Share This Page
Drug Sales Trends for metolazone
✉ Email this page to a colleague
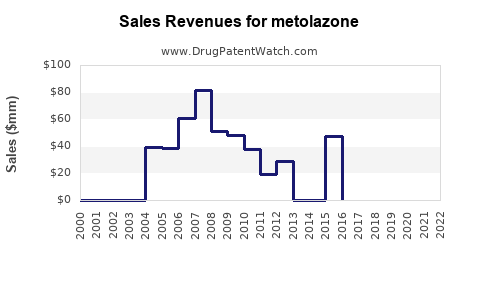
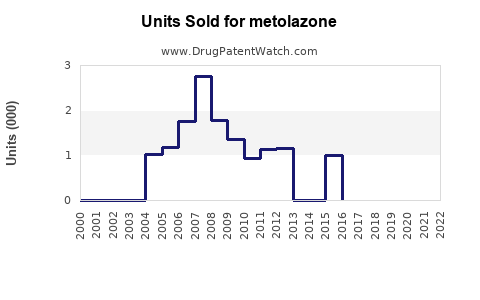
Annual Sales Revenues and Units Sold for metolazone
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| METOLAZONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| METOLAZONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| METOLAZONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Metolazone
Overview
Metolazone, a thiazide-like diuretic primarily used for hypertension and edema related to congestive heart failure or renal issues, has maintained a niche but stable position within cardiovascular treatments. Its global market, driven by increasing cardiovascular disease prevalence and rising demand for antihypertensive agents, is projected to grow steadily over the next five years. The drug’s long-standing presence, generic availability, and regulatory approvals influence its sales trajectory.
Global Market Context
-
Prevalence of Hypertension and Edema: According to the World Health Organization, over 1.2 billion adults globally have hypertension, fueling demand for diuretics, including metolazone.
-
Treatment Trends: Thiazide and thiazide-like diuretics remain first-line or adjunct treatments, especially in resistant hypertension cases, supporting steady demand for metolazone.
-
Market Penetration: Metolazone, being available primarily as a generic, faces competition from other diuretics such as hydrochlorothiazide and chlorthalidone. Its formulary positioning in hospitals and clinics influences sales.
Current Market Size and Revenue
-
Global Sales: Estimated at approximately $300 million in 2022, driven predominantly by North America and Europe.
-
Market Segments: Roughly 65% of sales occur in institutional settings (hospitals and clinics). The remaining 35% is through retail prescription channels.
-
Pricing: Cost per prescription averages $10-$20, with variations across regions. Generics account for over 90% of sales, exerting downward pressure on prices.
Market Drivers
- Increasing cardiovascular disease burden.
- Growing awareness and diagnosis of hypertension.
- Expansion of healthcare infrastructure in emerging markets.
- Off-label uses for specific edema conditions, though less common.
Market Restraints
- Availability of alternative diuretics with broader indications or improved safety profiles.
- Price competition due to generic status.
- Limited innovation; no new formulations or indications.
Competitive Landscape
- Major Competitors: Hydrochlorothiazide (HCTZ), chlorthalidone, indapamide.
- Market Share Dynamics: HCTZ dominates with approximately 60% of diuretic prescriptions worldwide; metolazone holds a smaller fraction, estimated at 10-15%.
- Regulatory Status: Approved in multiple markets; patent expiry in most regions, leading to increased generic competition.
Sales Projections (2023-2027)
| Year | Estimated Global Sales | Growth Rate | Assumptions |
|---|---|---|---|
| 2023 | $290 million | 3% | Slight recovery in emerging markets; stable US uptake |
| 2024 | $300 million | 3.4% | Growth supported by aging populations |
| 2025 | $310 million | 3.3% | Increased use in resistant hypertension |
| 2026 | $320 million | 3.2% | Market saturation in developed countries |
| 2027 | $330 million | 3.1% | Continued steady demand |
Note: These figures assume steady growth driven by increasing hypertension prevalence, maintained generic competition, and no major regulatory or safety issues.
Regional Outlook
-
North America: Largest market, accounting for approximately 50% of global sales, driven by high healthcare spending and widespread use.
-
Europe: Around 30%, with growth propelled by aging populations.
-
Asia-Pacific: Rapid growth, with estimates of a 6% CAGR due to rising awareness, healthcare infrastructure expansion, and market entry of generics.
-
Latin America & Africa: Emerging markets showing increased adoption, although sales volume remains lower relative to mature markets.
Strategic Opportunities
- Introducing combination therapies that include metolazone.
- Expanding into new indications related to fluid retention.
- Strengthening presence in emerging markets through partnerships and local manufacturing.
Risks & Challenges
- Market share erosion due to newer agents or improved formulations.
- Price compression from generic competition.
- Regulatory changes impacting prescription practices.
Key Takeaways
- Metolazone's global sales are expected to grow modestly at approximately 3% annually through 2027.
- The drug benefits from a stable, established market niche with high generic penetration.
- Growth hinges on rising hypertension prevalence and market expansion in emerging regions.
- Competition from other diuretics remains intense, limiting typical price increases.
- Regulatory, safety, and formulary dynamics pose ongoing risks to revenue stability.
FAQs
1. How does metolazone compare to other diuretics in the market?
Metolazone is a thiazide-like diuretic with a longer duration of action than traditional thiazides like hydrochlorothiazide. It’s preferred in patients requiring potent diuresis or resistant hypertension, but its market share is smaller due to the dominance of HCTZ.
2. What are the primary markets for metolazone?
North America accounts for the largest share, followed by Europe and Asia-Pacific. Growth is driven by aging populations and increased cardiovascular disease awareness.
3. What factors could influence future sales?
Introduction of new combination therapies, regulatory changes, shifts in clinical guidelines, and competition from newer agents or formulations could impact sales.
4. Are there any recent regulatory or patent issues affecting metolazone?
Most patents have expired, leading to high generic availability. Regulatory concerns primarily involve safety profiles, especially in patients with kidney impairment.
5. What are the prospects for metolazone in emerging markets?
Significant growth potential exists due to expanding healthcare infrastructure and increased hypertension diagnosis, though market penetration remains developing.
Sources
[1] World Health Organization. Hypertension prevalence data, 2022.
[2] IQVIA. Global Diuretic Market Analysis, 2022.
[3] FDA and EMA drug approval databases.
[4] MarketResearch.com. Cardiovascular Drugs Market Reports, 2022.
More… ↓
