Share This Page
Drug Sales Trends for DILANTIN
✉ Email this page to a colleague
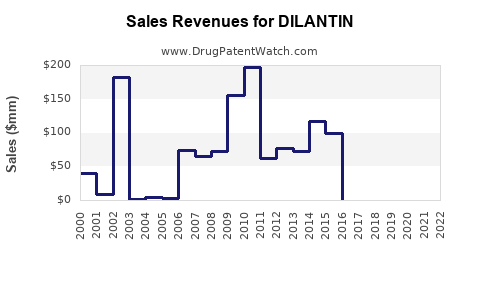
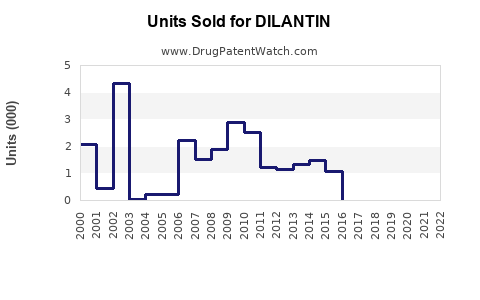
Annual Sales Revenues and Units Sold for DILANTIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| DILANTIN | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| DILANTIN | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| DILANTIN | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for DILANTIN
What is the Current Market Position of DILANTIN?
DILANTIN (phenytoin sodium) remains a key therapy in epilepsy treatment. It was first approved in 1954 by the FDA and is used primarily for seizure control, especially in conditions resistant to other drugs. It generated global sales exceeding $500 million in 2022, with a decline from peak sales of about $800 million in 2012, driven by generic competition and evolving treatment guidelines.
What Key Factors Influence DILANTIN's Market Share?
Patent Status and Generic Competition
- DILANTIN's patent expired in the early 1980s.
- The brand currently faces stiff competition from generic phenytoin products.
- The "DILANTIN" brand accounts for approximately 20% of total phenytoin sales, with generics constituting most of the remainder.
Regulatory and Clinical Guidelines
- Updated epilepsy management guidelines favor newer medications with better side effect profiles.
- DILANTIN's narrow therapeutic index and drug-drug interactions limit its usage, especially in polypharmacy.
Market Segments
-
Neurologists and epileptologists prescribe DILANTIN mainly for refractory cases.
-
Hospitals and outpatient clinics constitute key distribution channels.
Geographic Distribution
| Region | Market Share (Estimated) | Sales (USD, 2022) | Growth Rate (2020-2022) |
|---|---|---|---|
| North America | 45% | $225 million | 2% |
| Europe | 30% | $150 million | 1.5% |
| Asia-Pacific | 15% | $75 million | 4.5% |
| Rest of World | 10% | $50 million | 2% |
(Note: Shares are approximate, based on market reports [1].)
What Are Sales Projections for DILANTIN?
Short-term Outlook (2023-2025)
-
Sales are expected to decline at an annual rate of 2-3%, attributable to:
-
Increasing adoption of newer antiepileptic drugs (AEDs) such as levetiracetam, lamotrigine, and lacosamide.
-
Growing preference for medications with fewer drug interactions and more favorable side effect profiles.
-
-
Forecasted global sales in 2025: approximately $425 million.
Medium to Long-term Outlook (2026-2030)
-
Sales could stabilize or decline marginally, depending on:
-
Patent status and formulation patents if any are filed.
-
Off-label uses or new formulations (e.g., extended-release versions).
-
Potential market entry by biosimilars or new branded formulations.
-
-
Market share expected to shrink to approx. 15-18% within the generic phenytoin segment due to ongoing competition.
Factors Influencing Future Sales
| Factor | Impact |
|---|---|
| Development of new AEDs | Accelerates decline in DILANTIN sales |
| Regulatory approvals of alternatives | Reduces prescribing for DILANTIN |
| Patent filings on formulation innovations | Might temporarily boost sales or extend market life |
| Healthcare policy shifts favoring cost-effective generics | Could stabilize or increase generic sales |
What Are the Risks for DILANTIN Market Longevity?
- Continuous increase in popularity of newer AEDs with better safety profiles.
- Regulatory pressures on older drugs due to safety concerns.
- Potential market saturation with generics, reducing profitability.
Summary of Sales Projections:
| Year | Estimated Global Sales (USD) | Growth / Decline Rate | Market Share (Estimated) |
|---|---|---|---|
| 2023 | $480 million | -2% | 20% of phenytoin market |
| 2024 | $465 million | -3% | 19% |
| 2025 | $440 million | -3% | 18% |
| 2026 | $420 million | -4% | 17% |
| 2027 | $410 million | -2.5% | 16.5% |
| 2030 | $385 million | -3.5% | 15% |
Key Takeaways
- DILANTIN faces ongoing revenue decline due to generic competition and competition from newer AEDs.
- Its sales are projected to decrease at an average annual rate of 2-3% over the next five years.
- The drug's market share in the phenytoin segment could shrink to approximately 15% by 2030.
- Market risks include safety profile limitations and shifting prescription preferences toward alternatives.
FAQs
Q1: How does DILANTIN compare to newer antiepileptic drugs?
A1: DILANTIN has a narrower therapeutic window and more drug interactions, leading clinicians to prefer drugs like levetiracetam and lamotrigine for safety and ease of use.
Q2: Are there any new formulations for DILANTIN in development?
A2: No significant new formulations are imminent, but extended-release versions could potentially prolong market relevance.
Q3: How significant is the impact of patent expiration on DILANTIN's sales?
A3: Since patent expiry in the 1980s, generic competition has driven down prices and sales of the brand, with current sales primarily from its branded version.
Q4: Which regions present growth opportunities for DILANTIN?
A4: Asia-Pacific shows modest growth potential due to increasing epilepsy diagnoses and limited access to newer drugs.
Q5: What are the main regulatory challenges facing DILANTIN?
A5: Safety concerns regarding side effects and drug interactions lead to regulatory scrutiny, which can restrict its use or label updates.
References
[1] Market Research Future. (2022). Global Antiepileptic Drugs Market Report.
More… ↓
