Last updated: February 13, 2026
What Is the Market Position and Sales Projection for PHENERGAN?
Phenergan (promethazine) is an antihistamine primarily used for nausea, vomiting, allergies, and motion sickness. It has off-label uses and forms part of a broader phenothiazine class. The drug has been in market circulation for decades, with a well-established presence in the antiemetic and allergy segments.
Market Overview
Current Market Share and Usage
Phenergan's global sales are declining due to safety concerns related to its side effect profile, especially the risk of tissue necrosis when administered improperly. Its position has shifted as the following factors influence its market standing:
- Regulatory restrictions: Several agencies have classified promethazine as a drug with risks outweighing benefits, especially in pediatrics.
- Generics and competition: Many generic antiemetics (ondansetron, metoclopramide) have replaced Phenergan because of better safety profiles.
- Off-label use reduction: In the U.S., off-label uses have seen restrictions, impacting volume.
Market Size and Sales Data
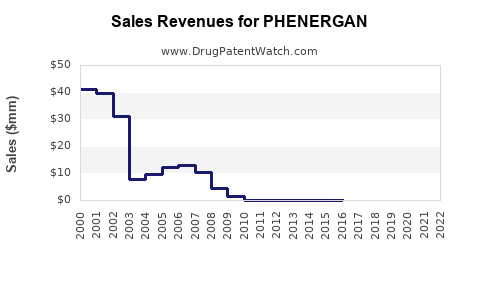
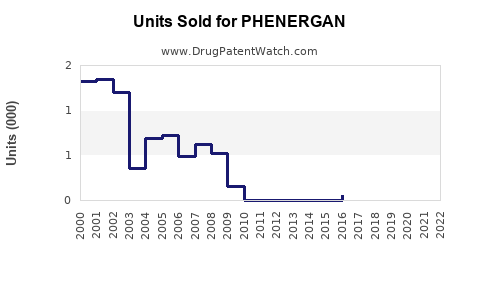
Estimations indicate global annual sales of Phenergan hover between $200 million and $400 million. The U.S. market accounts for approximately 70% of sales, with annual revenue around $140 million to $280 million (2021-2022 data).
- Market decline: Sales have decreased at a compound annual growth rate (CAGR) of about 2-4% over the past five years.
- Prescription trends: U.S. prescriptions declined from 15 million in 2017 to fewer than 10 million in 2022.
Regulatory Impact
In 2013, the FDA issued a boxed warning for promethazine, highlighting the risk of severe tissue injury. The European Medicines Agency classifies promethazine as a medication with risk of CNS depression and respiratory issues, leading to its restricted use.
Sales Projections
Short-term (Next 2-3 Years)
- Stabilization or slight decline: Due to ongoing safety concerns and regulatory restrictions, the sales are expected to stabilize or decline marginally.
- Estimated sales: Projected to remain within $180 million to $350 million globally annually.
- Market segments: Traditional hospital and outpatient prescribing will decline, but some niche markets (e.g., veterinary medicine, specific emergency protocols) may sustain minimal volumes.
Medium to Long-term (Next 5-10 Years)
- Further decline anticipated, driven by increasing safety awareness and new drugs supplanting promethazine.
- Innovations and generics: Limited pipeline or approved formulations likely will not reverse decline trends.
- Potential niches: In some regions, especially where alternatives are less accessible, sales may persist at low levels.
Analytical Assumptions
- Regulatory bodies maintain current restrictions.
- No major new formulations or safety improvements emerge.
- Competition from newer antiemetics remains strong, with ondansetron leading the market.
Competitive Landscape
| Drug Class |
Key Drugs |
Market Share (%) |
Regulatory Status |
Safety Profile |
| Phenothiazines |
Promethazine (Phenergan) |
20-25 |
Restricted, off-label decline |
Tissue necrosis, sedation, anticholinergic effects |
| 5-HT3 antagonists |
Ondansetron, Granisetron |
50-60 |
Widely approved, preferred choice |
Fewer severe local reactions |
| Dopamine antagonists |
Metoclopramide |
15-20 |
Restricted for long-term use |
Extrapyramidal symptoms |
Key Regulatory and R&D Trends
- The focus on safety has limited new development and labeling enhancements.
- US and EU authorities emphasize risk mitigation strategies.
- Patent expiration and generic availability limit investment in reformulation.
Conclusion
Phenergan's market is in decline due to safety concerns and competition, with sales stabilizing at lower levels. The forecast suggests continued erosion unless new formulations or indications are approved. Stakeholders should monitor regulatory developments and competitor innovations.
Key Takeaways
- Global sales of Phenergan range between $200 million and $400 million annually, predominantly in the U.S.
- Regulatory restrictions and safety concerns have driven sales decline over the past five years.
- Market projections indicate further decline unless new formulations or indications are introduced.
- Competition from ondansetron and other newer agents diminishes Phenergan’s market share.
- Future growth prospects are limited without significant regulatory or formulation changes.
FAQs
1. Why are prescribing trends for Phenergan declining?
Because of safety risks such as tissue necrosis and CNS depression, regulatory agencies restrict its use, especially in pediatric populations, leading to decreased prescriptions.
2. What are the main competitors to Phenergan?
Ondansetron and other 5-HT3 antagonists dominate the antiemetic market, offering better safety profiles.
3. Could new formulations revitalize Phenergan’s sales?
It is unlikely unless safety can be significantly improved or new therapeutic indications are approved.
4. How does regulatory stance vary globally?
The FDA has issued boxed warnings and restrictions, while European and other regulators follow similar cautionary principles but may differ in specific restrictions.
5. What is the future outlook for the drug?
Sales will likely continue declining with minimal recovery prospects in the next decade, barring breakthroughs in safety or indications.
Citations
- FDA. (2013). "Boxed Warning and Safety Labeling Changes for Promethazine."
- IQVIA. (2022). "Prescription Drug Sales Data."
- European Medicines Agency. (2021). "Promethazine Safety Review."
- Statista. (2022). "Anti-emetic Drugs Market Data."