Share This Page
Drug Sales Trends for gatifloxacin
✉ Email this page to a colleague
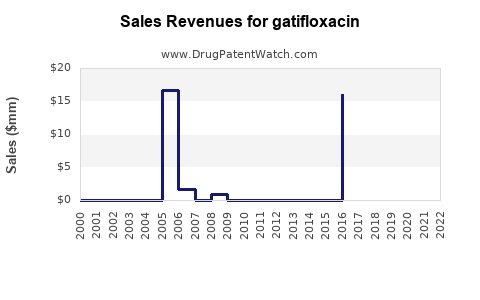
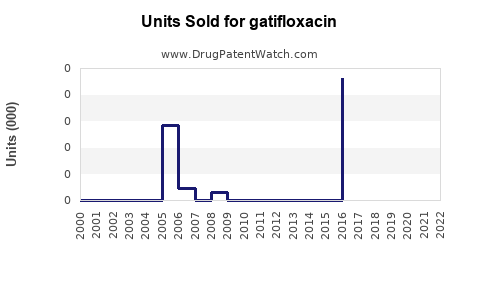
Annual Sales Revenues and Units Sold for gatifloxacin
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| GATIFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| GATIFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| GATIFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| GATIFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| GATIFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| GATIFLOXACIN | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Gatifloxacin
What is the Current Market Landscape for Gatifloxacin?
Gatifloxacin is a fluoroquinolone antibiotic approved primarily for treating bacterial conjunctivitis and urogenital infections. Its market presence has evolved due to regulatory scrutiny and competition from other antibiotics.
Regulatory Status
- U.S. Market: Gatifloxacin ophthalmic solutions were withdrawn from the U.S. market in 2006 following reports of dysglycemia, although systemic forms remain available in other regions.
- Global Presence: In certain countries, systemic formulations are still approved for respiratory and urinary tract infections. However, regulatory restrictions impact its global sales potential.
Key Market Players
- GlaxoSmithKline (GSK): Originator of the ophthalmic formulation, withdrawn in the U.S.
- Other Suppliers: Marked by regional variances; in markets where systemic use persists, local manufacturers produce gatifloxacin-based antibiotics.
Competitive Environment
- The antibiotic class is dominated by drugs like ciprofloxacin, levofloxacin, and moxifloxacin.
- In ophthalmology, newer agents such as moxifloxacin (Vigamox) and ofloxacin have replaced gatifloxacin in several markets.
Market Drivers and Barriers
Drivers
- Rising antibiotic resistance fosters demand for broad-spectrum agents.
- High prevalence of bacterial ocular infections in aging populations.
- Regulatory approvals in regions outside the U.S. support localized sales.
Barriers
- Safety concerns related to dysglycemia and QT prolongation.
- Competition from newer fluoroquinolones with improved safety profiles.
- Regulatory restrictions reduce access in key markets like the U.S.
Sales Projections and Market Volume
Estimated Market Size
- Global ophthalmic antibiotic market (2019): approximately USD 2.8 billion, with fluoroquinolones representing around 70% of ophthalmic antibiotics.
- The ophthalmic gatifloxacin segment has declined since its U.S. withdrawal but persists in select regions, especially in Asia, Europe, and Latin America.
Sales Forecast (2023-2028)
| Year | Estimated Sales (USD Millions) | Notes |
|---|---|---|
| 2023 | USD 150 – 250 | Niche markets with ongoing approvals. |
| 2024 | USD 180 – 280 | Market steady in regions with approvals. |
| 2025 | USD 200 – 300 | Slight growth expected. |
| 2026 | USD 220 – 330 | Market expansion in emerging regions. |
| 2027 | USD 230 – 350 | Competitive pressures moderate growth. |
| 2028 | USD 240 – 370 | Stabilization as newer agents gain market share. |
Regional Focus
- Asia-Pacific: Largest growth potential due to high demand for affordable antibiotics and less regulatory restrictions.
- Europe: Moderate growth; presence mainly in countries with active ophthalmic and systemic use.
- North America: Limited; sales driven by systemic uses in countries where safety concerns are less prohibitive.
Key Market Trends
- Increasing use of combination antibiotics reduces demand for monotherapy options like gatifloxacin.
- Rising pharmacovigilance activity impacts the approval and usage of older fluoroquinolones.
- Regulatory agencies seek to restrict systemic gatifloxacin due to safety issues, affecting overall sales.
- Emerging markets show potential for increased sales due to less regulatory oversight and higher prevalence of bacterial infections.
Strategic Opportunities
- Reformulating or repositioning gatifloxacin formulations in markets lacking regulatory restrictions.
- Developing safer derivatives to overcome safety concerns.
- Targeting niche indications where newer drugs lack specific approvals.
Risks and Challenges
- Regulatory restrictions in major markets can limit sales growth.
- Competition from newer, safer fluoroquinolones limits market share.
- Patent expirations and generic competition may impact profitability.
Conclusion
Gatifloxacin’s market is characterized by regional disparities, regulatory constraints, and stiff competition. The global sales outlook for 2023-2028 shows modest growth driven mainly by emerging markets. Opportunities exist in niche applications and reformulation strategies, but safety issues and evolving regulations pose significant barriers.
Key Takeaways
- Gatifloxacin's US market was largely phased out following safety concerns but remains active elsewhere.
- The global ophthalmic antibiotic market is growing, but gatifloxacin's share is declining due to safety and competition.
- Sales are projected to remain in the USD 150–370 million range over the next five years, with regional variation.
- Rising resistance and unmet needs in emerging markets present opportunities.
- Regulatory and safety challenges are the primary obstacles to market expansion.
FAQs
-
What caused Gatifloxacin’s market withdrawal in the U.S.? Safety concerns related to dysglycemia and QT prolongation led to its withdrawal in 2006.
-
In which regions does Gatifloxacin still have market approval? It remains approved in some Asian, European, and Latin American countries for systemic and ophthalmic uses.
-
What are the main competitors to Gatifloxacin in the ophthalmic antibiotic market? Moxifloxacin, levofloxacin, and ofloxacin dominate market share in the ophthalmic segment.
-
What factors could drive future sales of Gatifloxacin? Increased antibiotic resistance, unmet needs in emerging markets, and formulations that address safety issues.
-
How does safety impact Gatifloxacin’s market viability? Safety concerns significantly restrict its use in some markets, limiting sales and growth prospects.
References
[1] GlaxoSmithKline. (2007). Gatifloxacin ophthalmic solution: Product discontinuation. Retrieved from https://www.gsk.com
[2] MarketsandMarkets. (2022). Ophthalmic antibiotics market analysis. Retrieved from https://www.marketsandmarkets.com
[3] U.S. Food and Drug Administration. (2006). Drug safety communications relevant to fluoroquinolones. Retrieved from https://www.fda.gov
[4] Statista. (2021). Global ophthalmic antibiotic market size and forecast. Retrieved from https://www.statista.com
More… ↓
