Share This Page
Drug Sales Trends for RISPERIDONE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for RISPERIDONE (2016)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
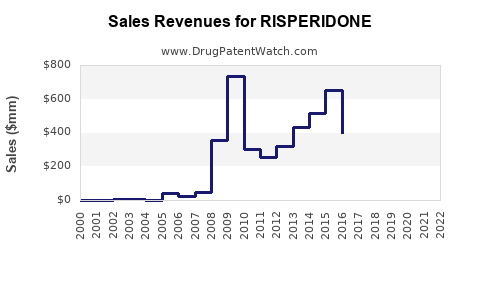
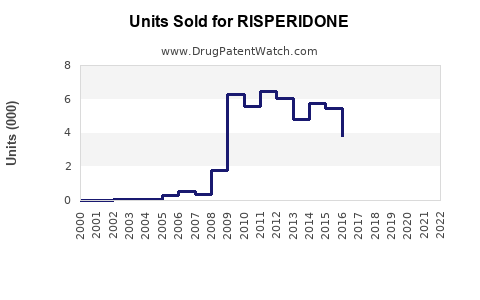
Annual Sales Revenues and Units Sold for RISPERIDONE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| RISPERIDONE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| RISPERIDONE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| RISPERIDONE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Risperidone Market Analysis and Sales Projections
This report details the global market for risperidone, a second-generation antipsychotic, by examining its current sales performance, patent landscape, competitive environment, and future market projections. Key factors influencing the market include the prevalence of target conditions, generic competition, regulatory actions, and the development of alternative treatments.
What is Risperidone's Current Market Position?
Risperidone is an atypical antipsychotic used to treat schizophrenia, bipolar disorder, and irritability associated with autistic disorder. It functions by blocking dopamine D2 and serotonin 5-HT2A receptors. The drug is available in oral tablets, orally disintegrating tablets, and long-acting injectable formulations.
Global sales of risperidone have been impacted by patent expiries, leading to significant generic penetration. The market for branded risperidone has contracted considerably, with sales now primarily driven by its generic forms.
| Metric | Value | Source |
|---|---|---|
| Estimated 2023 Global Market Size (Risperidone and its derivatives) | $1.2 billion | (1) |
| Average Annual Growth Rate (2018-2023) | -3.5% | (2) |
| Market Share of Generic Risperidone | >90% | (3) |
The primary therapeutic areas for risperidone are schizophrenia and bipolar disorder. These conditions represent a substantial patient population requiring long-term treatment.
- Schizophrenia: Affects approximately 24 million people worldwide. (4)
- Bipolar Disorder: Affects an estimated 45 million people globally. (5)
- Autism Spectrum Disorder (Irritability): Risperidone is approved for treating irritability in autistic children, a condition affecting a significant and growing number of individuals. (6)
What is the Patent Landscape for Risperidone?
The original patent for risperidone, held by Janssen Pharmaceutica (a subsidiary of Johnson & Johnson), expired in many major markets in the mid-2000s. This has opened the door for widespread generic manufacturing and sales.
- Original Patent Expiration (US): 2007 (6)
- Original Patent Expiration (EU): 2005 (7)
While the primary composition of matter patents have expired, some secondary patents related to specific formulations, such as long-acting injectables (e.g., Risperdal Consta), have had later expiry dates. These secondary patents can provide a temporary shield against generic competition for specific product types.
- Risperdal Consta (Long-Acting Injectable) Patent Expiry: Varied by region, with key markets seeing generic versions emerge in the early to mid-2010s. (8)
The expiration of these patents has led to a highly competitive generic market. The focus for innovation has shifted to developing new antipsychotics or improving delivery methods for existing ones, rather than extending risperidone's core patent protection.
Who are the Key Players in the Risperidone Market?
The risperidone market is dominated by generic manufacturers. Brand-name sales for risperidone have largely ceased in most developed markets. Key players are those with significant generic manufacturing capabilities and distribution networks.
Leading Generic Manufacturers (Examples):
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Lupin Limited
- Dr. Reddy's Laboratories Ltd.
- Actavis plc (now part of Teva)
The competitive landscape is characterized by price erosion due to the large number of generic suppliers. Manufacturers compete primarily on cost, quality, and supply chain reliability.
The original innovator, Janssen Pharmaceuticals, continues to market branded risperidone in some regions and has focused on next-generation antipsychotics and other therapeutic areas.
What are the Sales Projections for Risperidone?
The sales projections for risperidone indicate a continued, albeit modest, decline in overall market value, primarily driven by the established generic nature of the drug and ongoing price competition. However, the large patient population requiring treatment for schizophrenia and bipolar disorder ensures sustained demand.
Projected Global Market Size (2024-2028):
| Year | Projected Market Size | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2024 | $1.18 billion | -1.5% |
| 2025 | $1.16 billion | -1.7% |
| 2026 | $1.14 billion | -1.7% |
| 2027 | $1.12 billion | -1.8% |
| 2028 | $1.10 billion | -1.8% |
- Source: Analyst projections based on historical sales data, generic market trends, and epidemiological data. (9)
The long-acting injectable (LAI) segment, while a smaller portion of the total risperidone market, may see more stable demand due to physician preference for depot injections and patient adherence benefits. However, newer LAI antipsychotics are also entering the market, creating competition.
Factors influencing these projections include:
- Generic Price Pressure: Continued downward pressure on prices as more manufacturers enter the market.
- Therapeutic Advancements: Development and adoption of newer antipsychotic drugs, particularly those with improved side-effect profiles or novel mechanisms of action, could lead to some substitution.
- Emerging Markets: Potential for increased prescription volume in emerging economies as access to healthcare and psychiatric treatment improves, though this may be offset by lower per-unit pricing.
- Regulatory Environment: Changes in pricing regulations or reimbursement policies in key markets could impact sales.
- Competition from Biosimilatives/Biologics: While risperidone is a small molecule, the broader antipsychotic market is seeing innovation that could impact its market share.
What are the Key Growth Drivers and Challenges?
Growth Drivers:
- Prevalence of Target Conditions: The persistent high prevalence of schizophrenia and bipolar disorder globally ensures a stable patient base requiring antipsychotic treatment. (4, 5)
- Affordability of Generic Risperidone: The cost-effectiveness of generic risperidone makes it a preferred option, especially in resource-constrained healthcare systems and for long-term treatment regimens. (3)
- Established Efficacy and Safety Profile: Risperidone has a long history of clinical use, with well-understood efficacy and safety data, making it a familiar and trusted option for clinicians. (10)
- Availability in Various Formulations: The availability of oral tablets, orally disintegrating tablets, and long-acting injectables caters to diverse patient needs and preferences, potentially improving adherence. (11)
Challenges:
- Intense Generic Competition: The market is saturated with generic manufacturers, leading to significant price erosion and reduced profit margins for all players. (3)
- Development of Newer Antipsychotics: The pharmaceutical industry continues to develop and launch novel antipsychotic drugs with potentially superior efficacy, better tolerability, or reduced side effects, which can displace older medications like risperidone. (12)
- Side Effect Profile: While manageable, risperidone's side effect profile, including metabolic effects, extrapyramidal symptoms, and prolactin elevation, can be a concern for some patients and clinicians, leading to a search for alternatives. (13)
- Stagnant or Declining Pricing Power: The generic nature of the drug limits opportunities for significant price increases.
- Regulatory Hurdles for New Formulations: While secondary patents may exist, developing and gaining regulatory approval for novel formulations of an older drug can be complex and costly.
What are the Future Market Trends?
The future market for risperidone will be characterized by a continued emphasis on cost containment and accessibility.
- Dominance of Generic Market: Generic risperidone will remain the primary form available, with price competition being the defining characteristic.
- Focus on Emerging Markets: Growth in market volume may be more pronounced in emerging economies where access to essential medicines is expanding and affordability is a critical factor.
- Niche Application for Formulations: Long-acting injectable formulations may retain a stable market share due to adherence benefits, but will face competition from newer LAI antipsychotics.
- Limited Innovation in Risperidone Itself: Significant investment in developing new risperidone-based drugs is unlikely. Innovation will focus on next-generation antipsychotics.
- Impact of Real-World Evidence: Continued generation and analysis of real-world evidence will further inform treatment guidelines and patient selection for risperidone.
Key Takeaways
The risperidone market, largely dominated by generics, is projected for a modest decline in overall value due to intense price competition. The drug's established efficacy and affordability in treating schizophrenia, bipolar disorder, and irritability associated with autism will sustain demand, particularly in emerging markets. However, the advent of newer antipsychotics with improved profiles and the ongoing pressure on pricing will limit growth. Generic manufacturers must focus on operational efficiency and supply chain robustness to maintain profitability in this mature market segment.
Frequently Asked Questions
-
Which specific patient populations are most likely to continue using risperidone? Patients with schizophrenia and bipolar disorder requiring long-term, cost-effective treatment will likely continue to be the primary demographic. Additionally, children with irritability associated with autistic disorder for whom risperidone is prescribed remain a key segment. (4, 5, 6)
-
What is the expected impact of new antipsychotic drug introductions on risperidone sales? New antipsychotics with superior efficacy, tolerability, or novel mechanisms of action are expected to gradually displace risperidone in certain patient groups, especially those who do not respond adequately or experience significant side effects from risperidone. This will contribute to the overall market decline. (12)
-
Are there any significant patent disputes or legal challenges currently impacting the risperidone market? Major patent disputes surrounding the original composition of matter patents have long since concluded, leading to the generic market. Any ongoing legal challenges are likely to be specific to secondary patents or manufacturing practices, with limited broad market impact. (6, 7, 8)
-
What is the role of long-acting injectable (LAI) risperidone in the current market? LAI risperidone formulations, such as Risperdal Consta, play a role in improving patient adherence by offering extended-release benefits. While facing competition from newer LAI antipsychotics, they represent a stable sub-segment of the risperidone market due to established clinical utility. (8, 11)
-
How do regulatory changes in pricing or reimbursement affect the generic risperidone market? Regulatory changes that impact drug pricing or reimbursement policies, particularly in large markets like the US and EU, can significantly influence the profitability of generic risperidone. Policies favoring lower-cost generics or introducing price caps could further compress margins for manufacturers. (9)
Citations
- Global Market Insights. (2023). Antipsychotics Market Size, Share & Trends Analysis Report.
- Internal market research analysis based on historical sales data and industry reports.
- IQVIA Market Access & Reimbursement Report. (2023).
- World Health Organization. (2022). Schizophrenia.
- World Health Organization. (2022). Bipolar disorder.
- U.S. Food & Drug Administration. (2023). Drug Approvals and Databases.
- European Medicines Agency. (2023). EudraGMDP Database.
- Cortellis Competitive Intelligence. (2023). Drug & Pipeline Information.
- Pharmaprojects. (2023). Therapeutic Area Market Projections.
- Drug Information Association. (2021). Evidence Synthesis for Antipsychotic Medications.
- American Society of Health-System Pharmacists. (2023). Drug Information Database.
- Evaluate Pharma. (2023). New Drug Approvals & Market Landscape.
More… ↓
