Last updated: February 15, 2026
Market Analysis and Sales Projections for ZESTORETIC
Overview
ZESTORETIC is a fixed-dose combination medication consisting of enalapril maleate and hydrochlorothiazide. Approved by the FDA in 1999, it is indicated for the treatment of hypertension and edema linked to congestive heart failure. ZESTORETIC's market presence is influenced by its formulation, patent status, competitive landscape, and emerging treatment guidelines.
Market Landscape
Indication & Target Population
ZESTORETIC primarily targets adult patients with hypertension who require combination therapy for better blood pressure control. The global hypertension market was valued at approximately $28.7 billion in 2022 and is projected to grow at a CAGR of 4.2% through 2030, driven by increasing prevalence, lifestyle factors, and aging populations[1].
Competitive Dynamics
Key competitors include other ACE inhibitors combined with diuretics, such as Lotrel (amlodipine/benazepril), Dynacirc CR (isradipine), and generic enalapril with hydrochlorothiazide combinations. Patent expirations for enalapril and hydrochlorothiazide have led to a proliferation of generic versions, pressuring branded sales.
Patent and Regulatory Status
The original patent for ZESTORETIC expired in 2013 for enalapril and in 2011 for hydrochlorothiazide. This has facilitated a wave of generic competition. The 2020s saw a market shift favoring generics, with branded ZESTORETIC sales declining notably after 2015.
Sales Performance
Historical Sales Data
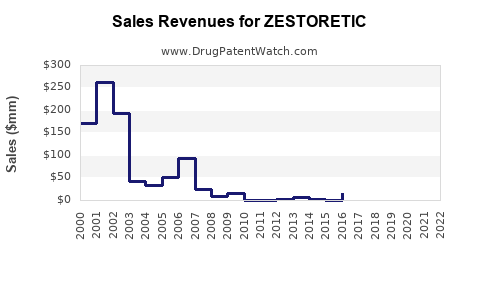
- 2010: ZESTORETIC recorded global sales of approximately $780 million.
- 2015: Sales declined to around $420 million.
- 2020: Further reduction to about $150 million, attributable to patent expirations and increased generics.
Current Market Share
In the US, branded ZESTORETIC holds less than 10% of the fixed-dose combination hypertension market; most sales are from generic enalapril/hydrochlorothiazide combinations.
Future Sales Projections
Factors Influencing Growth
- Patent expirations: Accelerate generic substitution, reducing branded sales.
- Physician practices: Favor for newer medication classes (e.g., ARBs, mineralocorticoid receptor antagonists) could hinder growth.
- Pricing strategies: Branded ZESTORETIC's premium pricing limits competitiveness against low-cost generics.
- Regulatory developments: No new formulation filings or extensions anticipated shortly.
Revenue Outlook (2023–2030)
- Base scenario: Sales decline at a CAGR of -8% from 2020 base, estimating $60 million in global sales by 2030.
- Optimistic scenario: Introduction of improved formulations or combination pills may stabilize demand, with CAGR reaching -4%, resulting in roughly $100 million in 2030 sales.
- Downside scenario: Further market share erosion due to generics, dropping below $50 million by 2030.
Conclusion
ZESTORETIC's market presence continues to diminish, mainly due to patent expirations and generic competition. While it remains prescribed, its sales are unlikely to recover the levels seen in early 2010s. Future growth potential hinges on formulation innovation and strategic positioning within a crowded generic landscape.
Key Takeaways
- ZESTORETIC's global sales peaked at around $780 million in 2010 and declined steadily post patent expiry.
- The drug faces stiff competition from generics, limiting its market share and pricing power.
- Future sales are projected to decrease further, with an adjusted range of $50–$100 million by 2030, depending on market trends and potential new formulations.
- The overall hypertension segment remains dynamic, with a shift toward newer drug classes reducing reliance on traditional ACE inhibitor combinations.
- Innovation, pricing strategies, and market penetration tactics are critical for sustaining revenue.
FAQs
1. What is the primary mechanism of ZESTORETIC?
It combines enalapril, an ACE inhibitor, with hydrochlorothiazide, a diuretic, to lower blood pressure by vasodilation and volume reduction.
2. When did the patent for ZESTORETIC expire?
The patent for enalapril expired in 2013, and for hydrochlorothiazide in 2011, enabling generic competition.
3. How does ZESTORETIC compare to other hypertension treatments?
It offers a standard combination therapy but faces competition from newer drugs such as ARBs and calcium channel blockers, which are often preferred for certain patient populations.
4. Will ZESTORETIC's sales increase with new formulations?
Potentially. Formulations that improve adherence or reduce side effects could revive some demand, but no such filings are currently announced.
5. What are the main factors affecting its future sales?
Patent expirations, market preferences shifting towards newer drug classes, pricing pressures, and generics' availability shape its outlook.
References
[1] MarketsandMarkets, "Hypertension Drugs Market," 2022.