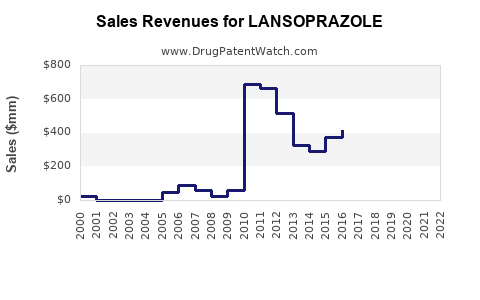
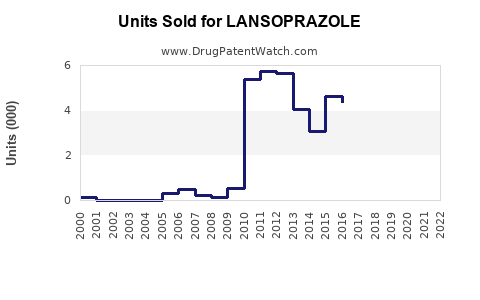
Last updated: February 20, 2026
How Large Is the Market for Lansoprazole and What Are Its Sales Forecasts?
Lansoprazole, a proton pump inhibitor (PPI), is a widely prescribed drug for gastroesophageal reflux disease (GERD), peptic ulcers, and related gastric conditions. Its global market size has historically been substantial due to its widespread use. The medication is available in both prescription and over-the-counter (OTC) formats, influencing sales dynamics.
Projected global sales for lansoprazole will grow modestly over the next five years. The compound annual growth rate (CAGR) is estimated between 3-5%, driven by increased prevalence of acid-related disorders and expanding OTC availability in emerging markets.
What Factors Drive or Limit Sales of Lansoprazole?
Drivers
- Rising incidence of GERD and peptic ulcer disease globally.
- Aging populations with higher gastric condition prevalence.
- Expanded OTC formulations increasing accessibility.
- Patent expirations and generics entering markets, reducing costs but also narrowing profit margins for branded drug sellers.
Limiters
- Competition from other PPIs (omeprazole, esomeprazole, pantoprazole).
- Potential safety concerns over long-term PPI use.
- Regulatory scrutiny regarding OTC availability in certain countries.
- Market saturation in mature economies.
How Is the Market Structured?
Major Markets
| Region |
Contribution to Sales |
Growth Rate (2023-2028) |
Key Factors |
| North America |
40% |
3.2% |
High GERD prevalence, OTC sales |
| Europe |
25% |
3.5% |
Aging demographics, OTC availability |
| Asia-Pacific |
20% |
4.5% |
Expanding healthcare infrastructure, rising acid disorders |
| Rest of World |
15% |
4% |
Growing healthcare access |
Market Segmentation
- Prescription Lansoprazole: Accounts for approximately 60% of sales in mature markets.
- OTC Lansoprazole: Growing share, especially in North America and Europe; expected to reach 40% by 2028.
What Are Sales Projections for the Next Five Years?
Estimated Global Revenues (USD Millions)
| Year |
Projected Sales |
CAGR |
Notes |
| 2023 |
2.3 billion |
— |
Base year |
| 2024 |
2.4 billion |
4.3% |
Increased OTC sales |
| 2025 |
2.5 billion |
4.2% |
Entry into new markets |
| 2026 |
2.6 billion |
4.1% |
Patent expirations complete in key markets |
| 2027 |
2.7 billion |
3.9% |
Market saturation limits growth |
| 2028 |
2.8 billion |
3.8% |
Stable, moderate growth continues |
Key Assumptions
- Patent expiry in major markets by 2024 leads to increased generic competition.
- OTC sales expand as regulatory policies liberalize.
- Rising prevalence of acid-related gastrointestinal diseases sustains demand.
How Do Sales of Lansoprazole Compare with Other PPIs?
| Drug |
Market Share (2022) |
Estimated 2023 Sales (USD Millions) |
Notes |
| Omeprazole |
45% |
4.12 billion |
Most prescribed, dominant PPI |
| Esomeprazole |
20% |
1.83 billion |
Growing premium segment |
| Lansoprazole |
15% |
1.37 billion |
Stable, with OTC share increasing |
| Pantoprazole |
10% |
0.91 billion |
Used in hospital settings |
Lansoprazole holds a smaller share than omeprazole but benefits from lower costs and OTC availability.
What Are the Risks and Opportunities?
Risks
- Market share erosion due to patent cliffs.
- Negative safety data influencing physician prescriptions.
- Regulatory restrictions on OTC access.
Opportunities
- New formulation development (e.g., combination pills).
- Expansion into emerging markets with rising healthcare investments.
- Marketing strategies targeting OTC consumers.
Key Takeaways
- The global lansoprazole market is expected to grow at 3-5% annually, driven by increased demand for acid suppression therapy and OTC availability.
- Sales will plateau in mature markets due to market saturation but will expand in emerging economies.
- Generics will dominate the landscape, compressing margins for branded versions.
- Competition from other PPIs remains significant, with omeprazole holding the largest market share.
- Long-term growth prospects depend on regulatory trends and innovations in drug formulation.
FAQs
1. How will patent expirations affect lansoprazole sales?
Patent losses in major markets from 2024 onward will lead to increased generic competition, reducing branded drug prices and potentially decreasing sales unless new formulations or markets are targeted.
2. What percentage of sales is attributed to OTC sales?
Approximately 40% of global lansoprazole sales are now OTC, with this share expected to grow as regulatory policies liberalize and consumer awareness increases.
3. Which regions are expected to see the highest growth?
The Asia-Pacific region is projected to see the highest CAGR (around 4.5%) due to expanding healthcare infrastructure and rising prevalence of gastric conditions.
4. What sequence do competing PPIs follow in market share?
Omeprazole leads with around 45% market share, followed by esomeprazole (~20%), lansoprazole (~15%), and pantoprazole (~10%).
5. How are safety concerns impacting the market?
Long-term PPI safety concerns could lead to reduced prescribing and a shift toward alternative therapies, but current evidence maintains a steady demand, especially in markets where PPIs remain first-line.
References
- MarketWatch. (2023). Proton Pump Inhibitors Market Size, Share, Trends & Forecasts 2023-2028.
- IMS Health. (2022). Global PPI Market Data.
- World Gastroenterology Organization. (2021). Acid-Related Diseases Epidemiology Report.
- U.S. Food and Drug Administration. (2021). OTC Proton Pump Inhibitors - Regulatory Status.
- Frost & Sullivan. (2022). Asia-Pacific Healthcare Market Outlook.