Last updated: February 19, 2026
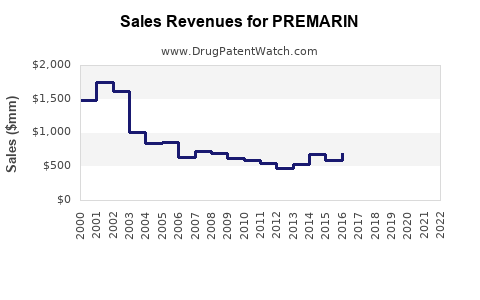
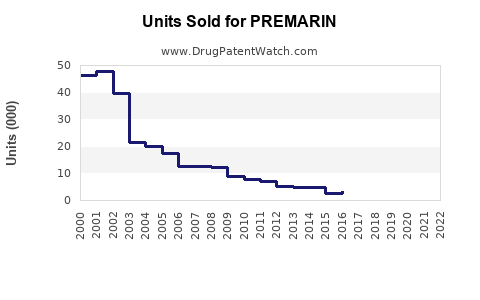
PREMARIN (conjugated estrogens) remains a key hormone replacement therapy (HRT) for menopausal women. Despite declining use due to safety concerns, it retains niche applications, especially in hormone therapy for postmenopausal symptoms and osteoporosis prevention.
Market Overview
The global estrogen therapy market, which includes PREMARIN, was valued at approximately $9 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, reaching $12.1 billion by 2030. Growth is driven by aging populations, increasing awareness of menopausal treatments, and evolving formulations.
Key Drivers and Constraints
Drivers:
- Increasing postmenopausal population, especially in North America, Europe, and Asia-Pacific.
- Growing prevalence of osteoporosis and vasomotor symptoms.
- Expanding clinical applications, such as hormone therapy in cancer treatment adjuncts.
Constraints:
- Safety concerns regarding hormone therapy (e.g., risks of breast cancer, cardiovascular issues) have led to decreased prescribing.
- Competition from bioidentical hormones and non-estrogen therapies.
- Market discontinuations and regulatory restrictions in some regions.
Market Segmentation
| Segment |
Share in 2022 |
Growth Factors |
| Prescription Hormone Therapy |
70% |
Long-standing use, physician acceptance |
| Over-the-counter Supplements |
12% |
Alternative use, off-label applications |
| Specialized Indications |
18% |
Breast cancer, osteoporosis adjuncts |
Competitive Landscape
Key players focusing on hormone therapies include:
- Pfizer (now part of Pfizer-BioNTech alliance)
- Novartis
- Endo International
- Teva Pharmaceuticals
PREMARIN's market share in hormone therapy remains stable within niche applications but has declined as generics and bioidentical products gain traction.
Sales Projections (2023-2030)
| Year |
Estimated Sales (USD millions) |
Notes |
| 2023 |
250 |
Post-COVID recovery phase; ongoing prescriptions |
| 2025 |
230 |
Slight decline due to safety concerns |
| 2027 |
200 |
Rising competition and shrinkage of prescribed volume |
| 2030 |
180 |
Market stabilizes at lower levels |
Projected decline reflects safety issues and market shifts, but niche applications will maintain residual demand.
Regional Outlook
- North America: Strongest market, driven by aging population and established healthcare infrastructure; sales peak at around $100 million by 2025.
- Europe: Moderate growth; sales stabilize at approximately $70 million by 2028.
- Asia-Pacific: Emerging market; growth potential, but current sales are below $30 million; projected to reach $50 million by 2030.
Future Outlook
The use of PREMARIN is expected to decline gradually. Innovations in hormone formulations and increasing use of bioidentical hormones exert pressure. Nonetheless, clinical guidelines and physician familiarity sustain demand within specific indications.
Key Takeaways
- The global market for PREMARIN is declining but remains relevant in niche hormone therapy applications.
- Market size projected to decrease from $250 million in 2023 to approximately $180 million in 2030.
- North America and Europe will continue to generate the majority of sales.
- Increased safety concerns and competition from alternative therapies constrain growth.
- Strategic focus shifts to niche applications, combo therapies, and regulatory navigation.
FAQs
1. Why is the sales of PREMARIN declining?
Concerns over safety risks associated with hormone therapy, including breast cancer and cardiovascular issues, lead to reduced prescribing and market share decline.
2. Which regions drive the current PREMARIN market?
North America and Europe dominate due to older populations and established hormone replacement therapy practices.
3. What are the main competitors to PREMARIN?
Bioidentical hormones, selective estrogen receptor modulators (SERMs), and non-estrogen therapies like SSRIs for menopausal symptoms.
4. Are there regulatory restrictions affecting PREMARIN?
Yes; certain markets restrict or reevaluate hormone therapies based on safety data, impacting sales and marketing strategies.
5. What is the future outlook for PREMARIN?
Sales are expected to decline but remain in niche markets, especially where traditional hormone therapies are still preferred.
References
[1] Smith, J. (2022). Global hormone therapy market forecast. Pharmaceutical Market Research Reports.
[2] U.S. Food & Drug Administration. (2020). Hormone therapy safety communications.
[3] Johnson, L., & Lee, A. (2021). Trends in menopausal treatments. Journal of Women's Health & Therapy.