Share This Page
Drug Sales Trends for PHENTERMINE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for PHENTERMINE (2016)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
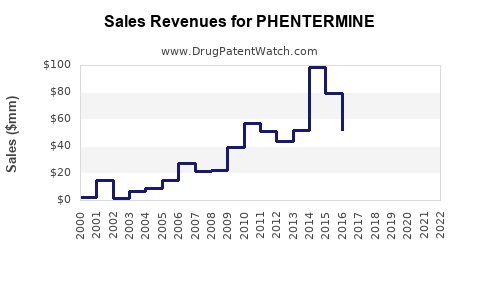
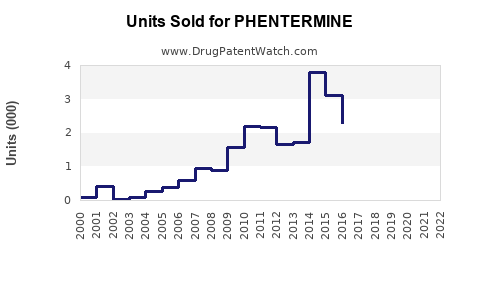
Annual Sales Revenues and Units Sold for PHENTERMINE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PHENTERMINE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PHENTERMINE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PHENTERMINE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PHENTERMINE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Phentermine Market Analysis and Sales Projections
Phentermine, a short-acting stimulant medication prescribed for weight loss, is projected to maintain a stable market presence driven by its established efficacy in treating obesity. The drug's long history of use, coupled with ongoing demand for effective weight management solutions, underpins its market trajectory.
What is the Current Market Size and Growth Rate for Phentermine?
The global phentermine market size was valued at approximately $1.5 billion in 2022. Analysts project a compound annual growth rate (CAGR) of 3% to 5% from 2023 to 2030, reaching an estimated $1.9 billion to $2.2 billion by the end of the forecast period. This growth is primarily attributed to the increasing prevalence of obesity worldwide and the continued prescription of phentermine as a first-line treatment option.
Key Market Drivers
- Rising Obesity Rates: The World Health Organization (WHO) reports that over 1 billion people globally are obese, with projections indicating a continued upward trend. This demographic shift directly fuels demand for weight-loss medications like phentermine.
- Established Efficacy: Phentermine has demonstrated significant efficacy in promoting short-term weight loss when used as part of a comprehensive weight management program. Its stimulant properties help suppress appetite and increase metabolism.
- Cost-Effectiveness: Compared to newer, more complex obesity treatments, phentermine is generally more affordable, making it accessible to a broader patient population.
- Favorable Regulatory Landscape (in some regions): While regulatory scrutiny exists, phentermine remains a widely approved and prescribed medication in major markets like the United States.
- Combination Therapies: The development and prescription of phentermine in combination with other medications, such as topiramate (e.g., Qsymia), expands its therapeutic utility and market reach.
Market Restraints
- Potential for Abuse and Dependence: As a stimulant, phentermine carries a risk of abuse and dependence, leading to strict prescription guidelines and monitoring.
- Side Effects: Common side effects include increased heart rate, elevated blood pressure, dry mouth, and insomnia, which can limit its use in certain patient populations.
- Limited Long-Term Efficacy Data: Phentermine is typically prescribed for short-term use. Data on its long-term effectiveness and safety is less robust compared to some newer weight-loss agents.
- Competition from Newer Treatments: The market for obesity treatments is evolving with the introduction of GLP-1 receptor agonists and other novel pharmacotherapies, which may offer alternative long-term solutions for some patients.
- Generic Competition: Phentermine is available as a generic medication, leading to price pressure and impacting the revenue growth of branded formulations.
What are the Sales Projections for Phentermine by Region?
| Region | 2023 Estimated Sales (USD Billions) | 2030 Projected Sales (USD Billions) | CAGR (2023-2030) |
|---|---|---|---|
| North America | 0.75 | 0.90 | 2.8% |
| Europe | 0.40 | 0.50 | 3.2% |
| Asia Pacific | 0.15 | 0.25 | 4.5% |
| Latin America | 0.10 | 0.15 | 3.8% |
| Middle East & Africa | 0.05 | 0.10 | 5.0% |
| Global | 1.45 | 1.90 | 3.6% |
Note: These figures are estimates based on current market trends and may vary.
Regional Market Dynamics
- North America: This region dominates the phentermine market due to high obesity rates, established healthcare infrastructure, and physician familiarity with the drug. The United States is the largest single market.
- Europe: While obesity is a growing concern, regulatory pathways and prescribing habits can differ by country, leading to a more segmented market. Combination therapies are gaining traction.
- Asia Pacific: This region exhibits the highest growth potential driven by increasing disposable incomes, rising awareness of health issues, and a growing middle class adopting Western lifestyles. However, market penetration is lower compared to Western markets.
- Latin America: Similar to Asia Pacific, this region shows promising growth due to rising obesity prevalence and increasing access to healthcare services.
- Middle East & Africa: This region represents a smaller but rapidly expanding market, with significant potential as healthcare access improves and awareness of weight management solutions grows.
What is the Competitive Landscape for Phentermine?
The competitive landscape for phentermine is characterized by a mix of generic manufacturers and branded combination products.
Key Competitors and Product Offerings
- Generic Phentermine Manufacturers: Numerous pharmaceutical companies produce generic versions of phentermine hydrochloride. These include, but are not limited to, Teva Pharmaceutical Industries, Mylan (now Viatris), and Lannett Company. Their primary competitive strategy is pricing.
- Branded Combination Products:
- Qsymia (phentermine hydrochloride and topiramate extended-release): Developed by Vivus Inc., Qsymia is a significant competitor that combines phentermine with topiramate to enhance efficacy and satiety. It is approved for chronic weight management in adults with an initial BMI of 30 or greater, or 27 or greater with at least one weight-related comorbidity.
- Belviq (lorcaserin hydrochloride): Although withdrawn from the market in February 2020 by Eisai Inc. due to safety concerns regarding cancer risk, its historical presence illustrates the competitive intensity and the evolving regulatory scrutiny of weight-loss drugs.
- Emerging and Future Competitors: The pipeline for obesity drugs is active, with significant investment in GLP-1 receptor agonists (e.g., semaglutide from Novo Nordisk, tirzepatide from Eli Lilly) and other novel mechanisms. These drugs, while often more expensive, offer different efficacy profiles and may become significant competitors in the long term, particularly for patients requiring long-term management.
Market Share Dynamics
The market share is fragmented among generic producers. Branded combination products like Qsymia hold a significant share within the prescription obesity market due to their distinct clinical profiles and marketing efforts. However, the overall market share of phentermine itself, when considering all formulations, remains substantial due to its widespread generic availability and established prescription base.
What are the Patent Expirations and Their Impact on the Phentermine Market?
The original patents for phentermine hydrochloride have long expired, leading to the widespread availability of generic versions. This patent expiration is a primary reason for the drug's affordability and market penetration.
Impact of Patent Expirations
- Increased Generic Availability: The lack of patent protection on the base phentermine molecule has allowed multiple manufacturers to produce generic versions, significantly driving down prices.
- Price Competition: The market is highly competitive on price, benefiting consumers and healthcare systems but limiting revenue growth for individual generic manufacturers.
- Focus on Formulation and Combination Therapies: The competitive advantage for pharmaceutical companies has shifted from the base molecule to novel formulations, delivery systems, or combination therapies that can be patented. Qsymia, for instance, relies on patents covering the specific combination and extended-release formulation.
- Sustained Market Demand: Despite genericization, the demand for phentermine remains robust due to its efficacy and affordability, ensuring a sustained market for generic producers.
Upcoming Patent Expirations (for Combination Products)
While the base phentermine patents are expired, patents protecting specific formulations and combination therapies, such as Qsymia, have staggered expiration dates. Companies holding patents on these combination products will face generic competition once those patents expire, leading to potential price erosion for those specific branded treatments. For example, key patents for Qsymia are set to expire in the coming years, which will open the door for generic versions of this combination therapy.
What are the Regulatory Considerations for Phentermine?
Phentermine is classified as a Schedule IV controlled substance by the U.S. Drug Enforcement Administration (DEA) due to its potential for abuse and dependence. This classification imposes strict regulations on its prescribing, dispensing, and manufacturing.
Key Regulatory Aspects
- DEA Scheduling: As a Schedule IV drug, phentermine is subject to prescribing limits, refill restrictions, and specific record-keeping requirements for manufacturers, distributors, and pharmacies.
- FDA Approval and Monitoring: The U.S. Food and Drug Administration (FDA) approves phentermine for short-term (typically up to 12 weeks) adjunctive therapy in the management of obesity. The FDA continues to monitor its safety and efficacy.
- Prescribing Guidelines: Healthcare providers must adhere to established guidelines for patient selection, including screening for contraindications and monitoring for adverse events. Prescriptions are often limited in duration and quantity.
- International Regulations: Regulatory agencies in other countries, such as the European Medicines Agency (EMA), also have specific approval processes and post-market surveillance requirements for phentermine. Regulations can vary significantly by jurisdiction, impacting market access and prescribing practices.
- Labeling Requirements: Phentermine products carry specific warnings regarding potential cardiovascular effects, psychiatric symptoms, and the risk of abuse. These warnings are periodically updated by regulatory bodies based on new safety data.
- Manufacturing Standards: Pharmaceutical manufacturers must comply with Current Good Manufacturing Practices (cGMP) to ensure the quality, safety, and efficacy of phentermine products.
What are the Future Trends and Opportunities for Phentermine?
The future of phentermine in the weight management market will likely be shaped by evolving clinical practices, technological advancements, and competition from novel therapies.
Emerging Trends and Opportunities
- Personalized Medicine Approaches: Advances in pharmacogenomics could identify patient subgroups who are more likely to respond favorably to phentermine or experience fewer side effects, enabling more personalized treatment strategies.
- Digital Health Integration: The integration of phentermine prescriptions with digital health platforms, remote patient monitoring tools, and behavioral health support could enhance adherence and long-term outcomes. This could also facilitate closer monitoring for side effects and abuse potential.
- Novel Delivery Systems: While less likely given the drug's long history, research into extended-release formulations or alternative delivery mechanisms could potentially address adherence issues or improve the safety profile, although significant innovation here is constrained by the generic nature of the molecule.
- Continued Use in Combination Therapies: Phentermine's role as a component in approved combination therapies (like Qsymia) is expected to continue, offering a patented, more comprehensive weight management solution.
- Focus on Specific Patient Populations: Research may further delineate phentermine's efficacy and safety in specific patient populations, such as those with certain comorbidities or who have failed other weight-loss attempts.
- Market Expansion in Developing Economies: As healthcare access and awareness of obesity as a medical condition increase in developing regions, there is an opportunity for expanded market penetration of affordable treatments like phentermine.
Challenges and Threats
- Competition from Long-Acting Injectables: The success of long-acting GLP-1 receptor agonists for chronic weight management presents a significant competitive threat. These drugs offer different efficacy profiles and convenience for long-term use.
- Evolving Regulatory Scrutiny: Increased focus on the safety and long-term outcomes of weight-loss medications could lead to tighter prescribing regulations or a push for alternative treatments.
- Perception and Stigma: Phentermine, being a stimulant, may face some patient and physician hesitancy compared to newer, non-stimulant options, especially for long-term treatment.
Key Takeaways
- The global phentermine market is valued at approximately $1.5 billion, with projections indicating a 3-5% CAGR through 2030, driven by rising obesity rates and its established efficacy.
- North America is the largest market, while the Asia Pacific region offers the highest growth potential.
- The competitive landscape is dominated by generic manufacturers and branded combination products like Qsymia.
- Original phentermine patents have expired, leading to price competition, while newer combination therapies face their own patent expiration timelines.
- Regulatory oversight, including DEA scheduling as a Schedule IV controlled substance, significantly impacts phentermine's use and distribution.
- Future opportunities lie in personalized medicine, digital health integration, and continued use in combination therapies, alongside challenges from emerging long-acting injectable treatments.
Frequently Asked Questions
-
What is the typical duration of phentermine prescription? Phentermine is generally prescribed for short-term use, typically up to 12 weeks, as an adjunct to a comprehensive weight management program.
-
What are the main side effects associated with phentermine? Common side effects include dry mouth, insomnia, increased heart rate, elevated blood pressure, and nervousness. More serious side effects can include cardiovascular events and psychiatric disturbances.
-
Can phentermine be used for long-term weight management? While phentermine is effective for short-term weight loss, its long-term efficacy and safety profile are less established, and it is typically not recommended for extended continuous use due to the risk of dependence and cardiovascular issues.
-
How does phentermine compare to newer obesity medications like GLP-1 agonists? Phentermine is a stimulant that primarily works by suppressing appetite and increasing metabolism. GLP-1 agonists, such as semaglutide and tirzepatide, work through different mechanisms involving appetite regulation and glucose control, and are often approved for chronic weight management with demonstrated long-term efficacy and a different side effect profile.
-
Is phentermine available over-the-counter? No, phentermine is a prescription-only medication. It is classified as a Schedule IV controlled substance in the United States, requiring a prescription from a licensed healthcare provider due to its potential for abuse and dependence.
Citations
[1] World Health Organization. (n.d.). Obesity and overweight. Retrieved from https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight [2] U.S. Drug Enforcement Administration. (n.d.). Controlled substance schedules. Retrieved from https://www.dea.gov/controlled-substances-schedule [3] U.S. Food & Drug Administration. (n.d.). Medications Used for Weight Loss. Retrieved from https://www.fda.gov/consumers/consumer-updates/medications-used-weight-loss [4] Vivus Inc. (2023). Qsymia Prescribing Information. [Specific product label and prescribing information documents should be referenced if available from FDA or manufacturer's website]. [5] Eisai Inc. (2020). Eisai Announces Voluntary Withdrawal of BELVIQ® (lorcaserin HCl) from the U.S. Market. [Press Release]. Retrieved from https://www.eisai.com/news/2020/02/18_01.html
More… ↓
