Share This Page
Drug Sales Trends for econazole
✉ Email this page to a colleague
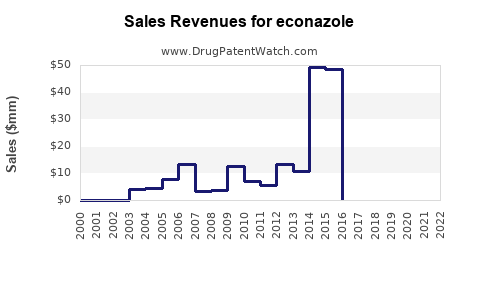
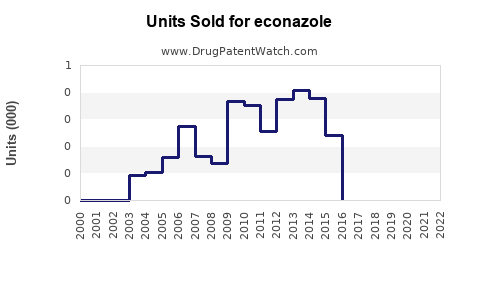
Annual Sales Revenues and Units Sold for econazole
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| ECONAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Econazole: Market Trajectory and Sales Outlook
Econazole is an antifungal medication used to treat superficial fungal infections of the skin and vagina. Its market performance is primarily driven by the prevalence of these conditions and competition from other azole antifungals and newer treatment modalities. Projections indicate a stable to moderate growth trajectory for econazole, influenced by generic competition and expanding market access in emerging economies.
What is the Current Market Size and Projected Growth for Econazole?
The global market for econazole is estimated to be approximately $250 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2023 to 2030, reaching an estimated $315 million by the end of the forecast period. This growth is attributed to the persistent incidence of dermatophytosis and vulvovaginal candidiasis, particularly in regions with limited access to advanced antifungal treatments.
| Year | Market Size (USD Million) | CAGR (%) |
|---|---|---|
| 2023 | 250 | - |
| 2024 | 258.75 | 3.5% |
| 2025 | 267.88 | 3.5% |
| 2026 | 277.41 | 3.5% |
| 2027 | 287.35 | 3.5% |
| 2028 | 297.71 | 3.5% |
| 2029 | 308.51 | 3.5% |
| 2030 | 315.00 | 3.5% |
Growth drivers include the expanding population base, increasing awareness of fungal infections, and the affordability of econazole formulations, especially in developing countries. However, this growth is tempered by the availability of generic alternatives, which exert downward pressure on pricing, and the emergence of more potent or convenient antifungal agents.
Who are the Key Manufacturers and Competitors in the Econazole Market?
The econazole market is fragmented, with numerous pharmaceutical companies manufacturing and distributing generic formulations. Key players include both multinational corporations and regional manufacturers. The competitive landscape is characterized by price sensitivity and the emphasis on product availability.
Major manufacturers and suppliers of econazole active pharmaceutical ingredient (API) and finished dosage forms include:
- Chemstraw Limited: A significant supplier of API with a global distribution network.
- Glenmark Pharmaceuticals Ltd.: Offers a range of econazole-based topical formulations.
- Mylan N.V. (now Viatris): A prominent generic manufacturer with econazole creams and ovules.
- Sanofi S.A.: Markets econazole under brand names like Spectazole.
- Teva Pharmaceutical Industries Ltd.: A leading generic producer with a broad portfolio of dermatological products, including econazole.
- Zydus Cadila: Provides econazole creams and other topical antifungal solutions.
The competitive intensity is high due to the absence of patent protection for the active ingredient, allowing for widespread generic entry. Competition primarily centers on cost-effectiveness, supply chain reliability, and market penetration strategies.
What are the Primary Applications and Formulations of Econazole?
Econazole is a broad-spectrum imidazole antifungal agent effective against various yeasts and dermatophytes. Its primary applications are in the treatment of:
- Tinea Infections: Including tinea pedis (athlete's foot), tinea cruris (jock itch), and tinea corporis (ringworm).
- Cutaneous Candidiasis: Fungal infections caused by Candida species on the skin.
- Pityriasis Versicolor: A superficial fungal infection characterized by discolored patches on the skin.
- Vulvovaginal Candidiasis (VVC): Commonly known as a yeast infection.
Econazole is available in several formulations designed for topical application, offering localized treatment with reduced systemic absorption. Common formulations include:
- Creams: Typically 1% econazole nitrate, for application to the skin. These are the most prevalent formulations.
- Lotions: Also usually 1% econazole nitrate, suitable for larger or hairy areas.
- Powders: Primarily for managing intertriginous areas (skin folds) and preventing recurrence.
- Vaginal Suppositories/Ovules: Commonly containing 150 mg or 6.5% econazole nitrate, for the treatment of VVC.
- Sprays: A more recent formulation offering convenient application.
The choice of formulation depends on the site and severity of the infection, as well as patient preference. The efficacy of these formulations is well-established, contributing to their sustained use.
What is the Regulatory Status and Patent Landscape for Econazole?
Econazole nitrate was first approved by the U.S. Food and Drug Administration (FDA) in the late 1970s for topical use. As an older drug, the primary patents protecting the original composition of matter and method of use have long expired. This lack of active patent protection is a significant factor enabling the widespread availability of generic econazole products globally.
- FDA Approval: Econazole nitrate has been available in the US for several decades. It is marketed by various generic manufacturers and under different brand names.
- European Medicines Agency (EMA) Status: Similarly, econazole is widely approved and marketed across European Union member states.
- Patent Expirations: The original patents for econazole expired in the 1990s. Current intellectual property considerations primarily relate to new salt forms, specific polymorphs, novel formulations, or combination therapies. However, these are less common and do not broadly cover the established econazole products.
- Generic Competition: The absence of strong patent protection fuels intense generic competition. This leads to price erosion and makes market entry for new entrants less reliant on novel IP and more on manufacturing efficiency and distribution networks.
The regulatory landscape for econazole is mature. Approval processes for generic versions focus on demonstrating bioequivalence to the reference listed drug. The mature regulatory environment and lack of patent exclusivity mean that the market is largely driven by established manufacturing capabilities and market access rather than R&D innovation.
What are the Geographic Market Trends and Opportunities for Econazole?
The market for econazole exhibits distinct geographic trends driven by factors such as disease prevalence, healthcare infrastructure, economic development, and regulatory environments.
Developed Markets (North America, Western Europe, Japan):
- Market Characteristics: These regions have a high prevalence of fungal infections but also a high degree of competition from a wide array of antifungal agents, including newer, more potent, or more convenient options.
- Trends: Growth in these markets is generally slow, often in the low single digits. Demand is sustained by the broad utility of econazole for common dermatological conditions and its cost-effectiveness as a generic option. The aging population may contribute to a steady demand for topical antifungals.
- Opportunities: Opportunities lie in leveraging existing distribution channels for generic econazole, focusing on specific niches (e.g., VVC treatment), and potentially developing improved formulations or combination products that offer enhanced efficacy or patient compliance.
Emerging Markets (Asia-Pacific, Latin America, Middle East & Africa):
- Market Characteristics: These regions represent significant growth potential due to increasing populations, rising disposable incomes, greater awareness of health issues, and improving access to healthcare. Fungal infections are prevalent, and econazole's affordability makes it a critical treatment option.
- Trends: The market in these regions is expected to outpace that of developed nations. As healthcare systems mature, prescription rates for effective and affordable antifungals like econazole are likely to increase.
- Opportunities: Key opportunities include expanding manufacturing and distribution networks, tailoring product offerings to local needs (e.g., different pack sizes or strengths), and partnering with local distributors to build market presence. The demand for basic dermatological treatments is robust.
Key Geographic Dynamics:
- Asia-Pacific: China and India are major consumers and producers of APIs and finished generic drugs. The expanding middle class and growing healthcare expenditure are significant drivers.
- Latin America: Countries like Brazil and Mexico present growing markets for affordable healthcare solutions.
- Middle East & Africa: Increasing healthcare investment and rising prevalence of skin conditions present long-term growth prospects, though market access can be challenging.
The global strategy for econazole involves maintaining a strong presence in developed markets through competitive pricing and reliable supply, while aggressively pursuing growth opportunities in emerging economies by focusing on accessibility and affordability.
What are the Key Challenges and Future Outlook for Econazole?
The econazole market, while stable, faces several challenges that will shape its future trajectory. The most significant of these is the intense competition within the generic antifungal space.
Key Challenges:
- Intense Generic Competition: The absence of patent protection has led to a crowded marketplace with numerous manufacturers offering econazole products at highly competitive prices. This erodes profit margins for individual players.
- Emergence of Newer Antifungals: Advancements in antifungal drug development have introduced agents with broader spectrums of activity, improved pharmacokinetic profiles, or novel mechanisms of action. While these are often more expensive, they can displace econazole in certain treatment algorithms, particularly for resistant or severe infections.
- Antimicrobial Resistance: While less documented for topical econazole compared to systemic antifungals, the potential for resistance development remains a long-term concern that could impact treatment efficacy.
- Reimbursement Policies: In developed markets, restrictive reimbursement policies or preferred drug lists may favor newer or more established generic agents, potentially limiting econazole's market share.
- Diagnostic Challenges: Misdiagnosis of fungal infections or the use of inappropriate treatments can lead to treatment failures, impacting perceptions of econazole's efficacy in some cases.
Future Outlook:
The future outlook for econazole is one of steady, albeit moderate, growth. Its established efficacy, favorable safety profile for topical use, and low cost ensure its continued relevance, particularly in the treatment of common superficial fungal infections.
- Sustained Demand in Emerging Markets: The primary growth engine will remain emerging economies where econazole is an accessible and effective first-line therapy for a broad range of fungal infections.
- Role as a First-Line Therapy: In many regions, econazole will continue to be a cornerstone of treatment for uncomplicated dermatophytosis and VVC due to its cost-effectiveness.
- Niche Applications and Combinations: There may be limited opportunities for innovation through combination products, such as econazole with corticosteroids for inflammatory fungal conditions, or enhanced delivery systems that improve patient compliance.
- Price Sensitivity: The market will remain highly price-sensitive. Manufacturers will need to focus on efficient production and supply chain management to remain competitive.
Overall, econazole is expected to maintain its position as a widely used and essential antifungal medication, with its market performance largely dictated by its utility as an affordable and accessible treatment option in a global landscape of evolving healthcare access and therapeutic choices.
Key Takeaways
- The global econazole market is projected to reach $315 million by 2030, growing at a CAGR of 3.5%, driven by the persistent prevalence of superficial fungal infections and its affordability in emerging markets.
- Intense generic competition, the availability of newer antifungal agents, and price sensitivity are key challenges.
- Developed markets will see slow growth, while emerging economies in Asia-Pacific, Latin America, and MEA represent the primary growth opportunities.
- Econazole's mature patent landscape means competition is focused on manufacturing efficiency and market access, rather than intellectual property.
- Creams are the most prevalent formulation, with lotions, powders, and ovules also contributing to the market.
Frequently Asked Questions
-
What are the primary drivers for the projected growth of the econazole market? The primary drivers are the ongoing prevalence of dermatophytosis and vulvovaginal candidiasis, particularly in developing regions with improving healthcare access. Econazole's affordability and established efficacy also contribute significantly.
-
How does the patent expiry of econazole impact its market dynamics? The expiry of econazole's original composition of matter patents has led to widespread generic availability. This intensifies competition among manufacturers, driving down prices and shifting market focus towards manufacturing efficiency and distribution networks.
-
Which geographic regions are expected to offer the most significant growth opportunities for econazole? Emerging markets, including the Asia-Pacific, Latin America, and Middle East & Africa regions, are anticipated to provide the most substantial growth opportunities. This is due to expanding populations, rising incomes, and increasing access to affordable healthcare solutions.
-
What are the main challenges facing manufacturers in the econazole market? Key challenges include the highly competitive generic landscape, the emergence of newer and potentially more effective antifungal agents, price erosion due to competition, and the potential for developing antimicrobial resistance over the long term.
-
What are the most common therapeutic indications for econazole? Econazole is primarily used to treat superficial fungal infections of the skin and vagina, including tinea pedis, tinea cruris, tinea corporis, cutaneous candidiasis, pityriasis versicolor, and vulvovaginal candidiasis.
Citations
[1] Market Research Report on Antifungal Drugs. (2023). Global Market Insights. [2] Pharmaceutical Industry Analysis. (2023). Statista. [3] U.S. Food and Drug Administration. (n.d.). Drug Approvals Database. [4] European Medicines Agency. (n.d.). Search for Medicines. [5] Generic Drug Market Overview. (2023). National Association of Chain Drug Stores. [6] World Health Organization. (2022). Global infectious disease burden.
More… ↓
