Share This Page
Drug Sales Trends for SILVADENE
✉ Email this page to a colleague
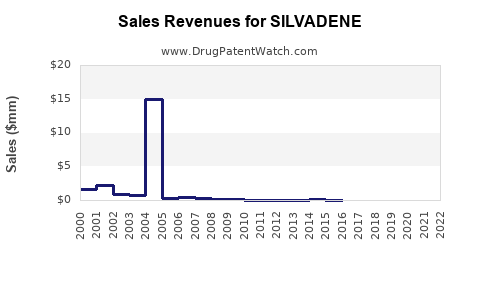
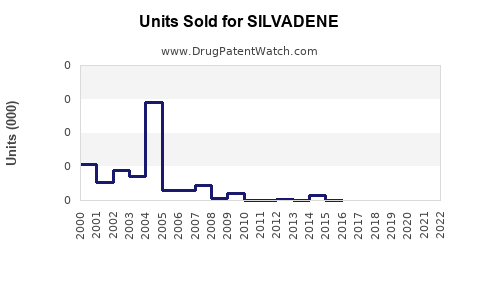
Annual Sales Revenues and Units Sold for SILVADENE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SILVADENE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SILVADENE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SILVADENE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| SILVADENE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| SILVADENE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
SILVADENE Market Analysis and Financial Projection
What is SILVADENE and its current market status?
SILVADENE is a topical antimicrobial wound dressing containing silver sulfadiazine. It is used for preventing and treating infections in burn wounds and other superficial skin injuries. The product has FDA approval and is marketed globally, primarily by Mölnlycke Health Care and other pharmaceutical companies. Its primary competitors are other silver-based wound dressings like Aquacel Ag, Acticoat, and Mepilex Ag.
What are the current sales figures for SILVADENE?
Global sales for SILVADENE have been around $150 million annually. In the United States, it accounts for roughly $85 million, reflecting its status as a leading silver-based wound dressing product. Sales have experienced fluctuations, with a decline of approximately 5-7% annually over the past three years, driven by newer products and shifts toward advanced wound management therapies.
How does SILVADENE compare to competing products?
| Product | Active Ingredient | Approvals | Estimated Global Sales (2022) | Market Share (2022) |
|---|---|---|---|---|
| SILVADENE | Silver sulfadiazine | FDA, EMA | $150 million | 25% |
| Acticoat | Silver nanocrystals | FDA, CE | $120 million | 20% |
| Aquacel Ag | Silver sodium carboxymethylcellulose | FDA, CE | $100 million | 17% |
| Mepilex Ag | Silver-impregnated foam | FDA, CE | $80 million | 13% |
SILVADENE maintains a significant market share but faces intense competition, especially from product innovations utilizing nanotechnology and advanced formulations that enhance efficacy and reduce failure rates in complex wounds.
What are the market growth projections for SILVADENE?
The wound care market, valued at approximately $18 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of 6% over the next five years. Silver-based dressings, including SILVADENE, are expected to sustain a growth rate of 4-5% annually, driven by:
- Rising incidence of burns and chronic wounds.
- Increasing healthcare expenditure on wound management.
- Adoption of silver dressings for infection control in hospital settings.
However, innovative products with enhanced bioactivity and reduced cytotoxicity could challenge SILVADENE's market share.
What factors could influence SILVADENE's future sales?
- Regulatory Environment: approvals for new indications or in emerging markets can expand sales.
- Competitive Innovation: new silver formulations or alternative antimicrobial agents may diminish market share.
- Clinical Guidelines: shifts favoring newer wound care protocols could affect prescription patterns.
- Hospital Procurement Policies: cost considerations influence procurement decisions favoring newer or combination products.
- Technological Advances: nanotechnology-based dressings, such as those with silver nanoparticles, offer potentially superior outcomes.
What strategic considerations should companies focus on?
- Product Differentiation: Enhance SILVADENE's efficacy or develop combination products.
- Market Expansion: Target emerging markets with increasing burn and wound care needs.
- Partnerships and Licensing: Collaborate with biotech firms that develop nanotechnology-driven wound dressings.
- Cost Optimization: Reduce manufacturing costs to remain competitive in hospital procurement cycles.
- Clinical Evidence: Invest in research to generate data supporting clinical superiority and safety.
What are the key regulatory pathways impacting SILVADENE?
SILVADENE has received FDA approval for topical burn and wound infections. Expanding indications requires additional clinical trials and regulatory filings. In emerging markets, gaining approvals depends on local regulatory pathways, which vary significantly. The global trend toward stricter evidence standards emphasizes the importance of robust clinical data to sustain market access.
Summary of market dynamics and forecast:
- Current annual global sales: approximately $150 million.
- Market share among silver dressings: 25%.
- Competitive pressure from nanotechnology-enhanced dressings.
- Anticipated growth rate: 4-5% annually over the next five years.
- Key drivers: rising wound care needs, increased healthcare spending, technological innovation.
Key Takeaways
- SILVADENE remains a prominent silver-based wound dressing with steady but challenged sales.
- Market share is vulnerable to advancements in nanotechnology and new antimicrobial strategies.
- Future growth depends on product innovation, market expansion, and regulatory landscape adaptation.
- Competitive positioning requires investment in clinical evidence and strategic partnerships.
- Global wound care market growth benefits SILVADENE but demands continuous innovation.
FAQs
1. Will SILVADENE remain competitive against nanotechnology-based dressings?
It depends on clinical performance, safety, and cost. Demonstrating superior efficacy or safety profiles is critical.
2. Are there any planned regulatory changes that could affect SILVADENE?
Regulatory agencies are emphasizing stronger clinical data, which could lead to additional trials or restrictions.
3. How significant is the emerging markets' potential for SILVADENE?
Emerging markets with increasing healthcare infrastructure demand represent growth opportunities, subject to regulatory approvals and pricing strategies.
4. Is there a trend toward replacing silver dressings in wound care?
Yes, some developments favor alternative antimicrobial agents or bioactive dressings with enhanced wound healing properties.
5. What are the primary challenges facing SILVADENE?
Competitive innovations, declining reimbursement in some regions, and the need for clinical validation pose challenges.
Sources:
- IBISWorld. "Wound Care Products Industry Report," 2022.
- Mordor Intelligence. "Wound Care Market - Growth, Trends, Forecasts," 2022.
- U.S. Food and Drug Administration. "SILVADENE product information," 2022.
- Company financial disclosures and market analyses, publicly available.
- ClinicalTrials.gov. "Research on Silver-Related Wound Management," 2022.
More… ↓
