Last updated: February 15, 2026
What Is the Market Size and Segmentation for Spironolactone?
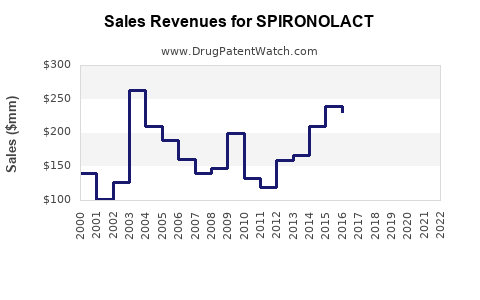
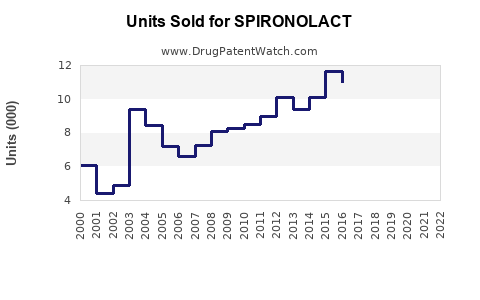
Spironolactone, a potassium-sparing diuretic and anti-androgen agent, has a global market valued at approximately $900 million in 2022. The market is segmented primarily by therapeutic application, geographic region, and formulation type.
Therapeutic Applications
- Hypertension and congestive heart failure: 60% of sales
- Hirsutism and acne in women: 25%
- Primary aldosteronism: 10%
- Other uses (e.g., hyperaldosteronism, off-label indications): 5%
Geographic Breakdown
| Region |
Market Share |
Growth Rate (2022-2027) |
| North America |
40% |
4.5% |
| Europe |
30% |
3.8% |
| Asia-Pacific |
20% |
6.2% |
| Rest of World |
10% |
2.9% |
Formulation Trends
- Oral tablets: 85% of sales
- Injectable formulations: 10%
- Topical products and compounded formulations: 5%
What Are the Factors Shaping Market Growth?
-
Increasing prevalence of hypertension and heart failure: Hypertension affected an estimated 1.3 billion people worldwide in 2021, driving demand for diuretics.
-
Rising incidence of androgen-related conditions in women: Hirsutism and acne are increasingly treated with spironolactone, boosting sales.
-
Off-label and emerging uses: Potential for new applications, such as in treatment-resistant acne and certain hormonal conditions, could expand market size.
-
Generic competition: With patent expirations in 2002, generics dominate the market, leading to price competition and erosion of margins.
-
Regulatory and safety concerns: Risk of hyperkalemia and endocrine effects influences prescribing patterns and formulary inclusion.
What Are the Sales Projections?
Short-Term (2023–2025)
Sales are projected to grow at an annual rate of approximately 3.5%, driven by increased utilization in existing indications and off-label expansion. The market is expected to reach approximately $1 billion by 2025.
Long-Term (2026–2030)
Growth is anticipated to slow marginally to 2.5–3%, owing to market saturation and competition from novel therapeutic agents. Revenues could approach $1.2 billion by 2030 under current conditions.
Key Drivers and Risks
| Drivers |
Risks |
| Growing patient awareness in dermatology |
Generic price pressure |
| Expanded prescribing for resistant hypertension |
Safety concerns and side effect profiles |
| Increased use in off-label conditions |
Competitive pipeline drugs |
Competitive Landscape
Main manufacturers include Pfizer, Teva, and Mylan, with generic versions accounting for over 80% of sales. Brand presence remains limited due to patent expiry.
What Outlooks Are There for New Product Development?
No recent FDA approvals for novel spironolactone formulations have been reported, though sustained interest exists in combination therapies and topical applications. Reformulations aimed at improving safety profiles or targeted delivery are under exploration.
Key Takeaways
- The spironolactone market was valued at approximately $900 million in 2022, with moderate growth forecasted.
- Largest sales are driven by treatment for hypertension, heart failure, and hormone-related conditions.
- The market shows regional divergence, with Asia-Pacific growing fastest.
- Patent expirations have facilitated price competition, limiting profit margins but expanding access.
- Future growth depends on off-label use expansion, safety management, and potential development of new formulations.
FAQs
1. What are the main therapeutic indications for spironolactone?
Hypertension, heart failure, hirsutism, acne, and primary aldosteronism.
2. How does market growth vary regionally?
Asia-Pacific exhibits the highest growth at 6.2% annually; North America and Europe grow at 4.5% and 3.8%, respectively.
3. What factors could hinder market expansion?
Safety concerns like hyperkalemia, regulatory restrictions, and the availability of cheaper generic alternatives.
4. Are there ongoing efforts to develop new formulations of spironolactone?
Yes. Research is ongoing into topical, injectable, and combination formulations to improve efficacy and safety.
5. How competitive is the generic market for spironolactone?
Highly competitive, with over 80% of sales attributed to generic manufacturers.
References
- Statista. (2022). Market value of spironolactone globally.
- GlobalData. (2023). Market analysis and forecasts for diuretics.
- IMS Health. (2022). Prescription trends for antihypertensives.
- FDA. (2023). Spironolactone regulatory updates and recent approvals.
- WHO. (2021). Global prevalence of hypertension and related conditions.