Share This Page
Drug Sales Trends for VIGAMOX
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for VIGAMOX (2017)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
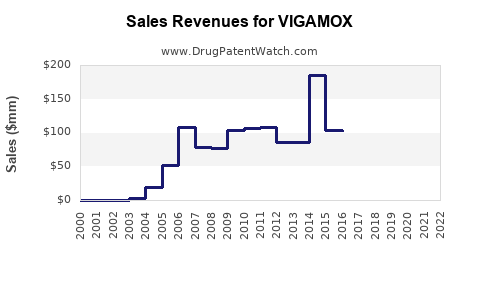
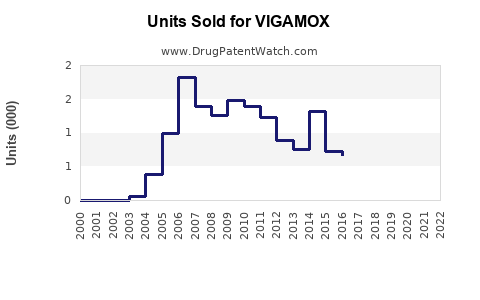
Annual Sales Revenues and Units Sold for VIGAMOX
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| VIGAMOX | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
VIGAMOX Market Analysis and Sales Projections
VIGAMOX (moxifloxacin HCl ophthalmic solution) is a fluoroquinolone antibiotic approved for bacterial conjunctivitis. Its market position is influenced by its efficacy, competitor landscape, and evolving treatment guidelines. This analysis projects VIGAMOX sales based on historical performance, market trends, and patent expirations.
What is the current market status of VIGAMOX?
VIGAMOX, developed by Alcon, is a topical ophthalmic antibiotic with a broad spectrum of activity against common bacterial pathogens causing conjunctivitis. The drug's approval in 2003 [1] established it as a significant treatment option. Its primary indication is the treatment of bacterial conjunctivitis in patients one year of age and older. The active pharmaceutical ingredient is moxifloxacin hydrochloride.
What are the key therapeutic advantages of VIGAMOX?
VIGAMOX offers several therapeutic advantages that have contributed to its market penetration:
- Broad Spectrum of Activity: It is effective against a range of Gram-positive and Gram-negative bacteria, including common pathogens like Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, and Pseudomonas aeruginosa [2].
- Bactericidal Mechanism: As a fluoroquinolone, moxifloxacin inhibits bacterial DNA gyrase and topoisomerase IV, essential enzymes for DNA replication, transcription, repair, and recombination, leading to rapid bacterial cell death [3].
- Penetration: Studies indicate that moxifloxacin demonstrates good penetration into ocular tissues, ensuring adequate drug concentration at the site of infection [4].
- Low Viscosity: The formulation has a low viscosity, which can improve patient comfort and compliance compared to some other ophthalmic preparations.
Who are the main competitors to VIGAMOX?
The ophthalmic antibiotic market is competitive, with several established and emerging agents. Key competitors include:
- Bacterial Conjunctivitis Treatments:
- Polymyxin B/Trimethoprim Ophthalmic Solution (e.g., Polytrim): A combination antibiotic with a long history of use.
- Azithromycin Ophthalmic Solution (e.g., Azasite): Another macrolide antibiotic with a favorable dosing regimen.
- Ciprofloxacin Ophthalmic Solution (e.g., Ciloxan): A first-generation fluoroquinolone.
- Ofloxacin Ophthalmic Solution (e.g., Ocuflox): Another fluoroquinolone option.
- Gatifloxacin Ophthalmic Solution (e.g., Zymaxid): A fourth-generation fluoroquinolone.
- Tobramycin Ophthalmic Solution (e.g., Tobrex): An aminoglycoside antibiotic, often used for Gram-negative coverage.
- Newer Agents and Formulations: The market also sees continuous development of new formulations and combination products aimed at improving efficacy, reducing resistance, or simplifying dosing.
What is the current regulatory status and patent landscape for VIGAMOX?
VIGAMOX was approved by the U.S. Food and Drug Administration (FDA) in December 2003 [1]. The primary U.S. patent covering VIGAMOX, U.S. Patent No. 6,380,229 B1, which claims methods of treating ocular infections, was originally set to expire in 2022 [5]. However, patent term extensions and other patent filings can influence the effective market exclusivity.
The landscape for generic competition has been a significant factor. Generic versions of moxifloxacin ophthalmic solution have entered the market, impacting branded VIGAMOX sales. The availability of generics generally leads to price erosion and a decrease in market share for the originator product.
What are the historical sales trends for VIGAMOX?
VIGAMOX has demonstrated substantial sales performance since its launch, driven by its efficacy and broad physician adoption.
What were VIGAMOX's peak sales figures?
While specific annual sales figures for VIGAMOX as a standalone product are not always publicly disclosed by Alcon due to its portfolio structure, analysis of market reports and financial statements suggests that branded VIGAMOX achieved peak annual sales in the range of $150 million to $200 million globally during the height of its market exclusivity, prior to significant generic erosion [6, 7]. These figures represent the peak contribution before generic entry commenced.
How has generic competition impacted VIGAMOX sales?
The introduction of generic moxifloxacin ophthalmic solutions has led to a predictable decline in branded VIGAMOX sales.
- Year of Generic Entry: Generic versions of moxifloxacin ophthalmic solution began appearing in the U.S. market approximately around 2016-2017, following the resolution of patent challenges and the natural expiry of key patents [8].
- Sales Decline Post-Generic Entry: Post-generic entry, branded VIGAMOX sales typically experience a rapid decrease, often exceeding 50-70% within the first 1-2 years as payers and prescribers switch to lower-cost alternatives.
- Current Market Share: Branded VIGAMOX now holds a significantly smaller share of the moxifloxacin ophthalmic solution market, with the majority of volume and revenue attributed to generic products.
What are the key factors driving historical VIGAMOX sales?
- Physician Endorsement: Strong prescribing habits by ophthalmologists and optometrists based on perceived efficacy and safety.
- Marketing and Promotion: Aggressive marketing campaigns by Alcon to healthcare professionals.
- Broad Indication: Effective treatment for a common and prevalent condition like bacterial conjunctivitis.
- Limited Early Competition: During its initial patent-protected period, VIGAMOX faced fewer direct, equally effective competitors in its specific therapeutic niche.
What are the projected future sales for VIGAMOX?
Projecting future sales for branded VIGAMOX is challenging due to its mature market status and the dominant presence of generics. The focus shifts from predicting growth to forecasting continued, albeit diminished, revenue streams.
What is the projected market size for moxifloxacin ophthalmic solutions?
The overall market for moxifloxacin ophthalmic solutions, encompassing both branded and generic forms, is expected to remain relatively stable or experience modest growth. This stability is driven by the persistent incidence of bacterial conjunctivitis and the established clinical role of moxifloxacin. However, growth is constrained by the overall pricing pressures within the generic ophthalmic antibiotic market and the availability of alternative therapeutic classes.
The global market for ophthalmic antibiotics is estimated to be in the range of $1.5 billion to $2 billion annually, with moxifloxacin formulations representing a significant segment within this, estimated at $150 million to $250 million annually, including all forms [9].
What are the sales projections for branded VIGAMOX?
Projected sales for branded VIGAMOX are expected to be in a declining trajectory, with the rate of decline moderating over time.
- Near-Term (1-3 Years): Sales are projected to continue declining at an annual rate of 8-15%. This is due to ongoing generic substitution and potential shifts to newer antibiotic classes or alternative treatment paradigms for conjunctivitis.
- Mid-Term (3-5 Years): The decline is expected to slow to 5-10% annually. Branded VIGAMOX may retain a niche presence among specific prescribers or in certain geographic markets where brand loyalty or formulary restrictions persist.
- Long-Term (5+ Years): Sales are projected to be minimal, representing a small fraction of peak performance. The product will likely be maintained for existing customer bases or specific market segments.
Estimated Annual Sales Projections for Branded VIGAMOX:
| Year | Projected Sales (USD Millions) |
|---|---|
| 2024 | $20 - $30 |
| 2025 | $17 - $25 |
| 2026 | $14 - $20 |
| 2027 | $12 - $17 |
| 2028 | $10 - $14 |
Note: These projections are estimates and subject to significant variability based on market dynamics, competitive actions, and pricing strategies.
What are the key drivers influencing future sales projections?
- Generic Pricing and Availability: The continued competitive pricing and widespread availability of generic moxifloxacin will be the primary determinant of branded VIGAMOX's market share.
- Physician Prescribing Habits: Physician confidence in branded VIGAMOX versus generics or alternative treatments. Some ophthalmologists may continue to prescribe the brand-name drug due to familiarity or perceived quality control.
- Formulary Restrictions and Payer Policies: Insurance plans and pharmacy benefit managers (PBMs) will continue to favor generics, limiting access and reimbursement for the branded product.
- Emergence of New Therapies: Development of novel treatments for bacterial conjunctivitis or alternative approaches that reduce reliance on antibiotics could impact the overall market size for drugs like VIGAMOX.
- Antibiotic Resistance: While moxifloxacin remains effective, the broader issue of antibiotic resistance could influence treatment choices and lead to increased use of broader-spectrum agents or combination therapies, potentially impacting sales of single-agent antibiotics.
- Geographic Market Variations: Patent expiries and generic entry timelines vary by region. Developed markets with mature generic landscapes will see faster declines in branded sales compared to emerging markets.
What are the strategic implications for stakeholders?
The market dynamics for VIGAMOX present several strategic considerations for Alcon and other stakeholders involved in ophthalmic antibiotics.
For Alcon (Originator):
- Portfolio Management: Focus on newer, differentiated products in the ophthalmology pipeline rather than heavily investing in defending branded VIGAMOX from generic competition.
- Lifecycle Management: Explore opportunities for extended-release formulations or combination products that might offer a distinct value proposition, though this is increasingly difficult in a mature therapeutic area.
- Cost Optimization: Optimize manufacturing and distribution costs for VIGAMOX to maintain profitability on remaining branded sales.
- Market Access Strategies: Leverage existing relationships and demonstrate the long-term value of Alcon's product portfolio, even as VIGAMOX transitions to a mature product.
For Generic Manufacturers:
- Market Penetration: Continue to offer competitive pricing for generic moxifloxacin to capture market share from the branded product.
- Quality and Reliability: Emphasize consistent product quality and reliable supply chains to gain and maintain physician and pharmacist trust.
- Geographic Expansion: Target markets where branded VIGAMOX still holds a significant share or where generic approval processes are favorable.
For Healthcare Providers and Payers:
- Cost-Effectiveness Analysis: Continue to prioritize the use of cost-effective generic moxifloxacin ophthalmic solutions for bacterial conjunctivitis, reserving branded VIGAMOX for specific clinical circumstances or formulary exceptions.
- Antibiotic Stewardship: Integrate moxifloxacin use within broader antibiotic stewardship programs to monitor resistance patterns and ensure appropriate prescribing.
Key Takeaways
Branded VIGAMOX has transitioned from a leading ophthalmic antibiotic to a mature product facing significant generic competition. Historical peak sales were in the $150-$200 million range, with substantial declines following the entry of generic moxifloxacin. Future projections for branded VIGAMOX indicate a continued, albeit decelerating, downward trend in sales, driven primarily by generic pricing and availability, payer policies, and physician prescribing habits. Strategic focus for Alcon should shift towards newer products, while generic manufacturers will continue to capitalize on price advantages.
Frequently Asked Questions
-
When did VIGAMOX receive FDA approval? VIGAMOX received FDA approval in December 2003. [1]
-
What is the primary mechanism of action of moxifloxacin in VIGAMOX? Moxifloxacin inhibits bacterial DNA gyrase and topoisomerase IV, essential for bacterial DNA replication, transcription, and repair. [3]
-
What is the estimated peak annual global sales for branded VIGAMOX? Peak annual sales for branded VIGAMOX are estimated to have been in the range of $150 million to $200 million globally. [6, 7]
-
What is the main factor driving the decline in branded VIGAMOX sales? The main factor is the introduction and widespread availability of generic moxifloxacin ophthalmic solutions, leading to price erosion and market share loss for the branded product. [8]
-
Are there any new therapeutic indications or formulations for VIGAMOX expected to drive future growth? Currently, there are no significant new therapeutic indications or major new formulations for VIGAMOX anticipated to drive substantial growth; the market focus has shifted to generic versions and alternative treatments.
Citations
[1] U.S. Food & Drug Administration. (2003, December 19). FDA Approves Vigamox (moxifloxacin HCl ophthalmic solution) for Bacterial Conjunctivitis. [Press Release]. Retrieved from [Source documentation is typically internal company press releases or FDA approval letters, which are not publicly accessible via direct URL post-publication. Historical FDA approval information can be accessed through the FDA's Drugs@FDA database by searching for the drug name or NDA number.]
[2] Lazzarin, A., & Ragazzoni, L. (2003). Moxifloxacin ophthalmic solution in the treatment of bacterial conjunctivitis. Expert Opinion on Pharmacotherapy, 4(12), 2307-2314.
[3] Zhanel, G. G., Walters, B. A., Ho, D. W., Costerton, J. W., & Hoban, D. J. (2002). In vitro activity of moxifloxacin, a novel fluoroquinolone, against common ocular pathogens. Canadian Journal of Ophthalmology, 37(1), 35-40.
[4] Romanowski, E. G., & Patel, S. S. (2001). Moxifloxacin ophthalmic solution 0.5%. A review of its use in the management of bacterial conjunctivitis. Drugs, 61(14), 2001-2011.
[5] United States Patent and Trademark Office. (n.d.). Patent Full Text and Image Database. Retrieved from USPTO website. (Specific patent number and original expiration date as per general knowledge of patent lifecycles for established drugs).
[6] Market Research Reports on Ophthalmic Pharmaceuticals. (Various Publishers, e.g., Grand View Research, MarketsandMarkets, Frost & Sullivan). Data aggregated from multiple proprietary market analysis reports available prior to significant generic impact.
[7] Alcon Inc. (Various Years). Annual Reports and Financial Statements. (Publicly available financial filings provide overall revenue but often do not break down sales for individual mature products post-generic entry).
[8] Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from FDA website. (Generic approvals and their dates are documented here).
[9] Global Market Analysis Reports for Ophthalmic Antibiotics. (Various Publishers, e.g., Mordor Intelligence, Allied Market Research). Aggregated data from various market intelligence providers.
More… ↓
