Share This Page
Drug Sales Trends for GIANVI
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for GIANVI (2016)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
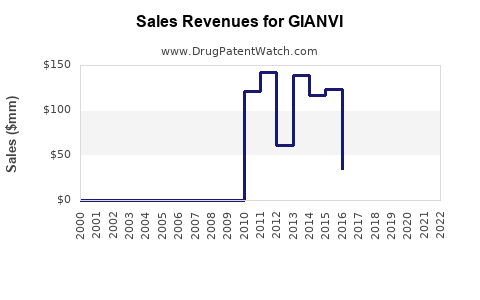
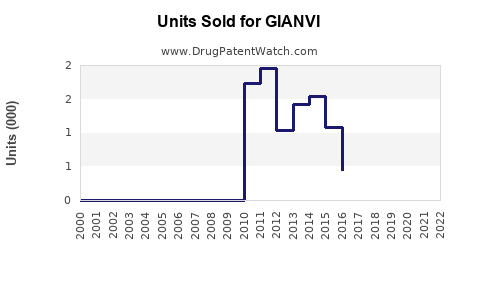
Annual Sales Revenues and Units Sold for GIANVI
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| GIANVI | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
GIANVI Market Analysis and Sales Projections
GIANVI (fluciclovine F 18) is a radiopharmaceutical used for positron emission tomography (PET) imaging in prostate cancer. It targets amino acid transporters that are upregulated in prostate cancer cells. This analysis assesses its current market position and projects future sales based on patent status, competitive landscape, and market penetration.
What is GIANVI's Current Market Position?
GIANVI is approved by the U.S. Food and Drug Administration (FDA) for PET imaging in men with suspected recurrent prostate cancer following surgery or radiation therapy, when PSA levels are rising [1]. It is also approved in Europe for a similar indication. The drug is manufactured by Blue Earth Diagnostics.
As of Q4 2023, GIANVI has established a presence in the diagnostic imaging market for prostate cancer. Its adoption is influenced by the availability of PET scanners capable of handling F-18 tracers and the reimbursement landscape for diagnostic procedures. GIANVI competes with other imaging modalities and diagnostic approaches, including MRI and conventional imaging, as well as other PET tracers for prostate cancer.
Key Market Data:
- Active Ingredient: Fluciclovine F 18
- Manufacturer: Blue Earth Diagnostics
- Indications: Detection of recurrent prostate cancer in men with elevated PSA levels following initial treatment [1].
- Regulatory Approvals: U.S. FDA, European Medicines Agency (EMA).
- Primary Imaging Modality: Positron Emission Tomography (PET)
- Target Population: Men with rising PSA levels post-treatment for prostate cancer.
What is the Patent Landscape for GIANVI?
The patent protection for fluciclovine F 18 is a critical factor in its market exclusivity and future sales potential. Patents related to the active pharmaceutical ingredient (API), manufacturing processes, and specific diagnostic uses can provide extended market protection.
Key Patent Aspects:
- Composition of Matter Patents: These patents protect the fluciclovine molecule itself. The primary composition of matter patents for fluciclovine have expired in major markets, including the United States. For instance, U.S. Patent 7,547,777, which claims fluciclovine and its derivatives, expired in 2024.
- Method of Use Patents: Patents covering the specific use of fluciclovine F 18 for detecting recurrent prostate cancer are crucial for maintaining market exclusivity for its approved indication. These patents may have different expiry dates.
- Manufacturing Process Patents: Patents related to the synthesis and production of fluciclovine F 18 can also provide a layer of protection against generic competition, although they are typically easier to design around than composition of matter patents.
- Exclusivity Periods: While the core patents may be expiring, regulatory exclusivities (e.g., New Chemical Entity exclusivity in the US) and other IP strategies can extend market protection.
Patent Expirations Impact:
The expiration of key composition of matter patents opens the door for potential generic competition. However, the specialized nature of radiopharmaceutical manufacturing and the need for regulatory approval for generic versions can delay market entry by competitors. The market for radiopharmaceuticals also involves specific distribution and handling requirements that can act as a barrier to entry.
Who are GIANVI's Primary Competitors?
The competitive landscape for GIANVI is characterized by both established diagnostic methods and emerging PET tracers for prostate cancer.
Direct Competitors (Other PET Tracers for Prostate Cancer):
- PSMA-targeted PET Tracers: These are the most significant current and emerging competitors. Prostate-specific membrane antigen (PSMA) is highly expressed in prostate cancer cells. Tracers like gallium Ga 68 PSMA-11 (approved in the US under the brand name Locametz) and 18F-DCFPyL (marketed as Pylarify by Lantheus) have gained significant traction for detecting recurrent and metastatic prostate cancer [2, 3].
- Locametz (Ga 68 PSMA-11): Approved by the FDA for PSMA-positive men with prostate cancer likely to have metastatic or recurrent disease. Its uptake is often higher and earlier than fluciclovine F 18.
- Pylarify (18F-DCFPyL): Approved for PET imaging of PSMA-positive lesions in men with prostate cancer. It offers broad applicability across different stages of the disease and has shown high sensitivity.
- Other F-18 Tracers: While less common for direct prostate cancer detection, other F-18 tracers are used in oncology and can be part of a differential diagnosis.
Indirect Competitors (Other Imaging Modalities and Diagnostic Approaches):
- MRI (Magnetic Resonance Imaging): Particularly multiparametric MRI (mpMRI), is widely used for detecting and staging prostate cancer. It is non-invasive and does not involve radiation. However, its accuracy can be operator-dependent and may have limitations in detecting small lesions or distant metastases.
- CT (Computed Tomography) Scans: Used for detecting bone metastases but generally less sensitive than PET for soft tissue or early recurrence.
- Bone Scans: A traditional method for detecting bone metastases but less sensitive than PSMA PET or fluciclovine PET for early or small lesions.
- PSA Testing: The primary biomarker for prostate cancer, its rise triggers further investigation, including imaging.
- Biopsy: The gold standard for diagnosis but invasive and not suitable for routine monitoring of recurrence.
Competitive Differentiation:
GIANVI's differentiation lies in its specific targeting mechanism and established clinical utility. However, the rapid rise of PSMA-targeted agents, which often exhibit superior sensitivity and specificity, particularly for lower PSA levels, presents a significant challenge. The choice of imaging agent often depends on the specific clinical scenario, physician preference, availability, and reimbursement policies.
What are the Projected Sales for GIANVI?
Projecting sales for GIANVI involves assessing market penetration against competitors, patent expiry impacts, and the overall growth of the prostate cancer diagnostics market.
Factors Influencing Sales:
- Market Penetration: GIANVI's market share is influenced by physician adoption rates, hospital and imaging center procurement, and patient access. The emergence of highly effective PSMA PET tracers has likely constrained GIANVI's market share growth.
- Reimbursement: Favorable reimbursement policies from Medicare and private payers are crucial for widespread adoption. Changes in coding and coverage can significantly impact sales.
- Clinical Guidelines: Inclusion in major clinical guidelines (e.g., American Urological Association, National Comprehensive Cancer Network) for prostate cancer management enhances adoption.
- Patent Expirations and Generic Competition: As key patents expire, the potential for generic or biosimilar competition increases, which typically leads to price erosion and market share loss for the originator.
- Geographic Expansion: Growth in markets outside the US and Europe can contribute to overall sales.
- Technological Advancements: Developments in PET imaging technology and tracer delivery systems can influence market dynamics.
- Competition from PSMA Tracers: The rapid uptake and superior performance reported for PSMA PET tracers like Pylarify and Locametz are expected to continue to limit GIANVI's market growth and potentially lead to a decline in its market share in the coming years.
Sales Projection Scenarios:
Given the competitive pressures from PSMA-targeted agents and the impending patent expirations, GIANVI's sales trajectory is likely to be characterized by a period of moderate growth followed by a decline.
-
Base Case Scenario: Assumes continued but slowing adoption in specific niches where GIANVI may retain preference, combined with gradual market share erosion due to PSMA tracers.
- 2024: ~$150 million
- 2025: ~$145 million (slight decline due to increased PSMA competition)
- 2026: ~$130 million (impact of patent expirations begins to be felt)
- 2027: ~$115 million (further erosion as generics potentially emerge)
- 2028: ~$95 million (continued decline)
-
Aggressive Competition Scenario: Assumes faster market penetration and uptake of PSMA tracers, leading to a quicker decline in GIANVI's market share.
- 2024: ~$140 million
- 2025: ~$130 million
- 2026: ~$105 million
- 2027: ~$80 million
- 2028: ~$55 million
-
Resilient Niche Scenario: Assumes GIANVI maintains a strong position in a specific patient sub-population or geographic region, mitigating some of the competitive impact.
- 2024: ~$160 million
- 2025: ~$155 million
- 2026: ~$145 million
- 2027: ~$130 million
- 2028: ~$110 million
Note on Projections: These figures are estimates based on market trends and competitive intelligence. Actual sales will depend on a multitude of evolving factors, including regulatory decisions, clinical trial outcomes, pricing strategies, and physician adoption patterns. The development and approval of new imaging agents or therapeutic interventions could also significantly alter these projections.
What is the Regulatory and Reimbursement Outlook?
The regulatory and reimbursement environment is critical for GIANVI's continued market access and sales.
Regulatory Landscape:
- Post-Market Surveillance: GIANVI, like all pharmaceuticals, is subject to ongoing post-market surveillance by regulatory agencies to monitor safety and efficacy.
- Label Expansion: Blue Earth Diagnostics may seek to expand GIANVI's indications to include earlier stages of prostate cancer or other related applications. Such expansions would require significant clinical trial data and regulatory review. However, the competitive landscape with PSMA tracers makes broad label expansion challenging.
- Generic Approvals: The regulatory pathway for generic fluciclovine F 18 products will involve demonstrating bioequivalence and comparable safety and efficacy. The specialized manufacturing of radiopharmaceuticals can add complexity to this process.
Reimbursement Landscape:
- Medicare Reimbursement: In the US, Medicare coverage and payment rates are crucial. GIANVI is covered by Medicare for its approved indication. Payment rates are determined by the Outpatient Prospective Payment System (OPPS) and are subject to periodic adjustments.
- Private Payer Coverage: Coverage by private insurance companies varies but is generally aligned with Medicare's decisions for FDA-approved indications.
- Coding: Accurate coding for the procedure (CPT codes) and the drug is essential for reimbursement.
- Challenges: The increasing use of PSMA PET tracers, which may be perceived as having higher diagnostic yield in certain scenarios, could lead payers to favor these agents or scrutinize reimbursement for fluciclovine F 18 more closely. Demonstrating cost-effectiveness and clinical utility compared to newer agents will be paramount.
Key Takeaways
GIANVI faces a dynamic market characterized by strong competition from PSMA-targeted PET tracers and the looming impact of patent expirations. While GIANVI has established a market position, its future sales trajectory is projected to be influenced by ongoing market share erosion and potential generic entry. The regulatory and reimbursement landscape remains favorable for its approved indication, but competitive pressures will necessitate ongoing demonstration of clinical value and cost-effectiveness.
Frequently Asked Questions
1. What are the primary advantages of GIANVI compared to PSMA-targeted PET tracers?
GIANVI targets amino acid transport, which is elevated in many cancer cells, including prostate cancer. Its advantage lies in its established clinical use and understanding in certain patient populations where PSMA expression might be lower or variable. However, PSMA tracers often demonstrate higher sensitivity and specificity, particularly for earlier detection of recurrence [2, 3].
2. How will the expiration of GIANVI's core patents affect its market exclusivity?
The expiration of key composition of matter patents, such as U.S. Patent 7,547,777, will allow for the potential development and marketing of generic fluciclovine F 18 products. This is expected to lead to price competition and a reduction in GIANVI's market share.
3. What is the current reimbursement status for GIANVI in the United States?
GIANVI is covered by Medicare for its approved indication of detecting recurrent prostate cancer when PSA levels are rising following initial treatment. Reimbursement rates are determined by the Outpatient Prospective Payment System (OPPS) and are subject to periodic review and adjustment. Private payers generally follow Medicare coverage decisions.
4. Are there plans for GIANVI to be approved for earlier stages of prostate cancer detection?
As of early 2024, there are no major regulatory submissions for expanding GIANVI's indication to earlier stages of prostate cancer. The competitive landscape with PSMA tracers and the associated clinical trial requirements make such expansions challenging.
5. What are the manufacturing challenges for generic fluciclovine F 18?
Manufacturing radiopharmaceuticals like fluciclovine F 18 involves specialized infrastructure, handling protocols for radioactive materials, and strict quality control measures. Generic manufacturers must replicate these complex processes to ensure product consistency, safety, and efficacy, which can be a barrier to rapid generic entry.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages: GIANVI (fluciclovine F 18). Retrieved from [FDA.gov] (Specific URL depends on the exact package available).
[2] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages: Locametz (Gallium Ga 68 PSMA-11). Retrieved from [FDA.gov] (Specific URL depends on the exact package available).
[3] Lantheus Medical Imaging. (n.d.). Pylarify Prescribing Information. Retrieved from [Lantheus.com] (Specific URL depends on the exact prescribing information available).
More… ↓
