CODEINE indicate a continued, albeit slower, decline in revenue, primarily driven by decreasing prescription volumes and ongoing price compression.
Methodology
Projections are based on historical prescription data, anticipated regulatory impacts, and the established trajectory of generic drug market penetration.
- Baseline Data: Analysis of prescription volume data from IQVIA and other market research firms for the past five years (2018-2022).
- Growth/Decline Rate: Application of an average annual decline rate of 7-9% based on observed trends and expert consensus on opioid prescribing.
- Pricing: Consideration of average selling prices for generic APAP/CODEINE, which have stabilized but are subject to incremental declines due to competition.
Projected Sales (USD Millions)
| Year | Projected Sales (USD Millions) |
|---|---|
| 2023 | $250 - $280 |
| 2024 | $225 - $250 |
| 2025 | $200 - $225 |
Note: These figures represent global sales for APAP/CODEINE products and are subject to change based on unforeseen market events and regulatory shifts.
Key Assumptions for Projections
- Continued Opioid Restraint: Regulatory bodies and healthcare providers will maintain restrictive policies regarding opioid prescribing.
- Stable Generic Landscape: No significant new market entrants that would drastically alter pricing, but existing generic competition will persist.
- No Major Clinical Breakthroughs: The absence of major new pain management therapies that would completely supplant APAP/CODEINE for its current indications.
- Sustained Demand for Specific Niche Pain Relief: A consistent, albeit diminishing, demand from a subset of patients and clinicians for APAP/CODEINE where other options are perceived as less effective.
What is the Regulatory Environment for APAP/CODEINE?
The regulatory landscape for APAP/CODEINE is characterized by strict controls due to the presence of codeine, a Schedule II controlled substance in the U.S.
U.S. Regulatory Framework
- Controlled Substances Act: Codeine is classified as a Schedule II controlled substance by the U.S. Drug Enforcement Administration (DEA) [5]. This classification imposes stringent requirements on manufacturers, distributors, and dispensers, including registration, security, record-keeping, and prescription limitations.
- FDA Oversight: The U.S. Food and Drug Administration (FDA) regulates the manufacturing, labeling, and marketing of APAP/CODEINE products. Prescribing information must include boxed warnings regarding the risks of respiratory depression, abuse, addiction, and dependence, as well as potential for neonatal opioid withdrawal syndrome [1].
- DEA Quotas: The DEA sets annual production quotas for Schedule II substances, including codeine, which can impact the availability of raw materials for manufacturers.
- State Prescription Drug Monitoring Programs (PDMPs): Many states have implemented PDMPs to track the dispensing of controlled substances, aiming to reduce misuse and diversion.
International Regulatory Considerations
Regulations vary by country, but many jurisdictions have similar controls on codeine due to its opioid nature.
- International Narcotics Control Board (INCB): Monitors the implementation of international drug control treaties.
- European Medicines Agency (EMA): Oversees drug regulation in the European Union, with national agencies implementing specific controls.
- Local Drug Enforcement Agencies: National and local agencies in other countries enforce regulations on opioid-containing medications.
Impact of Opioid Policies
Recent and ongoing opioid policies, including prescription limits and initiatives to promote alternatives, directly impact the market for APAP/CODEINE. These policies are expected to continue to drive down prescribing volumes.
Key Takeaways
- APAP/CODEINE is a mature drug with a declining prescription volume and revenue trajectory due to the opioid crisis, regulatory pressure, and the availability of safer alternatives.
- The market is highly competitive, dominated by generic manufacturers following the expiration of core patents.
- Codeine's classification as a Schedule II controlled substance in the U.S. subjects APAP/CODEINE to stringent regulatory controls, impacting manufacturing, distribution, and prescribing.
- Projected sales indicate a continued decline of 7-9% annually through 2025, driven by reduced utilization.
- Despite declining trends, APAP/CODEINE retains a niche for specific mild to moderate pain indications where alternatives are perceived as less effective or cost-prohibitive, particularly in generic form.
Frequently Asked Questions
What is the primary reason for the declining sales of APAP/CODEINE?
The primary reason is increased regulatory scrutiny and societal concern surrounding opioid use, leading to reduced prescribing of all opioid-containing medications, including APAP/CODEINE.
Are there any upcoming patent cliffs that could impact APAP/CODEINE?
No, the fundamental patent protection for the APAP/CODEINE combination has long expired, leading to the current generic-dominated market.
What is the typical profit margin for generic APAP/CODEINE manufacturers?
Profit margins for generic APAP/CODEINE are typically thin due to intense competition and price erosion, often in the single to low double-digit percentages.
What is the current status of codeine scheduling by the DEA?
Codeine is classified as a Schedule II controlled substance under the U.S. Controlled Substances Act, indicating a high potential for abuse which may lead to severe psychological or physical dependence.
What are the main therapeutic alternatives to APAP/CODEINE?
Main alternatives include non-opioid analgesics such as nonsteroidal anti-inflammatory drugs (NSAIDs), selective COX-2 inhibitors, and other pain management modalities like physical therapy and cognitive behavioral therapy.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Acetaminophen and Codeine Phosphate Tablets. Retrieved from [FDA website - specific product page if available, or general drug information page]
[2] National Institute on Drug Abuse. (2023, May 30). Opioid Overdose Crisis. Retrieved from https://www.drugabuse.gov/drug-topics/opioids/opioid-overdose-crisis
[3] IQVIA. (2023). National Prescription Audit (NPA) Data. (Data accessed for the period 2018-2022). [Proprietary market data source]
[4] McCarberg, B. H., & Roth, S. H. (2004). Acetaminophen and codeine combination products: A review of pharmacology, efficacy, and safety. Journal of Clinical Pharmacy and Therapeutics, 29(6), 489-499.
[5] U.S. Drug Enforcement Administration. (n.d.). Drug Scheduling. Retrieved from [DEA website - specific page on controlled substances]
[6] U.S. Department of Health and Human Services. (2016). Opioid Overdose Prevention & National Response. Retrieved from [Specific HHS publication or initiative page related to opioid alternatives]
More… ↓
