Last updated: February 19, 2026
XARELTO (rivaroxaban) is a direct oral anticoagulant (DOAC) manufactured by Bayer AG and Johnson & Johnson. Its primary indication is to reduce the risk of stroke and systemic embolism in patients with non-valvular atrial fibrillation (NVAF) and to treat and prevent deep vein thrombosis (DVT) and pulmonary embolism (PE).
What is the current patent status of XARELTO?
The foundational patent for XARELTO, U.S. Patent No. 7,348,332, covering rivaroxaban, expired in July 2023 in the United States. This patent was granted on March 25, 2008, with an expiration date of July 26, 2026, accounting for patent term extension (PTE). Secondary patents related to manufacturing processes, formulations, and specific uses may still be in effect. For example, U.S. Patent No. 9,750,749, concerning the prevention of recurrent venous thromboembolism, is set to expire in December 2033.
What are the key market segments for XARELTO?
XARELTO competes in the anticoagulant market, primarily with other DOACs such as Eliquis (apixaban) and Pradaxa (dabigatran etexilate), as well as warfarin, a vitamin K antagonist.
- Non-Valvular Atrial Fibrillation (NVAF): This is the largest indication for XARELTO. It is prescribed to prevent stroke and systemic embolism in patients with NVAF.
- Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE): XARELTO is used for the treatment of DVT and PE and for the extended treatment of DVT and PE to reduce the risk of recurrence.
- Coronary Artery Disease (CAD) and Peripheral Artery Disease (PAD): XARELTO, in combination with aspirin, is indicated for the prevention of major cardiovascular events in patients with chronic CAD or PAD.
What are XARELTO's sales performance and market share?
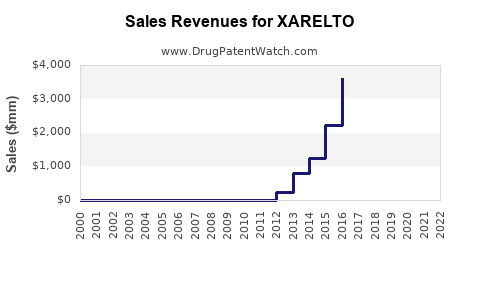
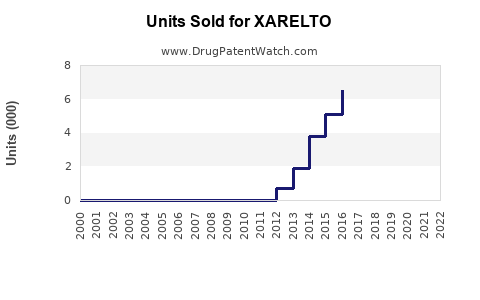
XARELTO has been a significant revenue driver for its manufacturers. In 2022, XARELTO generated approximately €4.7 billion (US$5.0 billion) in net sales globally, with Bayer reporting €2.7 billion and Janssen Pharmaceuticals (Johnson & Johnson) reporting US$2.2 billion [1, 2]. This represents a slight decrease from 2021 sales of approximately €4.9 billion (US$5.2 billion).
Global Net Sales (in billions USD):
| Year |
Bayer (Euro converted to USD) |
Johnson & Johnson (USD) |
Total (Approximate) |
| 2022 |
2.9 |
2.2 |
5.1 |
| 2021 |
3.0 |
2.2 |
5.2 |
| 2020 |
3.1 |
2.2 |
5.3 |
Note: Euro to USD conversions are based on average annual exchange rates.
XARELTO holds a substantial share of the DOAC market. While exact market share figures fluctuate, it is consistently ranked among the top DOACs, often vying with Eliquis for market leadership in certain regions or indications. Competition from Eliquis (apixaban) has been intense, with Eliquis demonstrating strong growth and market penetration, particularly in the US.
What are the projected future sales and market trends for XARELTO?
The expiration of its core patent in the US in July 2023 signals a shift in XARELTO's market trajectory. The market is now open to generic competition, which is expected to exert downward pressure on XARELTO's sales volume and average selling price.
- Generic Entry: The first generic versions of rivaroxaban have entered the US market following the patent expiration. This will lead to increased price competition and a reduction in market share for the branded product.
- Continued Demand in Key Indications: Despite generic competition, XARELTO is expected to maintain a significant presence in the NVAF and DVT/PE treatment segments due to its established efficacy and safety profile. Physician and patient familiarity with the drug will support continued prescription volumes, albeit at lower price points.
- Competition from Eliquis: Eliquis has shown robust growth and captured a considerable market share. It is expected to remain a dominant player in the DOAC market and will continue to be XARELTO's primary competitor.
- Combination Therapies: The use of XARELTO in combination with aspirin for patients with CAD/PAD represents a segment where it can continue to differentiate. However, this segment also faces competition from other antiplatelet and anticoagulant strategies.
Projected Sales Decline (Post-Patent Expiry): Analysts project a significant decline in XARELTO's sales in the US post-July 2023, with a more gradual erosion in ex-US markets where patent protections may extend further. For example, in Europe, key patents are expected to expire later, providing a longer period of market exclusivity.
What is the competitive landscape for XARELTO?
The anticoagulant market is highly competitive. XARELTO's main competitors include:
- Eliquis (apixaban): Developed by Bristol Myers Squibb and Pfizer. Eliquis has demonstrated superior efficacy in some studies and has gained significant market share, particularly in the US.
- Pradaxa (dabigatran etexilate): Developed by Boehringer Ingelheim. Pradaxa was one of the first DOACs to market and remains a significant competitor, though it has faced some safety concerns in its early years.
- Warfarin (Coumadin, Jantoven): A long-standing anticoagulant. While effective, warfarin requires frequent monitoring and dietary restrictions, making DOACs like XARELTO more convenient for many patients.
- Other Anticoagulants: Including heparin and low molecular weight heparins (LMWHs), primarily used for acute treatment or in specific patient populations.
Key Competitive Differentiators:
- Efficacy and Safety Profile: All DOACs have demonstrated non-inferior or superior efficacy compared to warfarin for stroke prevention in NVAF, with a lower risk of intracranial bleeding. Head-to-head studies and real-world data continue to shape prescribing patterns.
- Dosing Regimen: XARELTO's once-daily dosing is a key convenience factor for patients and healthcare providers. Eliquis offers twice-daily dosing.
- Indications: The range of approved indications for each drug influences its market penetration.
- Cost and Reimbursement: Generic entry for XARELTO will significantly alter its cost-effectiveness and reimbursement landscape, impacting its competitiveness.
What are the regulatory considerations and market access challenges?
Regulatory approval for XARELTO across various indications has been secured in major markets, including the United States (FDA) and Europe (EMA). Market access is influenced by:
- Health Technology Assessments (HTAs): Bodies like the National Institute for Health and Care Excellence (NICE) in the UK and the IQWiG in Germany evaluate the cost-effectiveness of drugs, influencing their reimbursement and formulary placement. XARELTO has undergone numerous such assessments, impacting its price negotiations and accessibility.
- Payer Policies: Insurance companies and national health systems establish formularies and utilization management policies that can restrict access or favor competing products based on cost and clinical evidence.
- Generic Competition: Post-patent expiry, payers are likely to favor generic rivaroxaban due to lower costs, further challenging branded XARELTO's market access.
- Physician Prescribing Preferences: Clinical trial data, real-world evidence, and physician education play a crucial role in shaping prescribing habits.
What are the future R&D and lifecycle management strategies for rivaroxaban?
Despite the patent expiry of its core compound, lifecycle management for rivaroxaban may involve strategies to extend its commercial life or leverage its established profile.
- New Formulations or Delivery Methods: Development of novel formulations could potentially lead to new patents, though significant innovation in this mature drug class is less probable.
- Combination Therapies: As seen with the CAD/PAD indication, further exploration of rivaroxaban in combination with other agents for different therapeutic areas could open new avenues.
- Life Cycle Management of Secondary Patents: Manufacturers will continue to defend and leverage any remaining patents related to manufacturing processes, polymorphs, or specific therapeutic uses.
- Focus on Branded Generics or Authorized Generics: In some markets, manufacturers may introduce their own generic versions or authorized generics to capture a portion of the generic market.
Key Takeaways
XARELTO's foundational patent in the United States expired in July 2023, opening the market to generic competition. This event is expected to significantly reduce the branded drug's sales in the US and put downward pressure on global revenue. Despite this, XARELTO remains a major player in the anticoagulant market, particularly for NVAF and DVT/PE treatment. Intense competition from Eliquis and other DOACs, coupled with the advent of generic rivaroxaban, will define its future market position. Lifecycle management strategies and the continued relevance of secondary patents will be crucial for its manufacturers.
FAQs
-
When did the primary patent for XARELTO expire in the United States?
The foundational patent for rivaroxaban (U.S. Patent No. 7,348,332) expired in July 2023.
-
What are the main competitors to XARELTO in the anticoagulant market?
Key competitors include Eliquis (apixaban), Pradaxa (dabigatran etexilate), and warfarin.
-
What are the primary medical conditions for which XARELTO is prescribed?
XARELTO is prescribed for reducing the risk of stroke and systemic embolism in patients with non-valvular atrial fibrillation (NVAF) and for the treatment and prevention of deep vein thrombosis (DVT) and pulmonary embolism (PE).
-
How are generic versions of XARELTO expected to impact its sales?
The introduction of generic rivaroxaban is anticipated to lead to a significant decrease in branded XARELTO sales due to price competition and market share erosion.
-
Will XARELTO continue to be prescribed after its patent expiration?
Yes, XARELTO is expected to remain a prescribed medication. Its established efficacy, safety profile, and physician familiarity will support continued use, though at a lower price point and with reduced market share compared to the pre-patent expiry period.
Citations
[1] Bayer AG. (2023). Bayer AG Annual Report 2022. Retrieved from https://www.bayer.com/en/investors/annual-report
[2] Johnson & Johnson. (2023). Johnson & Johnson 2022 Annual Report. Retrieved from https://www.jnj.com/investors/annual-reports