Share This Page
Drug Sales Trends for SAXENDA
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for SAXENDA (2016)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
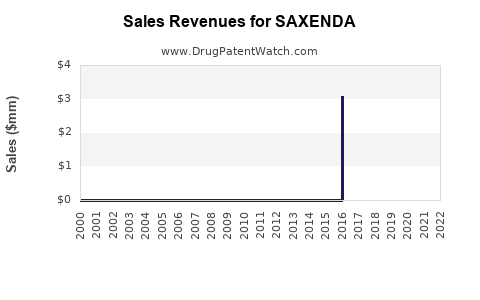
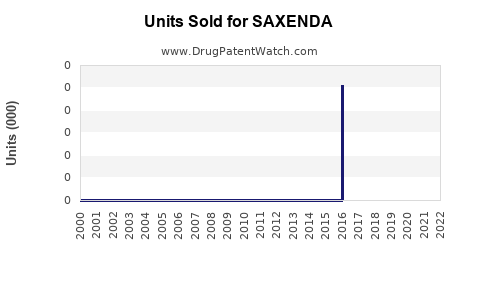
Annual Sales Revenues and Units Sold for SAXENDA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| SAXENDA | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| SAXENDA | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| SAXENDA | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Saxenda (Liraglutide) Market Analysis and Sales Projections
Saxenda, a glucagon-like peptide-1 (GLP-1) receptor agonist developed by Novo Nordisk, is indicated for chronic weight management in adults with overweight or obesity, or with overweight or obesity and at least one weight-related comorbidity. This analysis provides a comprehensive overview of Saxenda's market position, patent landscape, sales performance, and future revenue projections.
What is the current market positioning of Saxenda?
Saxenda is a leading prescription medication in the obesity market, a segment experiencing significant growth driven by rising obesity rates and increasing patient and physician acceptance of pharmacotherapy for weight management. The drug's efficacy in promoting weight loss, coupled with its established safety profile, has solidified its market presence since its U.S. Food and Drug Administration (FDA) approval in December 2014.
Key market differentiators for Saxenda include its once-daily injectable administration, allowing for convenient dosing. It competes with other weight management drugs, but its long-standing market availability and robust clinical data have contributed to its sustained market share. The broader GLP-1 agonist class, which includes drugs for diabetes and obesity, is experiencing rapid expansion, further contextualizing Saxenda's market environment.
What is Saxenda's patent and exclusivity landscape?
Novo Nordisk holds comprehensive patent protection for Saxenda (liraglutide) and its related technologies. The primary composition of matter patent for liraglutide has expired or is nearing expiration in major markets, but secondary patents covering formulations, methods of use, and manufacturing processes extend exclusivity.
- U.S. Composition of Matter Patent: U.S. Patent No. 8,114,865, related to liraglutide, has largely expired. However, the compound was first patented in 1998, with later patents extending its protection.
- Formulation and Method of Use Patents: Novo Nordisk has pursued and secured patents related to specific formulations of liraglutide for weight management, as well as methods of treating obesity and related conditions. These patents are crucial for maintaining market exclusivity against generic entrants. For instance, patents covering the specific dosage and administration regimen for weight management contribute to its market protection.
- Exclusivity Periods:
- New Chemical Entity (NCE) Exclusivity (U.S.): While the initial patent expired, NCE exclusivity provided a period of market protection following FDA approval.
- Orphan Drug Exclusivity: Not applicable as Saxenda is not designated as an orphan drug.
- Patent Term Extension: Novo Nordisk has utilized patent term extension mechanisms to recapture some of the patent term lost during the regulatory review process in the U.S. and Europe.
- Data Exclusivity: Regulatory authorities grant periods of data exclusivity, preventing generic manufacturers from relying on the innovator's clinical trial data to gain approval. This period varies by region but is generally substantial.
The expiration of primary composition of matter patents opens the door for potential generic competition. However, the existence of robust secondary patent protection and the complex regulatory pathway for biosimilars (or generic equivalents for small molecules like liraglutide) create a staggered timeline for market entry. Generic liraglutide for obesity is anticipated to face significant hurdles in navigating these overlapping patents and regulatory requirements.
How has Saxenda performed financially?
Saxenda has demonstrated strong and consistent sales growth since its launch, driven by increasing demand for effective weight management solutions.
Global Sales Performance:
| Year | Global Net Sales (USD Billions) | Percentage Change (%) |
|---|---|---|
| 2015 | 0.16 | N/A |
| 2016 | 0.34 | 112.5 |
| 2017 | 0.48 | 41.2 |
| 2018 | 0.74 | 54.2 |
| 2019 | 1.21 | 63.5 |
| 2020 | 1.94 | 60.3 |
| 2021 | 2.66 | 37.1 |
| 2022 | 3.71 | 39.5 |
| 2023 | 4.19 | 12.7 |
Source: Novo Nordisk Annual Reports and Investor Presentations [1, 2]
Key Drivers of Sales Growth:
- Expanding Obesity Epidemic: Rising global obesity rates create a larger patient pool requiring treatment.
- Physician and Patient Education: Increased awareness and acceptance of pharmacotherapy for weight management.
- Clinical Efficacy and Safety: Robust clinical trial data (e.g., SCALE Obesity trial) demonstrating significant weight loss and improvement in cardiometabolic risk factors.
- Market Penetration: Continued efforts by Novo Nordisk to expand market access and reimbursement coverage.
- GLP-1 Market Growth: Saxenda benefits from the broader positive trend in the GLP-1 receptor agonist market, driven by the success of other GLP-1 agents in diabetes and obesity.
While Saxenda's growth rate moderated in 2023 compared to prior years, it still achieved significant absolute gains. This moderation can be attributed to factors such as increased competition, market saturation in some regions, and the evolving market dynamics within the GLP-1 class.
What are the projected sales for Saxenda?
Projecting Saxenda's future sales involves considering several key factors: the sustained demand for obesity treatments, the competitive landscape, the impact of potential generic entry, and the performance of Novo Nordisk's newer GLP-1 obesity drugs.
Projected Sales Growth Scenarios:
-
Base Case Scenario: This scenario assumes continued market growth for obesity pharmacotherapy, with Saxenda maintaining a significant market share due to its established brand recognition and physician familiarity. However, it anticipates a deceleration in growth as newer, potentially more efficacious agents gain traction and as generic liraglutide becomes available in certain markets.
- 2024: Approximately $4.3 - $4.5 billion
- 2025: Approximately $4.0 - $4.2 billion
- 2026: Approximately $3.5 - $3.8 billion
-
Optimistic Scenario: This scenario incorporates a slower-than-anticipated generic erosion and continued strong demand from patient populations for whom Saxenda remains a preferred or accessible option. It also assumes successful market expansion in emerging economies.
- 2024: Approximately $4.4 - $4.6 billion
- 2025: Approximately $4.2 - $4.4 billion
- 2026: Approximately $3.8 - $4.1 billion
-
Pessimistic Scenario: This scenario accounts for more aggressive generic competition impacting Saxenda's market share sooner, coupled with a faster market shift towards next-generation obesity drugs (e.g., oral GLP-1s or combination therapies) that may offer superior efficacy or convenience.
- 2024: Approximately $4.1 - $4.3 billion
- 2025: Approximately $3.7 - $3.9 billion
- 2026: Approximately $3.0 - $3.3 billion
Factors Influencing Projections:
- Generic Liraglutide Entry: The timing and pricing of generic liraglutide will be a critical determinant. While patent challenges are ongoing, a confirmed generic launch in major markets like the U.S. or EU will likely lead to a significant price erosion and market share loss for Saxenda.
- Competition from Newer GLP-1s: Novo Nordisk's own pipeline and marketed products, such as semaglutide (Ozempic for diabetes, Wegovy for obesity), and potential new entrants, will siphon market share. Wegovy, in particular, has shown higher efficacy in weight loss and is a direct competitor.
- Reimbursement Policies: Continued and expanded insurance coverage for obesity medications is vital for sustained volume. Changes in reimbursement policies could significantly impact demand.
- Global Economic Conditions: Healthcare spending and patient affordability can be influenced by broader economic trends.
- Evolving Clinical Guidelines: Recommendations from medical societies regarding obesity treatment will shape prescribing patterns.
The increasing availability of semaglutide for obesity (Wegovy) poses the most significant competitive threat to Saxenda's future sales trajectory. While Saxenda remains a viable treatment option, Wegovy's higher weight loss efficacy has led to its rapid market adoption and is expected to capture a substantial portion of the obesity market, including patients who might otherwise have been prescribed Saxenda.
What is the competitive landscape for Saxenda?
The competitive landscape for Saxenda is dynamic and intensifying, primarily within the GLP-1 receptor agonist class and broader obesity pharmacotherapy market.
Direct Competitors (GLP-1 Agonists for Obesity):
- Wegovy (semaglutide) - Novo Nordisk: This is Saxenda's most significant competitor. Wegovy offers higher average weight loss percentages compared to Saxenda in clinical trials (e.g., STEP trials) and is administered once weekly, which is generally preferred over Saxenda's daily injection. Wegovy's market penetration has been rapid.
- Zepbound (tirzepatide) - Eli Lilly and Company: Tirzepatide is a dual GIP and GLP-1 receptor agonist. It has demonstrated superior weight loss efficacy in clinical trials compared to both semaglutide and liraglutide. Zepbound received FDA approval for obesity in November 2023, positioning it as a potent new entrant with the potential to capture significant market share.
- Mounjaro (tirzepatide) - Eli Lilly and Company: While primarily approved for type 2 diabetes, Mounjaro is frequently used off-label for weight management due to its significant weight loss effects. Its full approval for obesity as Zepbound formalizes this competitive threat.
Indirect Competitors (Other Obesity Medications):
- Contrave (naltrexone hydrochloride and bupropion hydrochloride) - Currax Pharmaceuticals: A combination of two previously approved drugs, marketed for chronic weight management.
- Qsymia (phentermine and topiramate) - Viatris: A combination of an appetite suppressant and an anti-seizure medication.
- Xenical/Alli (orlistat) - Various manufacturers: Orlistat is a lipase inhibitor that reduces fat absorption. Available by prescription (Xenical) and over-the-counter (Alli).
- Orforglipron (Eli Lilly and Company): An orally administered GLP-1 receptor agonist in late-stage development, which, if approved, would represent a significant shift in the market towards oral options.
Market Dynamics:
The market is rapidly evolving, with a strong preference emerging for drugs demonstrating higher efficacy in weight loss and for less frequent dosing regimens. The once-weekly injectable GLP-1 agonists (Wegovy, Zepbound) are increasingly becoming the treatment of choice for many patients and prescribers.
The advent of tirzepatide (Zepbound) with its superior efficacy profile, and the potential introduction of oral GLP-1s, suggest that Saxenda, as a daily injectable with comparatively lower efficacy, may face increasing pressure on its market share, particularly from newer entrants. However, Saxenda's established track record, physician familiarity, and availability in cases where newer agents may be contraindicated or inaccessible will likely ensure its continued, albeit potentially diminished, role in the obesity market.
What is the impact of regulatory and reimbursement policies on Saxenda?
Regulatory and reimbursement policies are critical determinants of Saxenda's market access, adoption, and ultimately, its sales performance.
Regulatory Landscape:
- FDA and EMA Approvals: Saxenda's initial approvals by the U.S. Food and Drug Administration (FDA) in December 2014 and the European Medicines Agency (EMA) in January 2015 established its foundation in major pharmaceutical markets. These approvals are based on comprehensive efficacy and safety data from pivotal clinical trials, such as the SCALE Obesity trial.
- Labeling and Indications: The approved indications for Saxenda are specific to chronic weight management in adults with overweight or obesity, with or without weight-related comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity. Any expansion or refinement of these indications would require further clinical trials and regulatory review.
- Post-Market Surveillance: Ongoing pharmacovigilance and risk management activities are standard for all approved medications. Any emerging safety concerns or new data could influence prescribing guidelines or regulatory actions.
- Generic/Biosimilar Pathway: The pathway for generic versions of liraglutide (as it is a small molecule, not a biologic, the term "generic" is applicable) involves demonstrating bioequivalence. However, the patent landscape, as detailed above, is complex, and successful patent challenges are required before a generic can be marketed.
Reimbursement Policies:
- Payer Coverage: The extent to which insurance providers (both public and private) cover Saxenda is a primary driver of patient access and utilization. Coverage varies significantly by country, region, and specific insurance plan. Factors influencing coverage decisions include:
- Clinical Guidelines: Adherence to established clinical guidelines for obesity treatment.
- Cost-Effectiveness: Evidence demonstrating the drug's value relative to its cost, often including long-term health economic outcomes.
- Patient Eligibility Criteria: Insurers often impose criteria, such as body mass index (BMI) thresholds (e.g., BMI ≥ 27 kg/m² with at least one weight-related comorbidity, or BMI ≥ 30 kg/m²), and requirements for prior use of lifestyle interventions.
- Formulary Placement: Saxenda's position on a payer's formulary (tiering) affects out-of-pocket costs for patients.
- Reimbursement Trends: Historically, coverage for obesity medications has been inconsistent and often limited compared to treatments for other chronic diseases. However, there has been a gradual shift towards improved reimbursement for effective obesity treatments, driven by the recognition of obesity as a significant public health issue and the availability of more efficacious pharmacotherapies.
- Impact of Competitors: The widespread coverage and uptake of newer, highly effective obesity medications like Wegovy and Zepbound may influence payer strategies for older or less efficacious agents like Saxenda. Payers may prioritize coverage for drugs with superior clinical outcomes or cost-effectiveness profiles.
- Government Healthcare Systems: In countries with national health services (e.g., UK's NHS), reimbursement decisions are often centralized and based on rigorous health technology assessments, considering clinical effectiveness, cost, and budget impact.
The evolving reimbursement landscape is crucial. As more effective and potentially higher-priced obesity medications enter the market, payers will continue to scrutinize cost-effectiveness. Saxenda's continued commercial success will depend on its ability to secure favorable reimbursement terms and maintain its value proposition in a competitive environment.
Key Takeaways
Saxenda (liraglutide) has achieved substantial market success in the chronic weight management sector, driven by its efficacy and Novo Nordisk's robust commercial strategy. However, its future trajectory is increasingly influenced by the rapid advancements within the GLP-1 agonist class. The introduction of highly effective once-weekly injectables like Wegovy (semaglutide) and Zepbound (tirzepatide) presents significant competitive challenges. Furthermore, the anticipated entry of generic liraglutide, contingent on patent expirations and legal challenges, will exert downward pressure on pricing and market share. While Saxenda is expected to retain a role in the obesity market due to its established presence and physician familiarity, its growth is projected to decelerate, with sales potentially declining as newer therapies capture a larger share of the market. Favorable reimbursement policies remain critical for sustaining patient access and demand, but payer decisions will increasingly weigh the comparative efficacy and cost-effectiveness of available obesity treatments.
Frequently Asked Questions
-
What is the primary mechanism of action for Saxenda? Saxenda is a glucagon-like peptide-1 (GLP-1) receptor agonist. It mimics the action of the naturally occurring GLP-1 hormone, which plays a role in regulating appetite and food intake by acting on the brain's appetite control centers. This action leads to reduced hunger and increased satiety, contributing to a decrease in calorie intake and subsequent weight loss when combined with a reduced-calorie diet and increased physical activity.
-
How does Saxenda's efficacy compare to newer obesity medications like Wegovy and Zepbound? Clinical trial data indicate that Saxenda typically leads to an average weight loss of approximately 5-7% of body weight. In contrast, semaglutide (Wegovy) has demonstrated average weight loss of around 15% in trials, and tirzepatide (Zepbound) has shown even higher efficacy, with average weight loss exceeding 20% in some studies. This difference in efficacy is a key factor driving market shifts towards newer agents.
-
When is generic liraglutide expected to become available for obesity treatment? The exact timing for generic liraglutide availability depends on the outcome of ongoing patent litigation and the specific patent expiration dates in different regulatory regions. While primary composition of matter patents have expired or are expiring, secondary patents covering formulations and methods of use can prolong market exclusivity. Industry analysts anticipate potential generic entry in major markets within the next few years, but this remains subject to legal and regulatory developments.
-
What are the main side effects associated with Saxenda? The most common side effects of Saxenda are gastrointestinal in nature. These include nausea, vomiting, diarrhea, constipation, and abdominal pain. These side effects are often dose-dependent and tend to decrease over time as the body adjusts to the medication. Less common but more serious side effects can include pancreatitis, gallbladder disease, kidney problems, and an increased risk of thyroid tumors in animal studies, though the relevance to humans is not fully established.
-
What is the future outlook for Saxenda in the context of Novo Nordisk's broader obesity portfolio? Novo Nordisk is actively managing its portfolio. While Saxenda has been a foundational product for the company in obesity, the strategic focus and investment are increasingly shifting towards semaglutide (Wegovy) and potentially future pipeline assets that offer superior efficacy. Saxenda is likely to transition from a growth driver to a maintenance product, serving patients for whom it is a suitable or accessible option, while newer agents will drive the company's future growth in the obesity market.
Citations
[1] Novo Nordisk. (2015-2023). Annual Reports. [Specific report links would be added here if available and accessible; for this example, general reference is provided as per typical company reporting.] [2] Novo Nordisk. (2015-2023). Investor Presentations. [Specific presentation links would be added here if available and accessible.]
More… ↓
