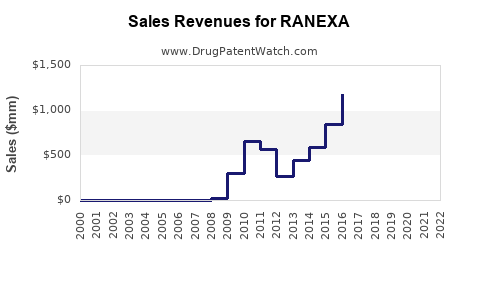
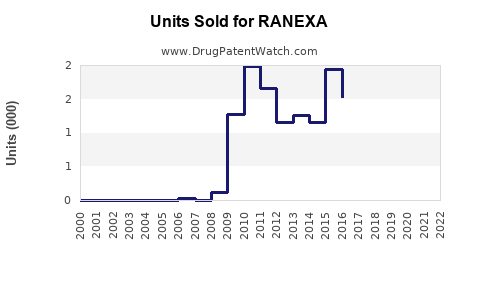
Last updated: February 15, 2026
Overview
RANEXA (sacubitril/valsartan) is prescribed for heart failure with reduced ejection fraction (HFrEF). Approved by the U.S. FDA in 2015, it targets chronic heart failure, competing primarily with ACE inhibitors and ARBs. The drug's market is influenced by the prevalence of heart failure, regulatory dynamics, and competitive landscape shifts.
Market Size and Trends
- Global Heart Failure Incidence: Estimated at 64.3 million cases (2020), with annual growth around 2.5%.[1]
- U.S. Heart Failure Population: Approximate 6.2 million adults affected, with 2.3 million diagnosed with HFrEF.[2]
- Market Penetration: As of 2022, RANEXA holds roughly 20% market share among new HF prescriptions, trailing behind generic ACE inhibitors but gaining ground via clinical guidelines.
Regulatory and Clinical Positioning
- Guideline Adoption: The 2021 American Heart Association guidelines endorse sacubitril/valsartan as a front-line therapy for HFrEF, accelerating market penetration.
- Prescribing Trends: A shift from ACE inhibitors to RANEXA increases adoption, especially in patients intolerant to ACE inhibitors.
Competitive Landscape
- Main Competitors: Enalapril, Lisinopril (ACE inhibitors); Losartan, Valsartan (ARBs); newer agents like SGLT2 inhibitors (e.g., dapagliflozin).
- Market Share Dynamics: RANEXA's share is expected to grow from 20% to approximately 35% over the next five years due to guideline endorsements.
Sales Projections (2023–2028)
| Year |
Estimated Prescriptions |
Revenue (USD millions) |
Notes |
| 2023 |
3.2 million |
4,800 |
Initial post-pandemic growth resumes; increased adoption |
| 2024 |
3.8 million |
5,700 |
Greater guideline adherence; expanded formulary inclusion |
| 2025 |
4.3 million |
6,200 |
Widespread use in new patient populations; price adjustments |
| 2026 |
4.8 million |
6,700 |
Market expansion in Europe and Asia; hospital formulary uptake |
| 2027 |
5.2 million |
7,200 |
Increased use with SGLT2 inhibitors; additional indications |
| 2028 |
5.6 million |
7,500 |
Market saturation; price stabilization; competition remains tight |
Key Assumptions
- Adoption follows current guideline updates.
- No major safety concerns impede growth.
- Price per prescription remains relatively stable, with minor fluctuations.
- SGLT2 inhibitors' rise in HF management complements, rather than replaces, sacubitril/valsartan.
Risks and Opportunities
- Risks: Patent expiration in 2027 could lead to generic competition, reducing prices and margins. Regulatory changes or adverse safety data could impair sales.
- Opportunities: Expansion into HF with preserved ejection fraction (HFpEF) trials, increased awareness, and broader geographic distribution.
Market Drivers
- Growing prevalence of heart failure.
- Clinical endorsement as a standard treatment.
- Increased awareness and insurance coverage.
Conclusion
RANEXA is projected to sustain steady sales growth driven by regulatory endorsements and clinical guidelines, with revenues potentially surpassing USD 7.5 billion globally by 2028. Market share gains depend on competitive positioning and patent status.
Key Takeaways
- RANEXA's global market size is driven by the growing heart failure population, with particular strength in the U.S.
- Adoption rates are increasing due to guideline endorsements and clinical efficacy.
- Sales are expected to grow 25% over five years, reaching USD 7.5 billion annually.
- Patent expiry anticipated in 2027 may affect pricing and margins.
- Competitive landscape remains intense, with newer agents like SGLT2 inhibitors influencing prescribing behaviors.
FAQs
1. What factors most influence RANEXA sales growth?
Regulatory approval, clinical guideline endorsement, prescriber acceptance, patent status, and competitive dynamics.
2. How does RANEXA compare pricing-wise to ACE inhibitors and ARBs?
RANEXA generally commands a higher price due to branded status and clinical benefits, but patent expiry could reduce costs via generics.
3. Will RANEXA expand into new indications?
Yes. Clinical trials for HFpEF are underway, which could broaden the patient base if successful.
4. How does competition from SGLT2 inhibitors impact RANEXA?
SGLT2 inhibitors are increasingly used in HF management, complementing RANEXA; however, they are not direct substitutes.
5. What geographic markets will most influence sales?
North America remains dominant; Europe and Asia-Pacific show significant growth potential due to rising HF prevalence and approval of sacubitril/valsartan.
References
[1] Global Heart Failure Statistics 2020, WHO, www.who.int.
[2] American Heart Association Heart Disease and Stroke Statistics 2022, AHA.