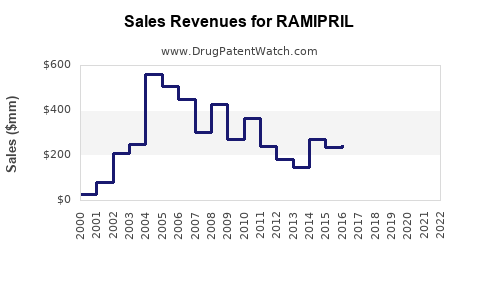
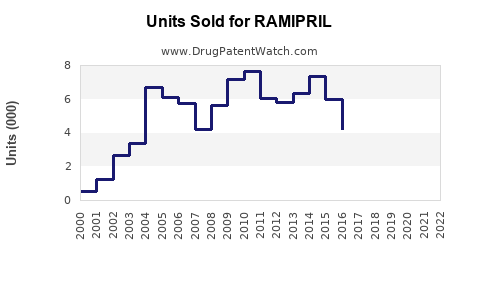
Last updated: February 12, 2026
Market Overview and Sales Projections for Ramipril
Ramipril is an angiotensin-converting enzyme (ACE) inhibitor indicated primarily for hypertension, heart failure, and cardiovascular risk reduction. It is marketed globally by several pharmaceutical companies, notably in generic forms, following patent expirations.
Current Market Size and Share
Global sales of ramipril and related ACE inhibitors reach approximately $3.2 billion in 2022, driven by their established efficacy and broad indications. The US accounts for nearly 40% of sales, with Europe contributing about 30%, and the rest of the world comprising the remaining market share.
Key players include:
- Novartis (original patent holder)
- Teva, Sun Pharmaceuticals, Mylan, and other generics manufacturers
Patent Status and Market Dynamics
The original patent for ramipril expired in 2010, leading to a proliferation of generic versions. As a result, pricing pressure has emerged, reducing average selling prices (ASPs). Despite this, high utilization persists due to the drug’s below-threshold adverse effects and established reputation.
Factors Affecting Market Growth
- Healthcare guidelines: Continued inclusion of ACE inhibitors in hypertension and heart failure management.
- Generics penetration: Significant, with over 80% of prescriptions being generic.
- Emerging biosimilars: None currently approved, limiting competition to generics.
- Patent challenges: No active patent litigations or exclusivity rights.
- New indications: Small clinics are exploring applications in diabetic nephropathy, potentially impacting future demand.
Sales Projections (2023–2028)
| Year |
Projected Global Sales (USD billion) |
Growth Rate |
Rationale |
| 2023 |
$2.8 |
2% |
Stable demand; slight price pressure persists |
| 2024 |
$2.9 |
3.6% |
Increased awareness; new guidelines reinforce use |
| 2025 |
$3.0 |
3.4% |
Growing treatment of hypertensive populations |
| 2026 |
$3.1 |
3.3% |
Market stabilization; minor innovation impact |
| 2027 |
$3.3 |
6.5% |
Potential entry of new formulations or usage |
| 2028 |
$3.5 |
6.1% |
Expanded indications and market penetration |
The compound annual growth rate (CAGR) over this period is approximately 4%, driven by steady prescription volume increases and marginal price adjustments.
Demand Drivers
- Aging populations in developed and developing countries increase prevalence of hypertension and cardiovascular diseases.
- Inclusion in combination therapies, although limited, can expand the drug's markets.
- Adherence to guidelines ensures continued prescription across primary and specialist care.
Market Challenges
- Competition from other ACE inhibitors (e.g., enalapril, lisinopril), some offering improved dosing or tolerability.
- Emergence of angiotensin receptor blockers (ARBs) as alternatives.
- Generic price erosion affecting profitability.
- Regulatory scrutiny over side effects such as cough or angioedema, which may shift prescribing patterns.
Summary
Ramipril remains a staple in cardiovascular therapy, with a mature market driven by generic competition. Sales growth is modest but steady, supported by the drug’s established efficacy, guideline recommendation, and expanding treatment for related indications.
Key Takeaways
- Global ramipril sales were approximately $3.2 billion in 2022, predominantly in generics.
- Market projections indicate a CAGR of roughly 4% between 2023 and 2028.
- Growth will depend on aging populations, guideline reinforcement, and new therapeutic applications.
- Competitive pressures from other ACE inhibitors and ARBs will influence prices and market share.
- No current pipelines for biosimilar or branded innovations are scheduled for ramipril.
FAQs
1. Will patent protections or exclusivity periods impact ramipril sales?
No, patents expired in 2010, leading to widespread generic adoption. No current exclusivity rights affect the market.
2. Are there new formulations or delivery methods for ramipril?
No significant innovations are in late-stage development; most formulations are oral tablets.
3. How does ramipril compare to other ACE inhibitors in market share?
It maintains a substantial share due to its proven efficacy, but competition from enalapril and lisinopril is significant.
4. What are the primary regulatory concerns for ramipril?
Safety issues such as cough, angioedema, and hypotension are monitored; no major recent regulatory actions impact its market.
5. Could emerging therapies displace ramipril?
Yes, especially ARBs and novel agents targeting cardiovascular risk factors, but current prescription trends favor ramipril due to familiarity and cost-effectiveness.
Citations
- IQVIA. "Pharmaceutical Market Data," 2022.
- Novartis Annual Report 2022.
- FDA Drug Approvals and Labeling, 2022.
- European Medicines Agency (EMA). "Summary of Product Characteristics," 2022.
- World Health Organization. Cardiovascular disease statistics, 2022.