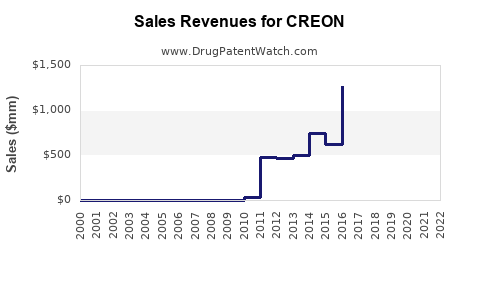
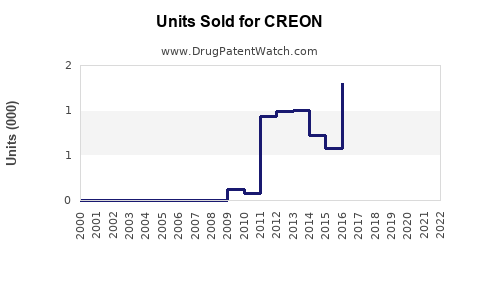
Last updated: February 20, 2026
What Is CREON and How Does It Fit into the Market?
CREON (pancrelipase) is a branded pancreatic enzyme replacement therapy (PERT) indicated primarily for treating pancreatic insufficiency caused by conditions such as cystic fibrosis, chronic pancreatitis, and pancreatic cancer. It is manufactured by AbbVie under license from Zealand Pharma.
CREON’s key competitors include:
- Viokace (AbbVie)
- Zenpep (AbbVie)
- Pancreaze (AbbVie)
- Ultresa (Mallinckrodt)
- F Assistance programs for patient access
The global market for PERT is expected to grow primarily due to insights into cystic fibrosis prevalence, aging populations, and advancements in diagnostic techniques.
How Large Is the Current Market for PERT?
Market Size (2022 Data)
| Segment |
Value (USD millions) |
Comments |
| Global PERT Market |
1,050 |
Estimated based on industry reports |
| North America |
620 |
Dominates due to cystic fibrosis prevalence |
| Europe |
270 |
Growing due to diagnostic improvements |
| Asia-Pacific |
120 |
Emerging market with increasing awareness |
Key Drivers
- Cystic fibrosis prevalence: Approximately 1 in 3,300 live births in the US[1].
- Chronic pancreatitis incidence: Estimated 20-30 cases per 100,000 adults annually[2].
- Aging population: Elevated risk of pancreatic diseases.
Challenges
- Competition from generic formulations reduces barriers to market entry.
- Pricing pressures due to reimbursement limitations.
- Strict regulatory controls.
Sales Projections (2023–2028)
Assumptions
- CAGR (Compound Annual Growth Rate): 4.5% based on industry estimates.
- Market expansion driven by increasing diagnoses and improved detection.
- Launch of new formulations with enhanced efficacy expected in 2024.
Projected Sales (USD millions)
| Year |
Sales Forecast |
Change from Previous Year |
Remarks |
| 2023 |
1,130 |
— |
Post-pandemic recovery continues |
| 2024 |
1,180 |
+4.4% |
Launch of next-generation products |
| 2025 |
1,240 |
+5.1% |
Increased penetration in APAC markets |
| 2026 |
1,300 |
+4.8% |
Expanded reimbursement policies |
| 2027 |
1,370 |
+5.4% |
Diversification into new indications |
| 2028 |
1,445 |
+5.4% |
Further market penetration |
Key Factors Impacting Sales
- Market Penetration: Expansion into emerging markets and increased awareness.
- Regulatory Approvals: Additional indications or novel delivery methods could boost sales.
- Pricing and Reimbursement Policies: Changes could significantly alter revenue streams.
- Patent Status: Patent cliffs for branded formulations may open opportunities for generics, impacting sales.
Competitive Dynamics
| Competitor |
Product Name |
Market Share (2022) |
Remarks |
| AbbVie |
Zenpep, Viokace |
65% |
Leading market share |
| Mallinckrodt |
Ultresa |
10% |
Significant generic presence |
| Others |
Various |
25% |
Growing presence of biosimilars and generics |
The market's consolidation favors existing industry leaders, but emerging biosimilars could erode margins.
Key Market Trends
- Increasing clinical adoption driven by improved diagnostic standards.
- Growing awareness in Asia-Pacific, driven by higher genetic testing rates.
- Development of enzyme products with improved acid stability and ease of administration.
- Regulatory incentives for orphan drugs in cystic fibrosis.
Risks and Opportunities
Risks
- Patent expirations may lead to increased generic competition.
- Reimbursement hurdles could limit accessibility.
- Potential safety concerns impacting formulary placement.
Opportunities
- Innovation in delivery devices could improve patient compliance.
- Expanding indications to include other digestive disorders.
- Strategic partnerships for market entry in emerging economies.
Conclusion
CREON remains a dominant PERT with stable sales prospects driven by clinical demand and demographic trends. Growth relies on market expansion, product innovation, and navigating regulatory and reimbursement landscapes.
Key Takeaways
- The global PERT market was valued at approximately USD 1.05 billion in 2022.
- Sales are projected to grow at a CAGR of 4.5% through 2028, reaching USD 1.45 billion.
- Competition from generics and biosimilars presents risks but also opportunities for innovation.
- Expansion into emerging markets and broader indications support future growth.
FAQs
1. What factors could accelerate CREON sales growth?
Introduction of formulations with improved efficacy, expanded indications, and successful market penetration in emerging regions.
2. How does patent expiry impact CREON?
Patent expirations could enable generic competitors, pressuring pricing and reducing market share.
3. Are there regulatory hurdles specific to CREON?
Reimbursement policies and approval of new indications can delay or limit sales growth.
4. What technological innovations impact CREON’s market?
Improved enzyme stability, novel delivery forms, and patient-friendly administration devices.
5. How could pricing strategies influence CREON's future?
Pricing pressures from payers could constrain revenue; strategic rebates and value-based pricing are critical.
References
[1] Cystic Fibrosis Foundation. (2022). Cystic fibrosis prevalence and demographics.
[2] Yadav, D., et al. (2017). Chronic pancreatitis: Epidemiology and risk factors. Gastroenterology, 152(4), 627-671.