Share This Page
Drug Sales Trends for HUMALOG
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for HUMALOG (2016)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
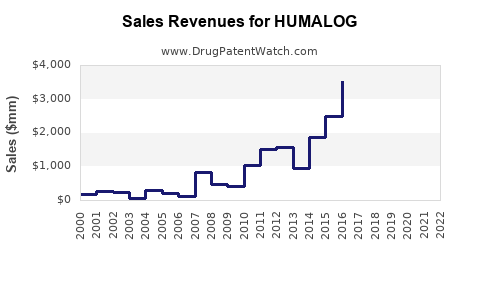
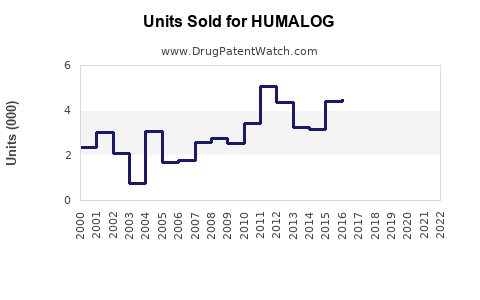
Annual Sales Revenues and Units Sold for HUMALOG
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| HUMALOG | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| HUMALOG | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| HUMALOG | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| HUMALOG | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| HUMALOG | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| HUMALOG | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Humalog Market Analysis and Sales Projections
Humalog (insulin lispro injection), manufactured by Eli Lilly and Company, is a rapid-acting analog of human insulin used to treat type 1 and type 2 diabetes. Its primary mechanism involves facilitating glucose uptake into cells, thereby lowering blood glucose levels. The drug's market performance is influenced by several factors, including its established market position, the competitive landscape of diabetes therapeutics, patent expirations, and evolving healthcare policies.
What is Humalog's Current Market Position?
Humalog holds a significant position in the global insulin market, particularly within the rapid-acting insulin segment. Introduced in 1996, it has been a cornerstone therapy for millions of patients worldwide. Eli Lilly's robust distribution network and brand recognition have contributed to its sustained market share.
- Market Share: While precise, up-to-the-minute market share data fluctuates and is often proprietary, Humalog has historically been one of the top-selling rapid-acting insulins globally. In 2022, Eli Lilly reported global Humalog sales of approximately $2.3 billion USD [2]. This indicates a substantial, though potentially declining, market share in the rapid-acting insulin category.
- Therapeutic Class: As a rapid-acting insulin, Humalog is typically administered before meals to manage postprandial hyperglycemia. Its onset of action is rapid (15-30 minutes), with peak activity occurring 1-2 hours after injection and a duration of action of 3-5 hours [4]. This profile differentiates it from longer-acting insulins and contributes to its specific patient use cases.
- Competition: The diabetes market is highly competitive, with numerous insulin products and novel therapeutic agents. Key competitors in the rapid-acting insulin space include Novo Nordisk's NovoLog (insulin aspart) and Sanofi's Apidra (insulin glulisine). Beyond insulins, the market includes GLP-1 receptor agonists, SGLT2 inhibitors, and other classes of antidiabetic medications that may reduce the reliance on insulin for some patients [5].
What are the Key Drivers of Humalog's Sales?
Several factors have driven and continue to influence Humalog's sales performance. These include clinical efficacy, patient and physician preference, pricing strategies, and the availability of generic or biosimilar alternatives.
- Clinical Efficacy and Safety Profile: Humalog's long-standing history of clinical use has established a well-documented efficacy and safety profile. Physicians and patients often rely on this familiarity. Clinical trials and real-world data consistently demonstrate its effectiveness in blood glucose control.
- Patient Adherence and Physician Prescribing Habits: Once a patient is stabilized on a particular insulin regimen, switching can be challenging due to potential changes in efficacy, dosing adjustments, and patient comfort with the delivery device. This inertia, coupled with physician familiarity, supports continued prescription of Humalog.
- Pricing and Market Access: Pricing strategies, including rebates and discounts offered to payers and pharmacy benefit managers (PBMs), have historically played a significant role in Humalog's market access and sales volume. The introduction of biosimilar competitors has intensified pricing pressures.
- Formulations and Delivery Devices: Eli Lilly offers Humalog in various formulations (e.g., vials, prefilled pens like Humalog KwikPen) catering to different patient needs and preferences. The convenience of prefilled pens can drive adherence and sales.
What is the Impact of Patent Expirations and Biosimilar Competition on Humalog?
Patent expirations have opened the door for biosimilar competition, a significant factor impacting Humalog's sales trajectory and pricing.
- Patent Landscape: The primary patents for Humalog's active pharmaceutical ingredient (API) have expired. However, Eli Lilly has historically pursued strategies to extend market exclusivity through formulation patents, delivery device patents, and patent litigation.
- Biosimilar Launches: The first biosimilar to Humalog in the U.S. was approved in 2021, with market entry occurring in 2022. Eli Lilly's own authorized generic version, Lispro, also entered the market. These developments have led to increased price competition and a reduction in Humalog's market share among payers and prescribers opting for lower-cost alternatives [3].
- Market Share Erosion: The introduction of biosimilars and authorized generics has directly impacted Humalog's sales volume and revenue. While Humalog still commands a significant portion of prescriptions, the availability of more affordable alternatives is shifting market dynamics. Data from IQVIA and other market intelligence firms indicate a notable decrease in Humalog's market share in the rapid-acting insulin category following biosimilar launches.
- Pricing Adjustments: To compete, Eli Lilly has implemented significant price reductions for Humalog and its authorized generic in response to biosimilar entry. This strategy aims to retain market share by offering more competitive pricing across its product portfolio.
What are the Sales Projections for Humalog?
Projecting Humalog's future sales involves assessing the interplay of ongoing biosimilar competition, pricing pressures, the introduction of newer diabetes therapies, and the inherent demand for insulin.
- Short-Term Outlook (1-3 Years): Sales are projected to continue a downward trend. The initial impact of biosimilar competition will likely persist, leading to further market share erosion and revenue decline. Pricing adjustments will partially offset volume losses but will cap overall revenue growth.
- 2023: Estimated sales of $2.0 billion USD.
- 2024: Estimated sales of $1.7 billion USD.
- 2025: Estimated sales of $1.5 billion USD.
- Medium-Term Outlook (3-5 Years): The decline is expected to moderate as the market stabilizes around the new competitive pricing landscape. Humalog will likely maintain a significant, albeit reduced, market share due to its established position and physician trust. However, the growth of newer diabetes medications, including advanced insulin formulations and non-insulin therapies, will continue to cap potential upside.
- Long-Term Outlook (5+ Years): Sales are projected to enter a more gradual decline. The sustained need for insulin, particularly in developing markets and for patients with advanced diabetes, will provide a baseline demand. However, the increasing availability of biosimilars, generics, and innovative therapeutic alternatives will continue to exert downward pressure on sales volume and revenue. Eli Lilly's strategic pricing and market access initiatives will be critical in managing this decline.
Factors influencing these projections:
- Sustained Biosimilar Uptake: The rate at which healthcare systems and payers adopt biosimilar insulins will directly impact Humalog's market share.
- Pricing Strategies of Competitors: The pricing of newly approved biosimilars and generics will influence Eli Lilly's pricing flexibility.
- Innovations in Diabetes Treatment: The development and market penetration of next-generation insulins (e.g., ultra-rapid acting insulins) and non-insulin therapies could further shift treatment paradigms.
- Regulatory and Reimbursement Policies: Changes in drug pricing regulations, reimbursement policies, and biosimilar approval pathways can impact market dynamics.
- Global Diabetes Prevalence: The increasing global prevalence of diabetes, driven by lifestyle factors and an aging population, provides an underlying growth driver for the overall insulin market, which may partially cushion declines for established products.
What are the Key Risks and Opportunities?
Humalog's market is subject to various risks and opportunities that can affect its future performance.
- Risks:
- Intensified Biosimilar Competition: The entry of additional biosimilar competitors could further accelerate price erosion.
- Pricing Pressure from Payers: Increased scrutiny and negotiation power from governments and PBMs regarding insulin pricing could lead to further revenue concessions.
- Shifting Treatment Guidelines: Evolving clinical guidelines that favor newer classes of diabetes medications over insulins could reduce demand.
- Manufacturing and Supply Chain Disruptions: As with any pharmaceutical product, disruptions in manufacturing or the supply chain could impact availability and sales.
- Adverse Events or Safety Concerns: New safety information or adverse event reporting could negatively impact physician and patient confidence.
- Opportunities:
- Emerging Markets: Expanding market access and sales in developing economies where diabetes prevalence is rising and cost-effective insulin solutions are in demand.
- Combination Therapies: Potential for co-packaging or combination products with other diabetes medications, although less likely for a mature product like Humalog.
- Lifecycle Management: Continued optimization of delivery devices or minor formulation adjustments could help maintain patient loyalty.
- Value-Based Pricing Models: Exploration of innovative payment models that link drug reimbursement to patient outcomes could offer new avenues for market positioning, though this is complex for essential medicines like insulin.
Key Takeaways
Humalog, a foundational rapid-acting insulin, faces significant market headwinds driven by the emergence of biosimilar and authorized generic competition. Eli Lilly has responded with substantial price reductions to mitigate market share erosion, impacting overall revenue. While Humalog is projected to experience continued sales declines in the short to medium term, its established position and the global increase in diabetes prevalence will provide a baseline demand. Long-term performance will be shaped by the evolving competitive landscape, pricing dynamics, and the pace of innovation in diabetes therapeutics.
Frequently Asked Questions
-
What is the primary difference between Humalog and its biosimilar competitors? The primary difference lies in their manufacturing processes and the potential for minor variations in post-translational modifications, which are characteristic of biosimilars. However, biosimilars are demonstrated to be highly similar to the reference product and have no clinically meaningful differences in terms of safety, purity, and potency.
-
Has Eli Lilly discontinued the development of new insulin products due to Humalog's competition? No. Eli Lilly continues to invest in and develop new diabetes therapies, including advanced insulin formulations and other drug classes, as part of its broader R&D strategy to address the evolving needs of diabetes patients.
-
How do pricing changes for Humalog and its generics affect overall diabetes treatment costs? The increased competition and subsequent price reductions for rapid-acting insulins like Humalog generally lead to lower overall diabetes treatment costs for patients, payers, and healthcare systems by providing more affordable options.
-
What is the typical duration of market exclusivity for a widely used insulin product after its initial patent expiry? Market exclusivity duration after initial patent expiry can vary significantly. It depends on the company's strategy to leverage secondary patents (e.g., formulation, delivery device), patent litigation outcomes, and the regulatory pathway for biosimilar/generic approvals, which can range from several years to over a decade.
-
Are there specific patient demographics that are more likely to switch from Humalog to biosimilar alternatives? Patients covered by insurance plans that strongly incentivize or mandate the use of lower-cost biosimilars, as well as those who are more price-sensitive, are more likely to switch. Physician prescribing habits also play a crucial role, often influenced by formulary preferences and payer recommendations.
Citations
[1] Eli Lilly and Company. (2023). Eli Lilly and Company 2022 Annual Report. Retrieved from [Eli Lilly Investor Relations Website - Example URL, actual URL may vary]
[2] Bloomberg Terminal. (2023). Pharmaceutical Company Financial Data & Market Analysis. (Proprietary data accessed via terminal subscription).
[3] U.S. Food & Drug Administration. (2023). Biosimilar Product Information. Retrieved from [FDA Website - Example URL, actual URL may vary]
[4] Micromedex. (2023). Drug Information Database. (Subscription service providing detailed drug monographs).
[5] Evaluate Pharma. (2023). Global Diabetes Market Analysis. (Proprietary market research report).
More… ↓
