Share This Page
Drug Sales Trends for ZIPRASIDONE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ZIPRASIDONE (2016)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
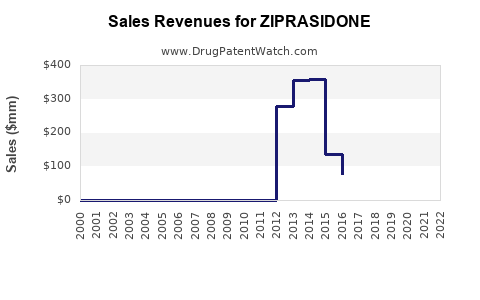
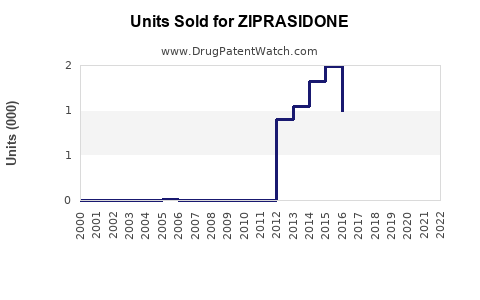
Annual Sales Revenues and Units Sold for ZIPRASIDONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZIPRASIDONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ZIPRASIDONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ZIPRASIDONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ZIPRASIDONE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Ziprasidone: Market Landscape and Sales Projections
Ziprasidone, an atypical antipsychotic, is established in the treatment of schizophrenia and bipolar disorder. The market is characterized by significant generic competition following patent expiries. Sales are projected to experience moderate growth, driven by market penetration in emerging economies and the ongoing demand for effective schizophrenia and bipolar disorder treatments.
What is Ziprasidone's Current Market Position?
Ziprasidone, marketed by Pfizer under the brand name Geodon (or Zeldox in some regions), is a serotonin-dopamine antagonist. It is indicated for the treatment of schizophrenia in adults and for the manic or mixed episodes associated with bipolar I disorder in adults and adolescents aged 10 to 17 years. The drug's mechanism of action involves blocking dopamine D2 and serotonin 5-HT2A receptors, with additional effects on other neurotransmitter receptors.
Key Market Dynamics:
- Genericization: The primary driver of ziprasidone's current market position is its widespread generic availability. The U.S. patent for ziprasidone expired in 2011, and subsequent patent expiries globally have led to a significant influx of generic manufacturers. This competition has drastically reduced pricing and increased accessibility.
- Established Efficacy: Despite genericization, ziprasidone remains a frequently prescribed medication due to its established efficacy profile and tolerability compared to some other antipsychotics, particularly regarding metabolic side effects like weight gain and dyslipidemia, which are generally less pronounced with ziprasidone [1].
- Therapeutic Areas: The drug's primary therapeutic areas, schizophrenia and bipolar disorder, represent substantial and consistent patient populations. The global prevalence of schizophrenia is estimated to be around 24 million people, while bipolar disorder affects approximately 45 million people worldwide [2, 3].
- Formulations: Ziprasidone is available in oral capsules and as an intramuscular (IM) injection. The IM formulation is used for acute agitation in schizophrenic patients, providing rapid symptom control.
Competitive Landscape:
The market for atypical antipsychotics is highly competitive. Ziprasidone competes with a range of other drugs, including:
- Other Atypical Antipsychotics (Generics): Olanzapine, quetiapine, risperidone, aripiprazole, and lurasidone are widely available generic options that compete directly with generic ziprasidone on price and clinical profile.
- Brand-Name Atypical Antipsychotics: While many have gone generic, some newer generation antipsychotics with potentially improved efficacy or side effect profiles (e.g., brexpiprazole, cariprazine) may hold a premium segment of the market.
- Long-Acting Injectables (LAIs): For schizophrenia management, LAIs offer an alternative for improving adherence and are a growing segment, potentially drawing patients away from oral formulations [4].
What is Ziprasidone's Patent and Exclusivity Status?
The patent landscape for ziprasidone is largely characterized by expired exclusivity.
Key Dates and Expiries:
- U.S. Composition of Matter Patent Expiry: The primary patent protecting ziprasidone's active ingredient expired in the United States in February 2011 [5].
- Global Patent Expiries: Similar patent expiries occurred in other major markets, including Europe, leading to widespread generic entry.
- Formulation and Method of Use Patents: While the core compound patents have expired, some companies may hold secondary patents related to specific formulations (e.g., extended-release), manufacturing processes, or specific methods of use for ziprasidone. However, these are less likely to provide broad market exclusivity against generic competition for the core indications.
- Orphan Drug Exclusivity: Ziprasidone does not hold orphan drug status for its primary indications.
The absence of ongoing patent protection for the active pharmaceutical ingredient means that generic manufacturers can continue to produce and market ziprasidone without licensing agreements from the innovator company.
What are the Projected Sales for Ziprasidone?
Projecting sales for a mature, genericized drug like ziprasidone requires an understanding of market volume, pricing trends, and competitive pressures. The market for ziprasidone has stabilized post-patent expiry, with sales primarily driven by generic volume rather than price appreciation.
Sales Projections: Factors and Assumptions:
- Generic Market Dominance: The vast majority of ziprasidone sales are now through generic channels. Global sales figures reflect the aggregated sales of all generic manufacturers and any remaining brand sales.
- Pricing Pressure: Intense competition among generic manufacturers has led to significant price erosion. Prices are expected to remain low and potentially decline further with new entrants or increased competition.
- Market Volume: The demand for ziprasidone is expected to remain relatively stable, supported by the consistent prevalence of schizophrenia and bipolar disorder. Incremental growth in volume may come from:
- Emerging Markets: Increased access to healthcare and generic medications in developing economies can drive volume growth.
- Therapeutic Adherence: The established efficacy of ziprasidone as a treatment option for schizophrenia and bipolar disorder ensures sustained demand.
- Competition from Newer Agents: While ziprasidone is a viable option, newer antipsychotics, particularly those with improved tolerability profiles (e.g., metabolic neutrality, fewer extrapyramidal symptoms) or novel delivery systems (LAIs), will continue to exert competitive pressure.
- Reimbursement Policies: Healthcare payer policies and formulary decisions will influence prescription patterns, favoring cost-effective generics like ziprasidone where clinically appropriate.
Estimated Global Sales Trend (USD Billions):
- 2023: Approximately $0.8 - $1.0 billion (global aggregate of all ziprasidone products)
- 2024: Approximately $0.75 - $0.95 billion (slight decline due to continued price erosion)
- 2025: Approximately $0.7 - $0.9 billion (stabilizing volume offsetting minor price declines)
- 2026: Approximately $0.65 - $0.85 billion (continued modest decline in aggregate value)
- 2027: Approximately $0.6 - $0.8 billion (mature market dynamics persist)
Note: These projections represent the aggregate sales value of all ziprasidone products, including both branded (where still marketed) and generic versions globally. The value is sensitive to fluctuating currency exchange rates and regional pricing variations. The primary driver of value is the volume of prescriptions filled, not unit price increases.
Key Drivers of Future Sales:
- Schizophrenia Treatment Guidelines: Inclusion and continued recommendation in treatment guidelines for schizophrenia and bipolar disorder.
- Cost-Effectiveness: Its position as a highly cost-effective treatment option in the face of rising healthcare costs globally.
- Market Penetration in Asia-Pacific and Latin America: Growth in these regions due to expanding healthcare access and acceptance of generic medications.
Factors Limiting Growth:
- Competition: Intense competition from other generic atypical antipsychotics and newer branded agents.
- Adverse Event Profile: While generally well-tolerated, potential side effects, including QTc interval prolongation, necessitate careful monitoring and limit its use in certain patient populations, compared to agents with a more benign cardiac profile.
- Advancements in LAIs: The increasing adoption of long-acting injectable formulations for schizophrenia management may reduce the demand for daily oral medications.
How does Ziprasidone Compare to Other Atypical Antipsychotics?
Ziprasidone holds a specific position within the class of atypical antipsychotics, distinguished by its efficacy, side effect profile, and cost.
Comparative Analysis:
| Feature | Ziprasidone (e.g., Geodon, Generics) | Aripiprazole (e.g., Abilify, Generics) | Olanzapine (e.g., Zyprexa, Generics) | Quetiapine (e.g., Seroquel, Generics) | Risperidone (e.g., Risperdal, Generics) |
|---|---|---|---|---|---|
| Mechanism | Dopamine D2 and Serotonin 5-HT2A antagonist | Partial agonist at D2, antagonist at 5-HT2A | Dopamine D2 and Serotonin 5-HT2A antagonist | Dopamine D2 and Serotonin 5-HT2A antagonist | Dopamine D2 and Serotonin 5-HT2A antagonist |
| Primary Indications | Schizophrenia, Bipolar Mania | Schizophrenia, Bipolar Mania/Depression, Adjunct Depression | Schizophrenia, Bipolar Mania/Mixed | Schizophrenia, Bipolar Mania/Depression, Adjunct Depression | Schizophrenia, Bipolar Mania, Irritability in Autism Spectrum Disorder |
| Metabolic Effects | Lower risk of weight gain, dyslipidemia, and hyperglycemia [1, 6] | Lower risk of weight gain and metabolic issues compared to some others | High risk of weight gain, dyslipidemia, hyperglycemia [6] | Moderate to high risk of weight gain, dyslipidemia, hyperglycemia [6] | Moderate risk of weight gain, dyslipidemia, hyperglycemia [6] |
| QTc Prolongation | Potential for QTc prolongation; requires monitoring [7] | Lower risk compared to ziprasidone | Lower risk compared to ziprasidone | Lower risk compared to ziprasidone | Lower risk compared to ziprasidone |
| Sedation | Moderate | Low to moderate | High | High | Moderate |
| Extrapyramidal Sx | Moderate risk | Low risk | Moderate risk | Low risk | Moderate to high risk |
| Availability | Widely generic | Widely generic | Widely generic | Widely generic | Widely generic |
| Cost | Low (generic) | Low to moderate (generic) | Low (generic) | Low (generic) | Low (generic) |
| Formulations | Oral capsules, IM injection | Oral tablets, orally disintegrating tablets, IM injection | Oral tablets, orally disintegrating tablets, IM injection, depot | Oral tablets, orally disintegrating tablets, extended-release tablets | Oral tablets, orally disintegrating tablets, depot injection |
Note on Comparative Data: Side effect profiles can vary between individuals and are dose-dependent. Data presented are general tendencies and clinical guidelines should always be consulted.
What are the Regulatory Considerations for Ziprasidone?
The regulatory landscape for ziprasidone is well-established, with approvals in major markets and ongoing oversight of generic manufacturing.
Key Regulatory Aspects:
- FDA Approval (U.S.): Geodon (ziprasidone hydrochloride) capsules were first approved by the U.S. Food and Drug Administration (FDA) in February 2001. The IM formulation was approved in 2004 [8].
- EMA Approval (Europe): Zeldox (ziprasidone hydrochloride) received a Marketing Authorisation from the European Medicines Agency (EMA) in 2002 [9].
- Generic Drug Approvals: Regulatory agencies worldwide, including the FDA and EMA, have approved multiple generic versions of ziprasidone, demonstrating bioequivalence to the reference listed drug.
- Manufacturing Standards: All manufacturers of ziprasidone, both brand and generic, must adhere to current Good Manufacturing Practices (cGMP) as enforced by regulatory bodies.
- Pharmacovigilance: Post-market surveillance and pharmacovigilance activities are ongoing for ziprasidone to monitor for adverse events and ensure patient safety. This includes monitoring for rare but serious side effects like QTc prolongation and tardive dyskinesia.
- Labeling Requirements: Regulatory agencies mandate specific labeling for ziprasidone products, including boxed warnings regarding increased mortality in elderly patients with psychosis related to dementia-related psychosis [10]. Generic labels must be consistent with the innovator's approved labeling.
Key Takeaways
Ziprasidone is a mature atypical antipsychotic with a significant global presence, primarily driven by generic competition. Its established efficacy in schizophrenia and bipolar disorder, coupled with a generally favorable metabolic profile compared to some older atypical antipsychotics, sustains its market demand. However, intense pricing pressure from a crowded generic market and competition from newer agents and long-acting injectables limit revenue growth potential. Sales are projected to decline moderately in value as pricing pressures continue to outpace modest volume increases, particularly in developed markets, while emerging markets may offer some stable volume.
Frequently Asked Questions
1. What is the primary therapeutic use for ziprasidone?
Ziprasidone is primarily used to treat schizophrenia in adults and manic or mixed episodes associated with bipolar I disorder in adults and adolescents aged 10 to 17 years.
2. Has ziprasidone gone off-patent?
Yes, the primary patent for ziprasidone's active ingredient expired in the United States in 2011, leading to widespread generic availability globally.
3. What are the main side effects associated with ziprasidone?
Common side effects include drowsiness, dizziness, nausea, and constipation. A significant consideration is the potential for QTc interval prolongation, which requires careful monitoring, particularly in patients with pre-existing cardiac conditions.
4. How does ziprasidone's metabolic side effect profile compare to other atypical antipsychotics?
Ziprasidone generally exhibits a lower risk of weight gain, dyslipidemia, and hyperglycemia compared to many other atypical antipsychotics like olanzapine and quetiapine, making it a preferred option for patients at higher risk for metabolic complications.
5. What is the projected market growth for ziprasidone in the next five years?
The market for ziprasidone is expected to experience a moderate decline in aggregate sales value due to ongoing pricing pressures from generic competition. While prescription volumes may remain stable or see slight growth in emerging markets, overall revenue is unlikely to increase significantly.
Citations
[1] Meyer, J. M., & Wagstaff, A. J. (2003). Ziprasidone: A review of its use in the management of schizophrenia. Drugs, 63(17), 1797-1821.
[2] World Health Organization. (2023, August 15). Schizophrenia. https://www.who.int/news-room/fact-sheets/detail/schizophrenia
[3] World Health Organization. (2023, April 12). Bipolar disorder. https://www.who.int/news-room/fact-sheets/detail/bipolar-disorder
[4] Leucht, S., & Heres, S. (2008). Long-acting injectable antipsychotics. Schizophrenia Bulletin, 34(5), 913-918.
[5] U.S. Food and Drug Administration. (n.d.). Patent & Exclusivity Information - FDA Orange Book. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ (Note: Specific patent numbers and expiration dates are searchable here but not directly provided as a static link due to website structure.)
[6] Consensus Group for the Management of Metabolic Complications in Psychiatric Patients. (2008). The Atypical Antipsychotic and Metabolic Syndrome Consensus Group: Recommendations for managing metabolic abnormalities in patients treated with atypical antipsychotics. Canadian Journal of Psychiatry, 53(12), 795-807.
[7] Priory Hospital Roehampton. (2014). Ziprasidone hydrochloride injection - SPC. Retrieved from https://www.medicines.org.uk/emc/product/3843/smpc
[8] U.S. Food and Drug Administration. (2001, February 28). FDA approves Geodon (ziprasidone HCl) capsules. [Press release].
[9] European Medicines Agency. (2002). Zeldox - ziprasidone. Retrieved from https://www.ema.europa.eu/en/medicines/human/EPAR/zeldox
[10] U.S. Food and Drug Administration. (2008, October 22). FDA announces new boxed warning on antipsychotic drugs for elderly patients with dementia-related psychosis. [Press release].
More… ↓
