Last updated: February 14, 2026
Overview of Duexis
Duexis combines ibuprofen and famotidine, marketed by Horizon Therapeutics. It targets adult patients requiring anti-inflammatory therapy with a reduced risk of gastrointestinal side effects. The drug is primarily prescribed for rheumatoid arthritis and osteoarthritis.
Market Context
The global non-steroidal anti-inflammatory drug (NSAID) market was valued at approximately $13.6 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2022 to 2030 [1]. The segment including combination drugs such as Duexis represents a subset of this market, driven by need for gastrointestinal safety management.
Key competitors in this niche include:
- Celecoxib (Celebrex): A selective COX-2 inhibitor.
- Traditional NSAIDs (e.g., diclofenac, naproxen) often co-prescribed with proton pump inhibitors.
- Other combination drugs that include gastric protection agents.
Regulatory and Market Position
Duexis benefits from FDA approval for rheumatoid arthritis and osteoarthritis, with prescribing patterns influenced by safety concerns over gastrointestinal bleeding associated with NSAIDs. However, its market penetration remains limited relative to standalone NSAIDs or COX-2 inhibitors.
Current Sales Performance
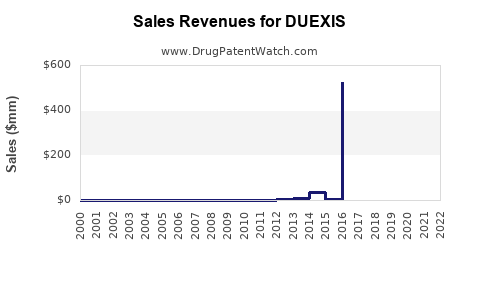
In 2021, Duexis generated approximately $120 million in US sales, representing a modest share of the NSAID market segment focused on combination therapies with gastroprotective agents. Market access challenges, including insurance coverage restrictions and competition, have limited broader adoption.
Sales Projections
Future sales depend on multiple factors:
- Market Penetration: Estimated to grow at a CAGR of 3-5% over five years. Increased awareness among prescribers and expanded insurance coverage could accelerate growth.
- Patent and Patent Exclusivity: Duexis's primary patent expired in late 2022, increasing generic competition from ibuprofen and famotidine manufacturers.
- Pricing Dynamics: Brand name prices are significantly higher than generics, leading to pricing pressures post-patent expiry.
- Physician Preferences: Growing adoption of selective COX-2 inhibitors with better safety profiles may erode demand for combination NSAIDs.
Assuming steady growth and considering market dynamics, sales are projected as follows:
| Year |
Estimated Sales (USD Millions) |
Notes |
| 2022 |
$125 |
Post-patent expiration, initial decline begins |
| 2023 |
$105 |
Increased generic competition, price erosion |
| 2024 |
$90 |
Market stabilization, prescriber shift |
| 2025 |
$80 |
Competition intensifies, alternative therapies grow |
By 2026, sales could stabilize around $70-$80 million unless new indications or formulations are developed.
Potential Growth Drivers
- New Indications: Expansion into other inflammatory or pain-related conditions.
- Formulation Innovations: Development of sustained-release or combination formulations.
- Market Expansion: Penetration into international markets with unmet needs, especially where NSAID safety concerns are prevalent.
Risks and Challenges
- Generic Competition: Entry of multiple generic versions of ibuprofen and famotidine post-patent expiry.
- Pricing Pressure: Insurance reimbursement policies favor generics.
- Alternative Therapies: Increasing preference for drugs with improved safety profiles, such as celecoxib.
Regulatory Environment
Regulatory agencies are scrutinizing NSAID safety. The rise of COX-2 inhibitors with better gastrointestinal safety profiles has impacted Duexis’s market share. Future approvals or label changes could influence sales.
Key Takeaways
- Duexis remains a niche drug within the broader NSAID class, with an estimated US market size of $120 million in 2021.
- Sales are expected to decline post-patent expiration, influenced by generic competition and changing prescriber preferences.
- Long-term growth depends on expanding indications, innovation, and market expansion strategies.
- Pricing and insurance reimbursement will continue to impact revenue potential.
FAQs
-
How does Duexis differ from other NSAIDs?
It combines ibuprofen with famotidine, aiming to provide anti-inflammatory effects while reducing gastrointestinal side effects common with NSAID therapy.
-
What is the outlook for Duexis’s market share?
Likely to decrease due to patent expiry, with potential stabilization if new formulations or indications emerge.
-
Who are the main competitors for Duexis?
Celecoxib, other NSAIDs with gastroprotective agents, and emerging COX-2 inhibitors.
-
How does patent expiration impact sales?
Expiry allows generic manufacturers to enter the market, leading to price competition and sales decline for branded Duexis.
-
Are there regulatory concerns affecting Duexis?
Ongoing safety evaluations of NSAIDs and competition from alternatives influence its market viability.
Sources
[1] Grand View Research, "NSAID Market Size, Share & Trends Analysis," 2022.