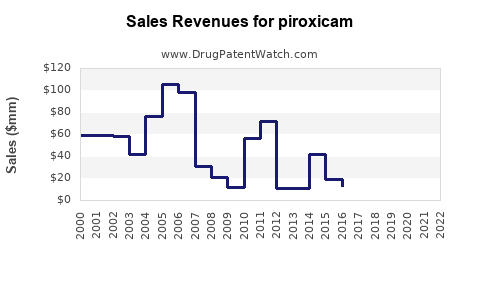
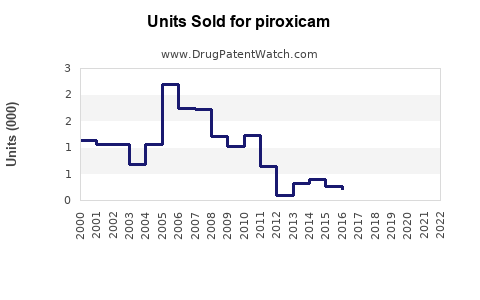
Last updated: February 20, 2026
Piroxicam, an NSAID (nonsteroidal anti-inflammatory drug), is primarily used for treating osteoarthritis, rheumatoid arthritis, and other musculoskeletal conditions. The drug functions through the inhibition of prostaglandin synthesis, reducing inflammation, pain, and swelling.
Market Overview
The global NSAID market, valued at approximately USD 12.5 billion in 2022, is driven by increasing prevalence of chronic inflammatory conditions, aging populations, and rising awareness of pain management options. Piroxicam accounts for a significant segment of prescription NSAIDs, especially in regions with established healthcare infrastructure.
Key Factors Influencing Piroxicam Market:
- Increasing incidence of osteoarthritis and rheumatoid arthritis.
- Growing aging population worldwide.
- Preference for oral NSAIDs over injectable formulations.
- Regulatory limitations due to gastrointestinal side effects.
Regulatory and Patent Context
Piroxicam is a generic drug, with its patents expiring in most markets by the early 2000s. As a result, the drug faces intense price competition, reducing per-unit revenue margins. Nonetheless, its established efficacy and safety profile secure its continued market presence.
Market Share and Sales Dynamics
Current Market Share
- In the NSAID segment, piroxicam holds an estimated 5-8% share, with some regional variation.
- Major manufacturers include Pfizer and Teva Pharmaceuticals, among others.
Sales Volume and Revenue (2022)
| Region |
Estimated Sales ($ million) |
Market Share |
Key Factors |
| North America |
150 |
6-8% |
High prevalence of arthritis, strong prescription norms |
| Europe |
120 |
5-7% |
Competitive generics market, regulatory constraints |
| Asia-Pacific |
80 |
4-6% |
Growing awareness, improved healthcare access |
| Other Regions |
50 |
2-4% |
Limited healthcare infrastructure |
Pricing Trends
Average wholesale prices for piroxicam tablets declined approximately 10% annually post-patent expiry due to generic competition. The average retail price per 20 mg tablet was around USD 0.20-0.30 in 2022, with regional variance.
Sales Projections (2023-2027)
Assumptions
- Steady population growth of chronic arthritis sufferers at 2% annually.
- Development of new formulations (e.g., topical gels) leading to modest growth.
- Regulatory pressure to limit gastrointestinal side effects reduces prescriber preference in some markets.
- Price erosion continues at 8-10% annually due to generic competition.
Forecast Summary
| Year |
Sales (USD million) |
CAGR |
Notes |
| 2023 |
300 |
0.5% |
Stable market, minor growth from new formulations |
| 2024 |
303 |
1.0% |
Slight increase, new regional approvals possible |
| 2025 |
310 |
2.2% |
Growing awareness of pain management options |
| 2026 |
312 |
0.6% |
Market saturation, price erosion continues |
| 2027 |
315 |
0.9% |
Market stabilizes, slight sales uptick due to generics |
Note: These projections are conservative; key drivers include the aging demographic and chronic disease prevalence.
Competitive Landscape
- No patent protections maintain a highly competitive market with multiple generics.
- Innovations focus on reducing gastrointestinal risk, including delayed-release formulations.
- Piroxicam's market share risks further decline due to emerging NSAIDs with better side-effect profiles.
Strategic Outlook
Pharmaceutical companies focusing on piroxicam should:
- Leverage existing brand recognition in markets with entrenched prescribing habits.
- Invest in formulations reducing adverse effects to improve adherence.
- Expand into emerging markets to offset pricing pressures in mature regions.
Key Takeaways
- Piroxicam's market remains stable but faces declining margins due to generic competition.
- Global sales are projected to grow slowly, buoyed by demographic trends and a focus on pain management.
- Regionally, North America and Europe are the largest markets; growth is expected primarily in Asia-Pacific.
- Price erosion and regulatory concerns about side effects influence sales strategies.
- Innovation in formulations may boost sales, but market share reduction is inevitable over the long term.
FAQs
1. What factors limit piroxicam's market growth?
Price erosion due to generics, safety concerns, and competition from NSAIDs with improved side-effect profiles limit growth.
2. Which regions present the most growth opportunities for piroxicam?
Asia-Pacific due to expanding healthcare access and increasing chronic condition prevalence.
3. How do regulatory restrictions impact piroxicam sales?
Restrictions on NSAIDs' gastrointestinal side effects lead prescribers to opt for alternatives, reducing demand.
4. What innovations are emerging in piroxicam formulations?
Topical gels and delayed-release tablets aim to mitigate side effects and expand user adherence.
5. How long will piroxicam remain in the market?
Until major generic competition diminishes profit margins, typically several more years, unless replaced by newer drugs with superior safety profiles.
References
[1] Grand View Research. (2022). NSAID Market Size, Share & Trends Analysis.
[2] FDA. (2021). Piroxicam Drug Label.
[3] IQVIA. (2022). Global Pain Management Market Reports.
[4] World Health Organization. (2022). Chronic Disease and Aging Population Statistics.
[5] Pharma Intelligence. (2022). Patent Expiry and Generic Launch Data.