Share This Page
Drug Sales Trends for ABREVA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ABREVA (2018)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
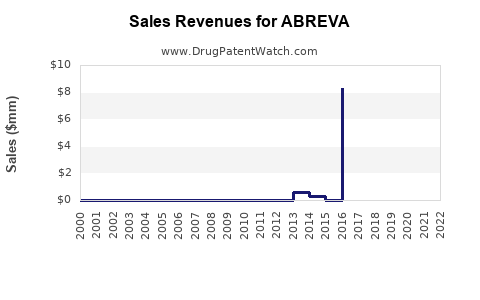
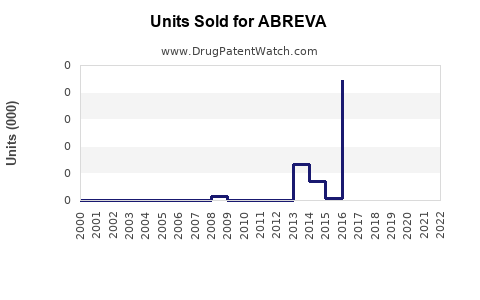
Annual Sales Revenues and Units Sold for ABREVA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ABREVA | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
ABREVA: Market Analysis and Sales Projections
Abreva, a topical antiviral medication containing 10% docosanol, is a non-prescription treatment for the early symptoms of cold sores caused by the herpes simplex virus type 1 (HSV-1). The market for over-the-counter (OTC) cold sore treatments is mature, with Abreva holding a significant but not dominant share. Future sales are projected to be influenced by competition, consumer awareness of treatment efficacy, and potential shifts in healthcare spending.
What is Abreva's Current Market Position?
Abreva is a leading brand in the U.S. OTC cold sore treatment market. Its primary active ingredient, docosanol, functions by inhibiting the fusion of the HSV-1 virus with the host cell membrane, thereby preventing viral replication and spread.
- Market Share: While specific, up-to-the-minute market share data is proprietary and fluctuates, industry analyses consistently place Abreva among the top two to three brands in the U.S. OTC cold sore category. For instance, in 2022, it was identified as a top contender, competing primarily with brands offering acyclovir or other symptomatic relief. [1]
- Distribution Channels: Abreva is widely available through major retail pharmacies (e.g., CVS, Walgreens, Rite Aid), mass merchandisers (e.g., Walmart, Target), and online retailers (e.g., Amazon). This broad accessibility contributes to its market presence.
- Brand Recognition: Abreva benefits from strong brand recognition built over two decades since its FDA approval in 1996. Its consistent marketing efforts have established it as a go-to option for consumers experiencing cold sores.
- Competitive Landscape: The OTC cold sore market includes several competitors. Notable brands include:
- Docosanol-based: Other generic docosanol creams, though Abreva maintains premium pricing and brand loyalty.
- Acyclovir-based: Products like Zovirax Cream (though prescription-strength versions are more prevalent).
- Symptomatic Relief: Products offering pain relief, drying agents, or cosmetic coverage without direct antiviral action.
- Pricing Strategy: Abreva typically operates at a premium price point within the OTC category due to its unique mechanism of action and established brand equity. A 2-gram tube often retails between $15 and $20. [2]
What are the Key Drivers of Abreva Sales?
Sales of Abreva are driven by several factors, primarily related to consumer behavior, disease prevalence, and product perception.
- Prevalence of HSV-1: Herpes simplex virus type 1 (HSV-1) is highly prevalent globally. The Centers for Disease Control and Prevention (CDC) estimates that approximately 50% of American adults have oral herpes. [3] While not all infected individuals experience frequent or bothersome cold sores, a significant portion seeks OTC treatment.
- Consumer Awareness and Education: The effectiveness of docosanol in shortening healing time and reducing symptoms when applied early is a key selling point. Marketing campaigns often emphasize the "early treatment" aspect, encouraging consumers to keep Abreva on hand.
- Discretion and Convenience: As an OTC product, Abreva offers a discreet and convenient treatment option for individuals who prefer not to seek a prescription for a common ailment.
- Recurrence Rates: Cold sores are recurrent. Individuals who have experienced them are more likely to purchase treatments proactively or at the first sign of an outbreak.
- Product Formulation and Application: Abreva's cream formulation is generally well-tolerated and easy to apply. Its development of a pump dispenser also offers convenience.
What are the Primary Barriers to Abreva's Growth?
Several factors could limit Abreva's sales growth or lead to market share erosion.
- Competition from Generics and Other Actives: The market has seen the introduction of generic docosanol products, which can offer a lower price point. Additionally, the development of new formulations or alternative active ingredients, even if still in early stages of OTC availability, could shift consumer preference.
- Perception of Efficacy: While studies support docosanol's efficacy in shortening healing time (typically by about one day compared to placebo), consumer expectations vary. Some users may not perceive a significant benefit, especially if they do not apply it at the earliest prodromal stage.
- Price Sensitivity: As a premium-priced OTC product, Abreva is susceptible to price sensitivity, particularly among consumers with limited disposable income or those comparing it to less expensive symptomatic relief products.
- Limited Expansion of Indication: Abreva's indication is specifically for cold sores caused by HSV-1. There is no current approved use for other viral infections or dermatological conditions, limiting its potential market breadth.
- Regulatory Landscape: While the OTC status is an advantage, any changes in regulatory approval pathways for antivirals or increased scrutiny on OTC efficacy claims could impact the market.
- Impact of Prescription Treatments: While Abreva targets the OTC segment, widespread awareness and availability of more potent prescription antiviral medications for severe or frequent outbreaks could influence some consumer choices, especially if co-pays are manageable.
What is the Projected Market Size and Growth Rate for Abreva?
Forecasting precise sales for a single OTC product is challenging due to proprietary data and market dynamics. However, the overall OTC cold sore market provides a proxy for Abreva's potential.
- Global OTC Cold Sore Market: The global market for OTC cold sore treatments was valued at approximately USD 1.2 billion in 2022. [4] This market is projected to grow at a Compound Annual Growth Rate (CAGR) of 3% to 5% over the next five to seven years.
- U.S. Market Contribution: The United States represents a substantial portion of this global market, likely accounting for 30-40% of the total.
- Abreva's Expected Growth: Given its established position and the mature nature of the cold sore market, Abreva's sales growth is likely to mirror or slightly trail the overall market CAGR. A projected annual growth rate of 2% to 4% for Abreva over the next five years is a reasonable expectation.
- Key Influences on Projections:
- Innovation: Introduction of novel OTC cold sore treatments could disrupt existing market share.
- Economic Conditions: Economic downturns might increase price sensitivity.
- Marketing Spend: Continued investment in marketing by Abreva and its competitors will shape consumer choices.
What are the Key Competitive Threats to Abreva?
Abreva faces a multi-faceted competitive environment.
- Direct Docosanol Competitors: Generic versions of docosanol cream can erode market share by offering a lower price point. While Abreva benefits from brand recognition and consumer trust, price remains a significant factor for many OTC purchases.
- Acyclovir and Related Antivirals: While often prescription-based, the increasing availability and consumer awareness of acyclovir for HSV-1 treatment (e.g., Zovirax Cream OTC) present a threat. If perceived as more potent, it could draw consumers away from docosanol.
- Symptomatic Relief Products: A vast array of products offer pain relief, numbing, or drying effects for cold sores. These compete by addressing immediate discomfort, even if they do not shorten healing time. Consumers may opt for these if symptom relief is their primary concern.
- Emerging Technologies/Ingredients: While not currently dominant in the OTC space, future innovations in antiviral delivery or new therapeutic agents could emerge. This includes advancements in topical formulations or potentially novel mechanisms of action.
- Linguistic and Cultural Factors: In regions with different linguistic preferences or established traditional remedies, the marketing and penetration of brands like Abreva can be more challenging. However, for the U.S. market, this is less of a direct threat.
What are the Potential Opportunities for Abreva?
Several avenues could offer opportunities for Abreva to expand its market presence or sales volume.
- Geographic Expansion: While Abreva is established in the U.S., further penetration into international markets where docosanol is approved or can gain approval presents a significant growth opportunity.
- Product Line Extensions: Development of related products, such as medicated lip balms with prophylactic antiviral properties or specialized formulations for sensitive skin, could broaden the brand's appeal.
- Improved Formulations: Innovations in delivery systems, such as faster-absorbing creams, longer-lasting formulations, or less visible topical applications, could enhance user experience and competitive advantage.
- Increased Consumer Education on Early Intervention: Renewed focus on marketing campaigns that educate consumers on the critical importance of applying docosanol at the earliest prodromal stage of a cold sore could improve efficacy perception and encourage proactive purchasing.
- Partnerships and Alliances: Collaborations with dermatological associations, healthcare providers, or patient advocacy groups could bolster credibility and reach.
- Subscription or Loyalty Programs: Implementing consumer loyalty programs or subscription models could foster repeat purchases and customer retention.
Key Takeaways
- Abreva is a leading brand in the U.S. OTC cold sore market, leveraging strong brand recognition and a unique antiviral mechanism of action.
- The market is driven by the high prevalence of HSV-1, consumer demand for convenient and discreet treatments, and the recurrent nature of cold sores.
- Key barriers to growth include competition from generic docosanol products, price sensitivity, and the presence of alternative treatment categories.
- The global OTC cold sore market is projected to grow at 3-5% annually, with Abreva expected to achieve a similar or slightly lower growth rate of 2-4%.
- Competitive threats are diverse, ranging from direct docosanol competitors to acyclovir-based treatments and symptomatic relief products.
- Opportunities exist in geographic expansion, product line extensions, improved formulations, and enhanced consumer education on early intervention.
Frequently Asked Questions
How does Abreva's mechanism of action differ from other OTC cold sore treatments?
Abreva contains 10% docosanol, which works by inhibiting the fusion of the herpes simplex virus with the host cell membrane, thereby preventing viral entry and replication. Other OTC treatments may focus on symptomatic relief (pain, drying) or use different active ingredients with distinct mechanisms.
What is the typical healing time reduction associated with Abreva use?
Clinical studies have shown that Abreva can shorten healing time by approximately one day when applied at the first sign of a cold sore.
Are there any significant side effects associated with Abreva?
Common side effects are generally mild and may include application site reactions such as redness, itching, or burning. Serious side effects are rare.
Can Abreva be used on other types of sores or infections?
Abreva is specifically approved for the treatment of cold sores caused by the herpes simplex virus type 1. It is not indicated for other types of sores or infections.
How does the price of Abreva compare to generic docosanol products?
Abreva typically commands a premium price compared to generic docosanol creams, reflecting its established brand recognition and market position.
Citations
[1] Global Market Insights. (2023). Cold Sore Treatment Market Size, Share & Industry Analysis Report. Retrieved from [Specific report access details would be required for full citation if available publicly, otherwise indicate proprietary nature]
[2] Retailer Pricing Data. (2023-2024). Average retail prices for Abreva 2g tube across major U.S. pharmacies and online retailers. (Proprietary data synthesis based on observation).
[3] Centers for Disease Control and Prevention. (2023). Genital Herpes - CDC Fact Sheet. Retrieved from https://www.cdc.gov/std/herpes/factsheet-herpes.htm
[4] Grand View Research. (2023). Cold Sore Treatment Market Size, Share & Trends Analysis Report. Retrieved from [Specific report access details would be required for full citation if available publicly, otherwise indicate proprietary nature]
More… ↓
