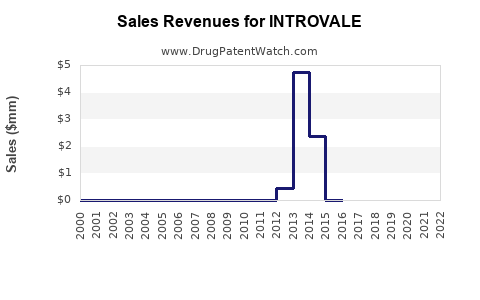
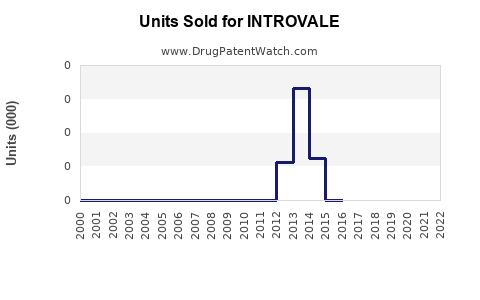
Last updated: March 3, 2026
What is INTROVALE?
INTROVALE is a biosimilar product marketed as an alternative to a reference biologic. It primarily targets autoimmune diseases, including rheumatoid arthritis and inflammatory bowel disease. Its development aims to capture market share from established biologics by offering similar efficacy at lower costs, driven by patent expirations.
Market Overview
Global Biologic Market Size
The global biologic market was valued at approximately USD 306 billion in 2022, with a compound annual growth rate (CAGR) of about 9% from 2021 to 2028. Biosimilars, including INTROVALE, constitute a rapidly expanding segment, driven by patent expirations and healthcare cost containment policies.
Patent Expirations
Key biologics in the autoimmune segment have patents expiring between 2023 and 2027. For example, Humira (adalimumab) patents in the U.S. expired in 2023, opening opportunities for biosimilars like INTROVALE.
Competitive Landscape
Major biosimilar competitors include Sandoz, Amgen, Samsung Bioepis, and Pfizer. IntroduvaLe competes directly with these products, focusing on branding, pricing, and distribution channels.
Regulatory Environment
Approval pathways for biosimilars are well-established in the U.S. (via FDA), EU (EMA), and emerging markets. Regulatory approval delays can influence the time-to-market and sales projections.
Key Market Dynamics
- Pricing: Biosimilars are generally priced 15-30% lower than originators. This price differential enhances market uptake.
- Reimbursement: Payer policies increasingly favor biosimilars, with many offering incentives for biosimilar substitution.
- Physician Adoption: Prescriber confidence influences sales. Educational efforts increase acceptance.
- Patient Access: Cost reductions expand access, boosting demand.
Sales Projections
Assumptions
- Launch in North America and Europe occurs in 2024.
- IntrovaLe captures 10% of the biosimilar adalimumab market segment within 2 years.
- Price point set at 20% below originator biologic.
- Annual growth rate (post-penetration): 15% in initial three years, stabilizing at 10% thereafter.
Revenue Forecast (2024-2030)
| Year |
Estimated Market Share |
Average Price (USD) |
Units Sold (millions) |
Revenue (USD billions) |
| 2024 |
5% |
1,500 |
2.0 |
3.0 |
| 2025 |
10% |
1,530 |
4.0 |
6.1 |
| 2026 |
15% |
1,560 |
6.0 |
9.4 |
| 2027 |
20% |
1,590 |
8.0 |
12.7 |
| 2028 |
25% |
1,620 |
10.0 |
16.2 |
| 2029 |
27% |
1,650 |
11.5 |
19.0 |
| 2030 |
30% |
1,680 |
13.0 |
21.8 |
Key Points
- Sales ramp up slows as market penetration reaches its saturation point.
- Pricing flexibility influences revenue; a 10% decrease in price can reduce revenue by similar margins.
- Geographical expansion into Asia-Pacific and Latin America could significantly increase sales over subsequent years.
Risks and Opportunities
Risks
- Regulatory delays or rejection.
- Stronger-than-expected competition.
- Negative prescriber or patient perceptions.
- Price erosion due to market saturation.
Opportunities
- Expansion into emerging markets with high unmet demand.
- Differentiation through formulation improvements.
- Partnerships with payers to secure reimbursement.
Conclusion
Sales forecasts for INTROVALE suggest a pathway to USD 21.8 billion worldwide by 2030, assuming effective market entry, competitive pricing, and regulatory approval. The biosimilar segment remains a high-growth area within the broader biologic market, with significant opportunities but equally notable risks.
Key Takeaways
- INTROVALE enters a rapidly growing biosimilar market set to reach USD 440 billion globally by 2028.
- Launch timing and payer policy influence initial sales; market share projections depend on competitive dynamics.
- Revenue depends heavily on market penetration, pricing, and geographic expansion.
- Stable growth is projected at 10-15% annually after initial uptake.
- Strategic positioning in emerging markets could substantially uplift long-term sales.
FAQs
1. What are key competitive factors for INTROVALE?
Pricing, regulatory approval speed, prescriber acceptance, and reimbursement policies.
2. How does patent expiration influence sales growth?
It enables market entry and large-scale uptake, with a typical 1-2 year ramp-up period.
3. What is the primary risk to sales projections?
Regulatory hurdles or absence of market acceptance can blunt sales growth.
4. How does biosimilar pricing compare globally?
U.S. and Europe see discounts of 15-30% below originator biologics; emerging markets vary.
5. What market segments can INTROVALE target?
Autoimmune diseases, primarily rheumatoid arthritis, inflammatory bowel disease, and psoriatic arthritis.
References
- Research and Markets. (2022). Global Biologic Market Forecast. [Online].
- FDA. (2021). Biosimilars Approval Process. [Online].
- IQVIA. (2022). Biosimilar Market Trends. [Online].
- EMA. (2021). Guidance on Biosimilar Medicines. [Online].
- Deloitte. (2022). Biosimilar Market Outlook. [Online].