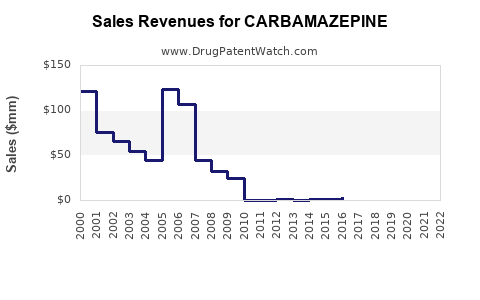
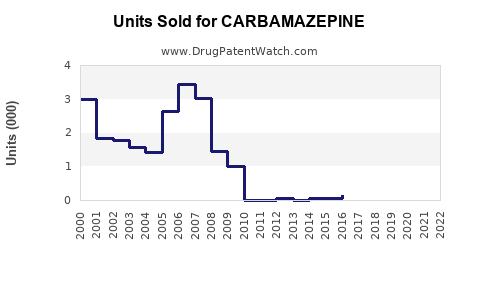
Last updated: February 19, 2026
Market Overview
Carbamazepine is an anticonvulsant and mood-stabilizing drug primarily used to treat epilepsy, trigeminal neuralgia, and bipolar disorder. It was first approved by the FDA in 1968. The drug remains a cornerstone in neurological and psychiatric treatment, with an estimated global market size driven by its formulary presence and longstanding clinical utility.
Global sales of carbamazepine were approximately $600 million in 2022. The compound is available in generic and branded formulations, with generics accounting for roughly 85% of sales. The drug's low cost, established efficacy, and FDA approval sustain its market share despite newer anticonvulsants.
Market Drivers
- Prevalence of Epilepsy and Neuralgia: Approximately 50 million people worldwide have epilepsy, with trigeminal neuralgia affecting 4-5 per 100,000 annually.
- Longstanding Generic Availability: Market penetration is high due to low-cost generics, impacting branded drug sales.
- Physician Preference: Carbamazepine remains preferred in settings with limited access to newer, more expensive medications.
- Regulatory Approvals: Approvals in emerging markets expand the geographic footprint.
- Adverse Effect Profile: Risk of hematologic and dermatologic side effects influences prescribing patterns.
Competitive Landscape
| Patent Status |
Market Share |
Key Players |
Formulations |
Pricing Dynamics |
| Patent expired |
15% |
Teva, Mylan, Sandoz |
Tablets, suspension |
Low-cost generics dominate market |
| Patent-held |
85% |
UCB (brand: Tegretol) |
Tablets, XR formulations |
Higher pricing for branded products |
Geographic Market Breakdown
| Region |
Market Share (%) |
Key Trends |
Average Annual Growth (2020–2022) |
| North America |
35% |
Stable, high penetration |
1.5% |
| Europe |
30% |
Slight decline in branded sales |
1.2% |
| Asia-Pacific |
20% |
Rapid generic adoption; emerging markets |
4.5% |
| Latin America, Africa |
15% |
Growing access, limited regulation |
3.8% |
Sales Projections (2023–2028)
| Year |
Global Market Size (USD millions) |
Compound Annual Growth Rate (CAGR) |
Major Growth Drivers |
| 2023 |
620 |
1.8% |
Increasing epilepsy diagnosis; expanding access abroad |
| 2024 |
640 |
1.8% |
Entry into new markets; use in bipolar disorder |
| 2025 |
660 |
1.8% |
Continued generic penetration; unmet needs in epilepsy |
| 2026 |
680 |
1.8% |
New formulations; improved tolerability |
| 2027 |
700 |
2.0% |
Adoption of narrow-spectrum formulations |
| 2028 |
720 |
2.1% |
Aging populations with chronic seizure conditions |
Barriers and Risks
- Drug Interactions: Carbamazepine induces hepatic enzymes, affecting metabolism of other drugs.
- Safety Concerns: Risks of agranulocytosis, Stevens-Johnson syndrome.
- Regulatory Changes: Stricter safety monitoring may mitigate use.
- Emerging Therapies: Newer anticonvulsants (e.g., levetiracetam, lamotrigine) gaining market share.
Revenue Impact of Patent and Formulary Changes
- Patent expiry in most regions led to a significant decline in premium-branded sales by 50% since 2015.
- Increased prescribing of generics has decreased per-unit revenue but expanded volume.
- Manufacturers' investment in controlled-release formulations aims to improve adherence and reduce side effects.
Future Opportunities
- Developing safer, more selective formulations.
- Expanding to pediatric populations.
- Integrating into combination therapies for complex seizure syndromes.
- Growing use in low- and middle-income countries as healthcare infrastructure improves.
Key Takeaways
- The carbamazepine market is mature with slow growth, driven mostly by expanding access in emerging markets.
- Generic competition limits revenue potential; branded sales are declining.
- New formulations targeting improved safety and tolerability could renew growth.
- Market growth remains modest at approximately 1.8–2.1% CAGR through 2028.
- A significant share of future revenues depends on regulatory landscape shifts and competitive responses.
Frequently Asked Questions
-
What is the primary indication for carbamazepine?
Treatment of epilepsy, trigeminal neuralgia, and bipolar disorder.
-
How does patent expiration affect market dynamics?
It causes branded sales decline, increases generic market share, and reduces per-unit pricing.
-
What are the main barriers to growth?
Safety concerns, drug interactions, and competition from newer agents.
-
Which regions exhibit the highest growth potential?
Asia-Pacific and Latin America, driven by increased access and healthcare infrastructure development.
-
Are there upcoming formulations or combinations anticipated to impact sales?
Yes, controlled-release formulations and combination therapies for complex seizure syndromes.
References
[1] U.S. Food and Drug Administration. (2021). Tegretol (carbamazepine) tablet, chewable tablet, suspension, extended-release capsule. FDA.
[2] MarketsandMarkets. (2022). Anticonvulsant drugs market by type, application, and region — Global forecast to 2027.
[3] IQVIA. (2022). Global prescription drug sales data.
[4] World Health Organization. (2019). Epilepsy fact sheet.
[5] European Medicines Agency. (2018). Safety communication on carbamazepine and adverse drug reactions.