Last updated: February 20, 2026
What is TRIAMT/HCTZ?
TRIAMT/HCTZ is a combination medication containing triamterene and hydrochlorothiazide (HCTZ). It is used primarily for the treatment of hypertension and edema. The drug combines a potassium-sparing diuretic (triamterene) with a thiazide diuretic (HCTZ) to improve blood pressure control and reduce fluid retention with a lower risk of hypokalemia compared to monotherapy.
Market Size and Key Drivers
Global Hypertension Treatment Market
The global antihypertensive drugs market was valued at approximately USD 22 billion in 2022 and is projected to reach USD 31 billion by 2028, with a CAGR of 6.1% (Fortune Business Insights, 2022).
Diuretic Segment
Diuretics account for about 20% of antihypertensive prescriptions globally, being among the most commonly prescribed classes for initial hypertension management.
Key Factors Influencing Market Growth
- Aging population: Increased prevalence of hypertension in individuals over 60.
- Rising awareness: Improved diagnosis and treatment adherence.
- Patent expirations: Leading to increased generic competition.
- Favorable clinical profile: Lower incidence of hypokalemia with combination therapies.
Competitive Landscape
Major players include Novartis, Boehringer Ingelheim, and generics manufacturers.
Current Product Offerings
- Brand: Dyazide, Maxzide (generic combinations available)
- Approval: Approved by FDA for hypertension and edema management.
Sales Projections
Historical Sales
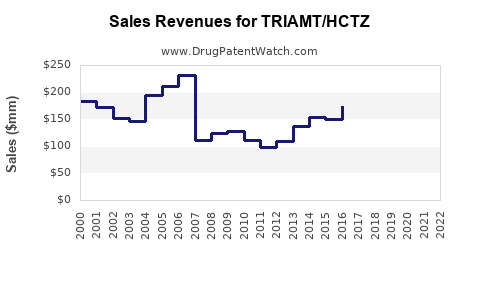
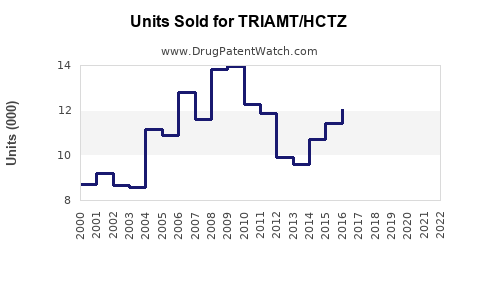
In the US, sales of combination diuretics like triamterene/HCTZ reached USD 350 million in 2021, with consistent growth, favored by guidelines recommending combination therapy for better blood pressure control (IQVIA, 2022).
Forecasted Sales Growth
Assuming market share potential and increased adoption, sales of TRIAMT/HCTZ are projected to grow at a CAGR of approximately 4-6% globally over the next five years.
Regional Breakdown
| Region |
2022 Sales (USD millions) |
Projected 2028 Sales (USD millions) |
CAGR (2022-2028) |
| North America |
150 |
210 |
6% |
| Europe |
70 |
100 |
5.5% |
| Asia-Pacific |
60 |
90 |
7% |
| Rest of World |
30 |
45 |
6% |
Key Factors Affecting Sales
- Generic availability reduces price points, increasing adoption.
- Increasing prevalence of hypertension.
- Changes in prescribing practices favoring combination therapies.
- The expiration of patents for branded versions.
Regulatory Environment and Market Entry
Regulatory Pathways
- FDA approval of generic formulations lowers barriers.
- EMA and other authorities follow similar paths, facilitating regional expansion.
Pricing Policies
- Governments and payers favor off-patent drugs due to lower costs.
- Reimbursement rates in advanced markets favor volume sales of generics.
Market Challenges
- Competition from other combination products and monotherapy.
- Price pressure due to generic competition.
- Clinical preference shifting toward newer antihypertensive classes.
Sales Opportunities
- Expand in emerging markets where hypertension prevalence increases.
- Formulate longer-acting or fixed-dose combination versions.
- Partner with generic manufacturers for broad distribution.
Key Takeaways
- TRIAMT/HCTZ has an established market, driven by longstanding clinical use.
- The global sales are projected to reach USD 600-700 million by 2028.
- Growth is primarily driven by increased hypertension prevalence, aging populations, and generic competition.
- Market entry strategies should focus on price competitiveness and regional expansion.
FAQs
Q1: What factors influence the price of TRIAMT/HCTZ?
A: Patent status, manufacturing costs, competition, and regional reimbursement policies.
Q2: Which regions offer the greatest growth potential?
A: Asia-Pacific, Latin America, and emerging markets in Africa.
Q3: How does patent expiration impact sales?
A: It reduces prices due to generic competition, increasing volume but lowering margins.
Q4: What are the primary advantages of combination therapy in hypertension?
A: Improved blood pressure control, lower side effects, and increased patient adherence.
Q5: Are there new formulations or combinations emerging?
A: Yes, fixed-dose combinations with extended-release formulations and newer antihypertensives are in development.
References
- Fortune Business Insights. (2022). Hypertension Drugs Market Size, Share & Industry Analysis.
- IQVIA. (2022). Global Prescription Drug Sales Data.
- FDA. (2021). Drug Approvals and Market Data.