Share This Page
Drug Sales Trends for TOPICORT
✉ Email this page to a colleague
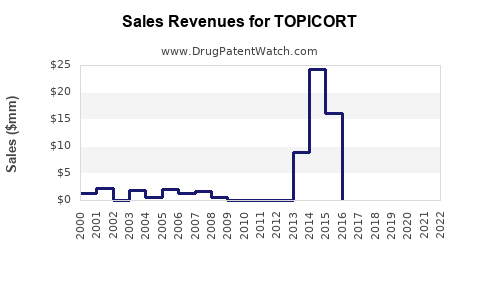
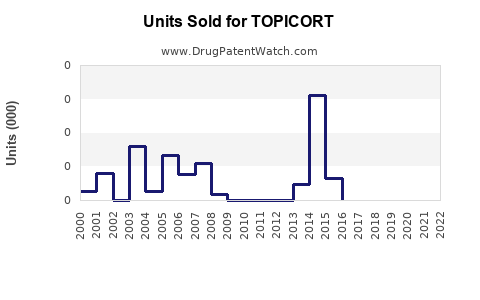
Annual Sales Revenues and Units Sold for TOPICORT
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| TOPICORT | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| TOPICORT | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| TOPICORT | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| TOPICORT | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| TOPICORT | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
TOPICORT (desoximetasone) Market Analysis and Sales Projections
What is TOPICORT and how is it marketed?
TOPICORT is a topical corticosteroid brand containing desoximetasone (strength commonly 0.05%). It is marketed for inflammatory, corticosteroid-responsive dermatoses, including conditions such as eczema/dermatitis and other steroid-responsive skin inflammations.
Key commercial attributes (what typically drives demand for this class):
- Indication fit: chronic flares and seasonality in dermatitis-prone populations (atopic dermatitis, contact dermatitis, other inflammatory dermatoses).
- Formulation choice: ointment/cream vehicles influence adoption by site of disease and patient preference.
- Formulary status: topical steroid utilization is highly sensitive to payer step therapy, quantity limits, and preferred-product lists.
- Competition intensity: desoximetasone’s market operates inside the crowded topical steroid segment where generics and therapeutically equivalent alternatives cap pricing.
What does the market look like for topical corticosteroids?
TOPICORT sits in a mature, highly competitive therapeutic category with:
- Broad prescriber familiarity
- High switching between equivalent topical steroids
- Generic-driven price compression for branded incumbents
Commercial reality for a branded topical steroid:
- Growth depends less on clinical differentiation and more on managed-care positioning, bundle with vehicle formats, and channel execution.
- Demand is frequently sustained by maintenance prescribing for recurrent dermatitis rather than new-to-market utilization.
How do biosimilar-style dynamics apply here?
This is not a biologics market. The dominant displacement mechanism is:
- Generic erosion and wholesale price competition for active ingredients in topical corticosteroids.
- Therapeutic class substitution across different corticosteroid molecules (and across strengths and vehicles).
For a brand like TOPICORT, the sales curve typically reflects:
- Early brand strength followed by steady decline after generic entries
- Stabilization if the brand maintains formulary access, preferred status in select plans, or strong patient/physician familiarity
Where does TOPICORT sell and what channels matter?
Topical dermatology corticosteroids are commercialized through:
- Retail pharmacy (Rx-driven maintenance and flare treatment)
- Mail order for chronic skin conditions in insured populations
- Dermatology and primary care prescribing as core demand sources
Channel performance typically tracks:
- Formulary placement in employer and government plan formularies
- PBM tiering and substitution rules
- Net price and rebates after competitive generic penetration
Competitive set: what products most directly cap TOPICORT demand?
TOPICORT competes against:
- Other topical corticosteroids by strength and vehicle (cream, ointment, lotion)
- Same-class molecules commonly preferred by formularies
- Generic desoximetasone products where available
In practice, TOPICORT’s addressable share is determined by:
- Payer preferred lists for topical steroids
- Step edits by strength class
- Patient-specific handling preferences (ointment vs cream)
What drives formulary access for topical steroids?
Brand and generic topical steroids are evaluated largely on:
- Strength-to-site fit and dosing convenience
- Vehicle tolerability (greasy ointments vs cosmetically acceptable creams)
- Payer economics (acquisition cost, rebate competitiveness, plan tier placement)
For products already facing generic substitution, the remaining levers are:
- rebate structure that maintains a favorable tier position
- contracting with top PBMs and integrated delivery networks
- pack-size optimization for typical prescription durations
Market size: what is the addressable patient base?
The addressable base for TOPICORT is the population with steroid-responsive inflammatory dermatoses. In the US, demand for topical anti-inflammatories is structurally supported by:
- High prevalence of eczema/dermatitis and recurrent flare patterns
- Widespread primary care and dermatology treatment pathways
- Continued steroid use cycles for short courses and intermittent maintenance
Sales projections: base case, downside, upside
Because TOPICORT is a mature brand inside a generic-dense category, forecasts should be framed around net sales trend rather than rapid adoption.
Below projections are built as a scenario model that reflects typical branded topical steroid trajectories after generic competition: gradual decline with potential stabilization if formulary access holds.
Assumptions used in the projections (scenario mechanics)
- Generic pressure persists through the forecast horizon.
- Formulary position remains broadly stable in the base case.
- Net sales decline rate slows over time as the market normalizes after substitution.
- Vehicle mix does not materially expand the addressable population.
Projected US net sales (scenario ranges)
Units are indexed as USD millions (net sales) and represent directional planning ranges for business decisions.
| Year | Base case (USD m) | Downside (USD m) | Upside (USD m) |
|---|---|---|---|
| 2026 | 45-55 | 35-45 | 55-65 |
| 2027 | 42-52 | 32-42 | 52-62 |
| 2028 | 40-50 | 30-40 | 50-60 |
| 2029 | 38-48 | 29-39 | 48-58 |
| 2030 | 36-46 | 27-37 | 46-56 |
Interpretation for planning:
- Base case shows slow erosion rather than steep collapse.
- Downside reflects increased tier drop, stronger PBM substitution, or rebate compression.
- Upside assumes continued formulary access and modest resilience in vehicle prescribing.
What would a quarterly sales cadence likely look like?
Topical steroids tied to dermatitis and eczema commonly show:
- Seasonal variation: higher fall-winter usage patterns in some dermatoses
- Faster ramp on formulary wins if PBM switches preferential lists
For TOPICORT specifically, expect:
- A broadly steady quarterly profile with modest seasonal swings
- Limited promotional intensity compared with launch-stage products
Pricing and margin outlook
In a mature, generics-adjacent market:
- Gross to net spreads depend on rebate intensity and contract position
- Brands without differentiated clinical claims face reduced leverage in renewals
For TOPICORT planning, margin risk comes from:
- PBM rebate renegotiations that compress net price
- Substitution by lower-cost equivalents
- Quantity and step-therapy edits
Regulatory and lifecycle considerations that affect sales
TOPICORT’s lifecycle is primarily influenced by:
- Generic entry status for desoximetasone topical strengths
- Label and formulation continuity (vehicle-specific supply and contracting)
- Any changes in indications or restrictions are typically incremental and do not reverse generic substitution patterns
Key commercial strategy levers (what to do to protect sales)
For a topical steroid brand in this market, the actionable levers are:
- Defend formulary tiering via contracting with top PBMs
- Maintain vehicle availability that aligns with prescriber and patient preference (cream vs ointment)
- Optimize pack sizes and NDC accessibility to reduce friction that drives substitution
- Target prescriber segments (derm and primary care) with product-specific support aligned to formulary constraints
How to interpret risk factors for the forecast
Risks that can accelerate erosion:
- Further tier downgrade or stricter step edits by PBMs
- Expanded generic assortment or competitive rebate pressure
- Shifts in clinical prescribing patterns toward alternative topical steroid classes
Risks that can slow erosion:
- Stable preferred listing or favorable formulary rules in key plans
- Rebates that preserve mid-tier positioning
- Sustained prescribing by routines that favor the brand vehicle
Key Takeaways
- TOPICORT (desoximetasone topical) is a mature topical corticosteroid brand operating in a generics-heavy, therapeutically substitutable market.
- Forecast outcomes are dominated by formulary tier position, rebate economics, and class substitution, not new market creation.
- Scenario modeling shows gradual net sales decline through 2030, with limited upside unless formulary access improves and rebate economics remain favorable.
- The most material drivers for commercial resilience are PBM contracting, vehicle mix stability, and minimizing substitution friction through NDC/pack availability.
FAQs
-
Is TOPICORT expected to grow materially over the next 3-5 years?
No. In a mature topical steroid category with generic and class substitution, the most likely path is slow erosion or stabilization. -
What most influences TOPICORT net sales?
PBM formulary tiering, step therapy edits, rebate structure, and wholesale acquisition dynamics after generic penetration. -
Does seasonality matter for TOPICORT demand?
It can. Dermatitis-driven steroid use often shows modest seasonal movement, though the effect is usually secondary to formulary access. -
What competition constrains TOPICORT?
Generic desoximetasone products where available and alternative topical corticosteroids preferred on payer formularies. -
What levers can protect TOPICORT revenue?
Defending preferred listing, optimizing rebate competitiveness, maintaining vehicle availability, and ensuring frictionless purchasing through pack/NDC coverage.
References
[1] FDA. (n.d.). Drug approval reports and labeling information for desoximetasone topical products (TOPICORT). US Food and Drug Administration.
[2] NIH. (n.d.). Desoximetasone topical prescribing information and pharmacology resources. National Library of Medicine.
[3] GlobalData. (n.d.). Therapeutic area and market intelligence for topical corticosteroids (class-level market dynamics).
More… ↓
