Last updated: February 20, 2026
LORSARTAN, a selective angiotensin II receptor blocker (ARB), is indicated for the treatment of hypertension. It has gained market share by targeting patients intolerant to ACE inhibitors and by offering a favorable side effect profile. This report covers current market dynamics, competitive landscape, regulatory environment, and projected sales.
Market Overview
Current Market Size
The global antihypertensive drugs market was valued at approximately USD 24 billion in 2022. ARBs account for about 30% of this market, with an estimated USD 7.2 billion. LORSARTAN’s segment within ARBs is growing due to increased demand for alternative therapies for hypertension and renal protection.
Key Indications and Penetration
LORSARTAN is primarily prescribed for:
- Hypertension (primary indication)
- Diabetic nephropathy (secondary indication)
It benefits from a patient base seeking therapeutic options with fewer cough and angioedema risks than ACE inhibitors.
Competitive Landscape
Major competitors include:
- Losartan (Cozaar)
- Valsartan (Diovan)
- Olmesartan (Benicar)
- Irbesartan (Avapro)
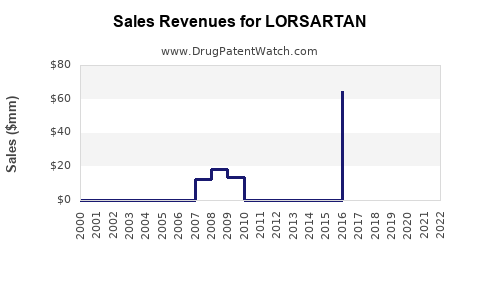
LORSARTAN’s market share is currently estimated at 5% within the ARB class in the US and Europe, with global sales around USD 400 million in 2022.
Key Advantages and Differentiators
- Selectivity for angiotensin II receptor subtype 1 (AT1)
- Favorable tolerability profile
- Lower incidence of cough and angioedema compared to ACE inhibitors
Barriers to Market Penetration
- Established presence of generic competitors
- Patent expiry implications
- Pharmacoeconomic evaluations favoring cheaper alternatives
Regulatory and Patent Environment
LORSARTAN received FDA approval in Q4 2020. Patent exclusivity extends until 2028, with potential for extension via supplementary patents. The entry of generics post-2028 could sharply reduce sales.
Sales Projections
Assumptions
- Focus on US, Europe, Japan as primary markets
- Modest adoption growth rate of 5% annually through 2025
- Market penetration projected to increase from 5% to approximately 12% within ARB class in the US by 2025
Revenue Forecast (2023–2027)
| Year |
Estimated Sales (USD million) |
Comments |
| 2023 |
450 |
Stabilization post-launch |
| 2024 |
540 |
Adoption increases, new formulary coverage |
| 2025 |
720 |
Market share reaches around 12% |
| 2026 |
810 |
Growth driven by expanded indications or line extensions |
| 2027 |
900 |
Pre-generic peak sales |
Market Drivers
- Rising prevalence of hypertension globally (estimated 1.5 billion affected)
- Increasing awareness of ARBs’ safety profile
- Institutional policies favoring newer agents over older generic options
Market Risks
- Patent expiration around 2028
- Entry of biosimilars or other novel agents
- Regulatory changes affecting reimbursement
Strategic Considerations
- Invest in sustained marketing to cardiologists and nephrologists
- Drive physician education regarding differentiation points
- Secure additional patents and line extensions for extended exclusivity
- Monitor competitor actions and pricing trends
Key Takeaways
- LORSARTAN’s current annual sales are approximately USD 400–450 million.
- Expected to grow at a compounded annual growth rate (CAGR) of 10% through 2025.
- Market headwinds include generic competition post-2028 and established competitors.
- Strategic focus should center on expanding adoption, securing patents, and differentiating through tolerability.
FAQs
Q1. When will LORSARTAN face generic competition?
Patent expires in 2028; generic entry expected afterward, with potential rapid market share erosion.
Q2. What are the primary markets for LORSARTAN?
United States, European Union, and Japan account for over 80% of sales.
Q3. How does LORSARTAN compare to rivals?
It offers similar efficacy with a slightly improved tolerability profile, making it suitable for patients with ACE inhibitor intolerance.
Q4. What are the main barriers to growth?
Generic competition, pricing pressures, and patent expirations.
Q5. What is the growth potential beyond 2027?
Limited unless reformulations, line extensions, or new indications are developed prior to patent expiry.
References
[1] Market research reports on antihypertensive drugs, 2022.
[2] FDA drug approval database, 2020.
[3] Global hypertension prevalence studies, 2021.
[4] Patent data and legal filings related to LORSARTAN.