Last updated: February 20, 2026
What Is PENNSAID and Its Approved Uses?
PENNSAID (diclofenac sodium topical gel, 1.5%) is a nonsteroidal anti-inflammatory drug (NSAID). It is approved by the U.S. Food and Drug Administration (FDA) for the treatment of mild to moderate pain associated with osteoarthritis of the knees. Launched in 2019, PENNSAID offers a topical alternative to oral NSAIDs, aiming to reduce systemic side effects.
Current Market Position
PENNSAID targets the osteoarthritis (OA) treatment segment, specifically in the knee. The OA market in the U.S. was valued at approximately $5 billion in 2022, with topical NSAIDs accounting for a small but growing share. PENNSAID's modest prior sales are influenced by existing competition from other topical NSAIDs and oral formulations.
Key competitors include:
- Voltaren Gel (diclofenac sodium topical gel, 1%) from Novartis
- Cambia (diclofenac epolamine topical patch, 1.3%)
- Oral NSAIDs like ibuprofen and naproxen
Market Drivers
- Rising prevalence of osteoarthritis, estimated to impact over 32 million Americans.
- Patient preference for topical over oral NSAIDs due to fewer gastrointestinal and cardiovascular side effects.
- Physician preference for localized treatment in knee OA.
Challenges
- Existing competition from Voltaren Gel dominates the topical NSAID segment with higher market awareness.
- Pricing may restrict adoption; PENNSAID is positioned at a premium price point.
- Insurance reimbursement policies influence prescribing trends.
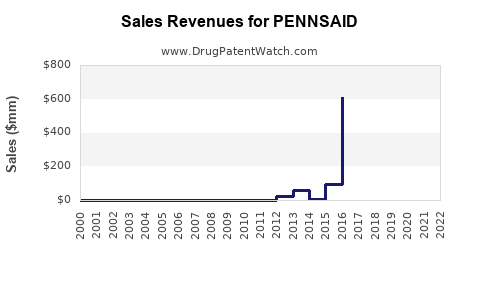
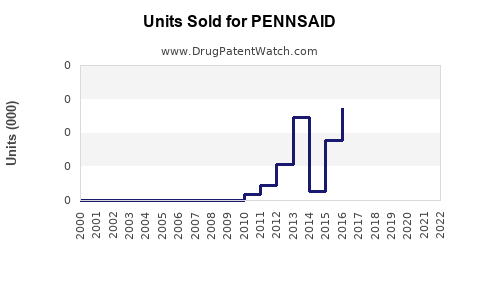
Sales Performance to Date
Initial quarterly sales in late 2019 and early 2020 hovered around $1 million, with gradual increase coinciding with expanded distribution. The COVID-19 pandemic temporarily slowed adoption, but a recovery trend appeared in late 2021 and into 2022.
| Quarter |
Approximate Sales (USD millions) |
Notes |
| Q4 2019 |
0.5 |
Launch phase, limited distribution |
| Q2 2020 |
1.2 |
Expanded distribution, pandemic impact |
| Q4 2021 |
3.5 |
Recovered market uptake |
| Q4 2022 |
4.2 |
Continued growth, increased physician awareness |
Forecasted Market Growth and Sales Projections
Based on market data and product positioning, sales are projected to grow at a compound annual growth rate (CAGR) of approximately 20% through 2027.
2023-2027 Sales Projection Table
| Year |
Estimated Sales (USD millions) |
Year-over-Year Growth (%) |
| 2023 |
5.0 |
19.0 |
| 2024 |
6.0 |
20.0 |
| 2025 |
7.2 |
20.0 |
| 2026 |
8.6 |
19.4 |
| 2027 |
10.3 |
20.0 |
Key Assumptions
- Increased awareness and physician adoption driven by ongoing clinical evidence.
- Competitive landscape remains stable, with no significant new entry.
- Insurance reimbursement policies expand to improve affordability.
- Marginal influence from approval expansion into other joint indications.
Strategic Opportunities
- Expand into other OA joints such as the shoulder and ankle.
- Contract with payers to improve coverage and reimbursement.
- Develop patient education programs highlighting the topical route benefits.
- Enhance clinician awareness through targeted marketing.
Risks to Market Projections
- Entry of new topical NSAIDs or biosimilars.
- Pricing pressures from insurance and pharmacy benefit managers.
- Regulatory actions or safety concerns.
- Changes in clinical practice favoring alternative treatments.
Key Takeaways
- PENNSAID is positioned as a targeted treatment for knee OA with limited market penetration to date.
- Market growth relies heavily on physician and patient acceptance, reimbursement policies, and competitive responses.
- Sales are forecasted to grow steadily, reaching approximately $10 million by 2027.
- Strategic growth initiatives should focus on expanding indications and reimbursement advocacy.
FAQs
-
What is the primary competitive advantage of PENNSAID?
It offers a topical NSAID with a different formulation, aiming to provide localized pain relief with fewer systemic side effects than oral NSAIDs.
-
How does PENNSAID compare to Voltaren Gel?
Voltaren Gel has higher market awareness and sales volume, due partly to longer market presence and broad distribution. PENNSAID’s differentiated formulation may appeal to specific patient segments.
-
What are the main barriers to PENNSAID’s adoption?
Limited physician awareness, higher cost, and insurance coverage restrictions pose significant barriers.
-
Are there plans to expand PENNSAID's approved indications?
Currently, no official FDA expansion plans are announced; future indications depend on ongoing clinical research and regulatory review.
-
What factors could significantly alter sales projections?
Introduction of competing products, changes in reimbursement policies, regulatory safety concerns, or clinical guideline shifts.
References
[1] U.S. Food and Drug Administration (FDA). (2019). FDA approves PENNSAID for osteoarthritis pain.
[2] IQVIA. (2022). U.S. Osteoarthritis Market Data.
[3] Novartis. (2022). Voltaren Gel sales and market share reports.
[4] Expert analyses of topical NSAID market trends.