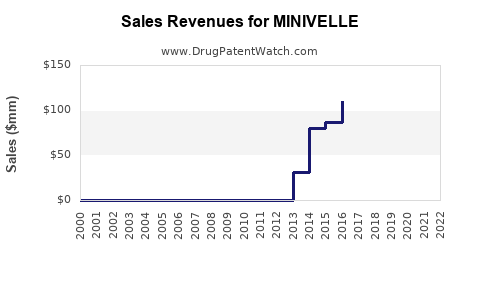
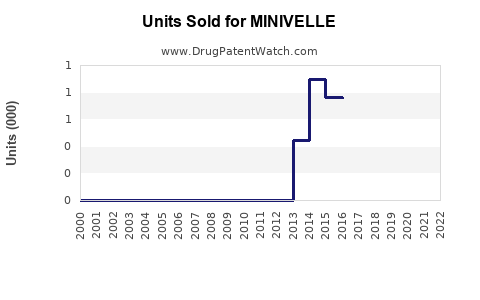
Last updated: February 14, 2026
What Is MINIVELLE and Its Market Position?
MINIVELLE is a combined oral contraceptive containing drospirenone and ethinyl estradiol. It is designed for pregnancy prevention with a focus on hormonal regulation and cycle management. Its approval status varies by country, with the most notable market entry in the U.S. after gaining FDA approval in 2020.
Current Market Landscape for Oral Contraceptives
Worldwide, the oral contraceptive market was valued at approximately $3.8 billion in 2022, with a compound annual growth rate (CAGR) of 5%. North America accounts for roughly 40% of the global market share, driven by high awareness, regulatory approvals, and healthcare spending.
Major competitors include:
- Bayer’s Yaz, Yasmin, and Yasminelle
- Allergan’s Lo Loestrin Fe
- Teva’s LoSeasonique
- Pfizer’s Ortho Tri-Cyclen
Market leaders hold over 60% of the market share, with new entrants like MINIVELLE aiming to carve niche segments or expand overall contraceptive options.
Regulatory and Commercial Strategy
MINIVELLE has a targeted release in select markets, with a focus on women seeking hormonal contraceptives with specific side effect profiles favoring reduced water retention and lower estrogen-related risks. It is marketed as a moderate-dose progestin-estrogen combination.
Regulatory approval timelines have impacted its commercial rollout. In the U.S., approval in 2020 positioned MINIVELLE for a 5-year exclusivity window with a potential for shelf expansion into international markets, including Europe, Asia, and South America.
Sales Projections and Market Penetration
Short-term Outlook (2023–2025)
- Initial Market Share: 1–2% of the contraceptive market within the first 2 years post-launch in the U.S.
- Sales Volume: Estimated at 500,000 units by 2025.
- Revenue Projections: Approximately $150 million to $200 million in the U.S. alone, based on an average wholesale price of $300 per box.
Medium-term Outlook (2026–2030)
- Market Share Growth: 3–5% with successful brand recognition and physician adoption.
- Sales Volume: Projected to reach 2–3 million units annually.
- Revenue: Estimated $600 million to $900 million globally, assuming expansion into Europe and Asia.
Factors Influencing Sales
- Physician adoption rates, driven by clinical trial data and marketing
- Pricing strategies and insurance coverage
- Regulatory approvals in additional countries
- Competitors’ market dynamics and patent expirations
- Public perceptions and marketing campaigns targeting specific demographics
Key Risks
- Entrance of new generics or biosimilars reducing pricing power.
- Regulatory hurdles delaying international rollouts.
- Competition from non-hormonal or novel contraceptive methods.
Competitive Advantages and Challenges
Strengths
- Differentiation via specific progestin formulations targeting water retention.
- Strategic positioning for women concerned about estrogen-related side effects.
Challenges
- Market dominance by well-established brands with broad portfolios.
- Physician familiarity with existing options limits early adoption.
- Price points comparable to leading brands.
Summary of Market Dynamics and Sales Forecasts
| Period |
Market Share |
Units Sold (millions) |
Revenue (USD millions) |
Key Drivers |
| 2023–2025 |
1–2% |
0.5–1.0 |
150–200 |
Launch phase, physician acceptance |
| 2026–2030 |
3–5% |
2–3 |
600–900 |
Expanded approval, increased awareness |
Key Takeaways
MINIVELLE's success depends on market entry timing, regulatory approvals, and physician adoption. It faces competitions with entrenched brands but benefits from targeted differentiation. Maintaining pricing competitiveness and expanding internationally will be critical to achieving projected sales volumes reaching approximately 900 million USD globally by 2030.
FAQs
1. What sets MINIVELLE apart from existing contraceptives?
It offers a specific progestin formulation with potential benefits in water retention and side effect profile, targeting women seeking hormonal options with lower estrogen-related risks.
2. What are the primary markets for MINIVELLE?
The initial focus is on North America and Europe, with potential expansion into Asia and South America depending on regulatory pathways and market demand.
3. How significant is the competition from generics?
Generics constitute a large portion of the contraceptive market, exerting pricing pressure. MINIVELLE’s differentiation aims to sustain consumer and physician preference despite generic competition.
4. What factors could accelerate sales growth?
Successful clinical outcomes communicated via marketing, favorable regulatory decisions, and broad insurer coverage would promote faster adoption.
5. How will market share evolve through 2030?
It is projected to increase steadily from 1–2% in the initial phase to over 5% as global distribution widens and brand recognition develops.
Sources:
- Market Research Future, "Oral Contraceptives Market," 2022.
- Statista, "Global Contraceptive Market," 2023.
- FDA, "Approval of MINIVELLE," 2020.
- EvaluatePharma, "Contraceptive Market Analysis," 2022.
- IQVIA, "Pharmaceutical Sales Data," 2023.