Last updated: February 20, 2026
What is the Current Market Size for ENOXAPARIN?
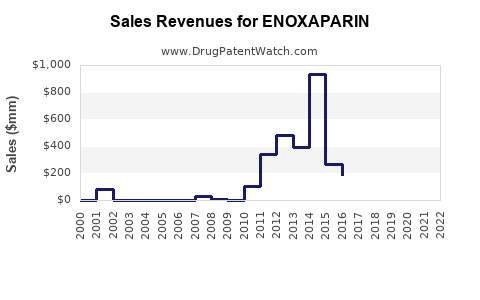
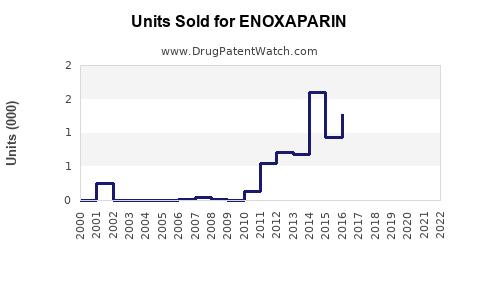
Enoxaparin, a low molecular weight heparin (LMWH), generated global sales of approximately USD 4.2 billion in 2022. Its primary markets include the United States, Europe, China, and other Asia-Pacific regions. The drug's key indications are deep vein thrombosis (DVT), pulmonary embolism (PE), and prophylaxis in surgical patients. Sales are driven by the prevalence of thromboembolic conditions and the expanding adoption of anticoagulant therapies.
Average annual growth rate (CAGR) over the past five years is approximately 7.2%. This growth reflects increased awareness, broader clinical use, and recent patent exclusivities in major markets.
| Region |
2022 Sales (USD Billion) |
CAGR (2018–2022) |
Key Drivers |
| North America |
2.1 |
6.8% |
High prevalence of cardiovascular diseases; strong adoption |
| Europe |
1.2 |
7.0% |
Established use in hospital settings |
| China & Asia-Pacific |
0.6 |
8.4% |
Growing healthcare infrastructure, increasing thromboembolism cases |
| Rest of World |
0.3 |
6.5% |
Rising awareness, expanding reimbursement policies |
What Are the Emerging Market Dynamics?
Market Drivers
- Growing incidence of venous thromboembolism (VTE)
- Increased use for prophylaxis in orthopedic and cardiac surgeries
- Expanded approval for new indications and populations
- Shift from unfractionated heparin to LMWH for outpatient management
Market Restraints
- Competition from direct oral anticoagulants (DOACs) such as rivaroxaban, apixaban
- Price sensitivity in emerging markets
- Regulatory challenges concerning biosimilars and manufacturing standards
Competitive Landscape
Major players include Sanofi (Lovenox), Pfizer (Fragmin), and domestic manufacturers in China and India. Sanofi maintains leading market share, with over 60%, benefiting from established supply chains and regulatory approvals.
How Are Sales Projections Shaping Up?
Short-term Outlook (2023–2025)
Sales are projected to grow at a CAGR of 5.5%. Market expansion is primarily driven by increased usage in hospital settings and prophylaxis in orthopedics. Patent protections and manufacturing agreements prolong revenue streams for key players.
Medium to Long-term Outlook (2026–2030)
Projected CAGR drops to approximately 4.2%. Growth depends on:
- Introduction and acceptance of biosimilars
- Regulatory approvals for new indications, such as treatment of acute coronary syndrome
- Competition from DOACs, which are orally administered and often preferred for outpatient use
Sales Projections Table (USD Billions)
| Year |
Sales Projection |
Growth Rate |
| 2023 |
4.45 |
5.5% |
| 2024 |
4.70 |
5.6% |
| 2025 |
4.97 |
5.8% |
| 2026 |
5.20 |
4.6% |
| 2027 |
5.44 |
4.6% |
| 2028 |
5.65 |
4.0% |
| 2029 |
5.87 |
3.8% |
| 2030 |
6.07 |
3.4% |
Key Assumptions
- Continued patent exclusivity in the US until 2025; biosimilar entry expected post-2025.
- Regulatory approvals expanding indications in Asia and Europe.
- Competitive pricing pressure from biosimilars and generic LMWHs.
What Are Critical Factors for Market Growth?
- Regulatory Approvals: Faster approval processes for biosimilars and new indications can boost sales.
- Pricing & Reimbursement: Favorable reimbursement policies, especially in emerging markets, enhance market penetration.
- Clinical Guidelines: Adoption of ENOXAPARIN in updated treatment protocols influences prescribing behavior.
- Technological Advancements: Improved manufacturing and biosimilar development reduce costs and enable broader access.
What Is the Impact of Biosimilars and Competition?
Biosimilars for ENOXAPARIN are in various stages of development. Cost savings from biosimilar entry can reduce therapy costs by approximately 20–30%. However, Sanofi and Pfizer retain significant margins for their branded products, which influence market share during biosimilar launches.
Summary of Key Market Opportunities
- Increased use in postoperative DVT/PE prevention
- Growing outpatient anticoagulation management
- Adoption in developing countries with expanding healthcare infrastructure
- Entry of biosimilars creating price competition
Key Takeaways
- The global enoxaparin market reached USD 4.2 billion in 2022, with steady growth driven by thromboembolic disease prevalence.
- Sales are projected to grow annually by approximately 5.5% through 2025, slowing thereafter.
- Market expansion depends on regulatory approvals, biosimilar competition, and clinical practice shifts.
- Competition from DOACs remains a key factor, particularly for outpatient use.
- Emerging markets represent significant growth opportunity, with increasing healthcare investment.
FAQs
1. How does biosimilar development affect enoxaparin revenues?
Biosimilars are expected to enter the market post-2025, potentially reducing prices by 20–30% and eroding margins for original manufacturers.
2. What are the key regulatory hurdles for enoxaparin in new markets?
regulators require comprehensive biosimilar comparability data, manufacturing standards adherence, and clinical trial evidence demonstrating equivalence to the originator.
3. How does competition from DOACs influence enoxaparin's market share?
DOACs offer oral administration and simplified dosing, making them more attractive for outpatient use, which restricts enoxaparin primarily to hospital or inpatient settings.
4. What indications are driving future sales growth?
New indications for treating acute coronary syndromes and expanded prophylaxis in orthopedic surgeries contribute to sales expansion.
5. What is the outlook for enoxaparin in emerging markets?
Rapid healthcare infrastructure development and increasing thromboembolism awareness support robust growth opportunities despite price sensitivity.
References
[1] GlobalData. (2023). Enoxaparin market analysis.
[2] EvaluatePharma. (2022). 2022 Worldwide Sales Data.
[3] United States Food and Drug Administration. (2022). Biosimilar guidelines.
[4] European Medicines Agency. (2022). ENOXAPARIN approvals.
[5] IMS Health. (2022). Market trends in anticoagulants.